 | |
| Clinical data | |
|---|---|
| Trade names | Prostetin, Roxenone |
| Other names | TSAA-291; 16β-Ethyl-19-nortestosterone; 16β-Ethylestr-4-en-17β-ol-3-one |
|
Routes of administration | Intramuscular injection [1] [2] [3] [4] |
| Drug class | Steroidal antiandrogen; Progestogen; Progestin |
| Pharmacokinetic data | |
| Bioavailability | Oral: Very low (1% in dogs) [5] |
| Elimination half-life | IM: 5.0–6.6 days. [4] [6] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C20H30O2 |
| Molar mass | 302.458 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Oxendolone, sold under the brand names Prostetin and Roxenone, is an antiandrogen and progestin medication which is used in Japan in the treatment of enlarged prostate. [7] [8] [9] [10] [11] However, this use is controversial due to concerns about its clinical efficacy. [11] Oxendolone is not effective by mouth and must be given by injection into muscle. [5] [1] [2] [3] [4]
Oxendolone is an antiandrogen, and hence is an antagonist of the androgen receptor, the biological target of androgens like testosterone and dihydrotestosterone. [12] [13] [14] [15] [6] It is also a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone. [12] [13] [14] [15] Due to its progestogenic activity, oxendolone has antigonadotropic effects. [16] [17] Oxendolone has no other important hormonal activity...
Oxendolone was introduced for medical use in 1981. [8] It is used only in Japan. [8] [11]
Medical uses
Oxendolone is used in the treatment of benign prostatic hyperplasia (BPH) in Japan. [8] [11] It has been used at a dosage of 200 mg once every 2 weeks via intramuscular injection. [17] Although it is approved for the treatment of BPH in Japan, concerns have been raised about its use for this condition due to poor efficacy seen in clinical trials. [11]
Side effects
Pharmacology
Pharmacodynamics
Oxendolone binds to the androgen receptor (Ki = 320 nM) and progesterone receptor (Ki = 20 nM) and acts as a weak but clinically relevant inhibitor of 5α-reductase ( IC50 = 1.4 μM). [12] [13] [14] [15] The relative binding affinity of oxendolone for the androgen receptor is 0.8 to 3.6% of that of metribolone. [18] [19] Oxendolone is not a silent antagonist of the androgen receptor but is rather predominantly antagonistic with weak agonistic activity; [13] for this reason, it has been described as a selective androgen receptor modulator. [20] The medication has potent antigonadotropic effects via its progestogenic activity. [16] It has been found to suppress luteinizing hormone and testosterone levels to an equivalent extent as allylestrenol and chlormadinone acetate, which are two progestins that are similarly used at high doses to treat BPH. [17]
Pharmacokinetics
The oral bioavailability of oxendolone in dogs is extremely low, 1% at most. [5] Due to its low oral bioavailability, oxendolone is administered by intramuscular injection in humans. [1] [2] [3] [4] Its elimination half-life via this route is 5.0 to 6.6 days. [4]
Chemistry
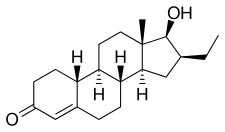
Oxendolone, also known as 16β-ethyl-19-nortestosterone or 16β-ethylestr-4-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of testosterone and 19-nortestosterone (nandrolone). [7] [8]
The acetate ester of oxendolone is known as TSAA-328, while the caproate ester of oxendolone is known as TSAA-330. [21] They were never marketed. [21]
History
Oxendolone has been marketed in Japan by Takeda since 1981. [8]
Society and culture
Generic names
Oxendolone is the generic name of the drug and its INN, USAN, and JAN. [7] [22] It is also known by its developmental code name TSAA-291. [7] [22]
Brand names
Oxendolone is or has been sold under the brand names Prostetin and Roxenone. [7] [22]
Availability
Oxendolone is marketed only in Japan. [22]
References
- ^
a
b
c Henkler G, Klotzbach M, Koch H, Müller W, Richter J (November 1982). "[Progress in the area of drug development. 15]". Die Pharmazie (in German). 37 (11): 753–765.
PMID
6131442.
[Oxendolone] has been clinically tested in Japan (weekly intramuscular injection of 200-400 mg) in prostatic hypertrophy.
- ^
a
b
c Hikichi Y, Yamaoka M, Kusaka M, Hara T (October 2015). "Selective androgen receptor modulator activity of a steroidal antiandrogen TSAA-291 and its cofactor recruitment profile". European Journal of Pharmacology. 765: 322–331.
doi:
10.1016/j.ejphar.2015.08.052.
PMID
26335395.
According to the clinical data of TSAA-291, the plasma level of TSAA-291 after weekly intramuscular administration at 400 mg/kg for 12 weeks is approximately 100 nM (Drug Information).
- ^
a
b
c Ostri P, Swartz R, Meyhoff HH, Petersen JH, Lindgård G, Frimodt-Møller C, et al. (1989). "Antiandrogenic treatment of benign prostatic hyperplasia: a placebo controlled trial". Urological Research. 17 (1): 29–33.
doi:
10.1007/bf00261046.
PMID
2466359.
S2CID
30551043.
Thirty patients were treated with weekly injections of oxendolone 200 mg during a 3 months' period, and 30 patients were allocated to placebo treatment.
- ^
a
b
c
d
e Midgley I, Fowkes AG, Darragh A, Lambe R, Chasseaud LF, Taylor T (April 1983). "The metabolic fate of the anti-androgenic agent, oxendolone, in man". Steroids. 41 (4): 521–536.
doi:
10.1016/0039-128x(83)90092-2.
PMID
6419414.
S2CID
41224726.
After intramuscular administration of 16β-ethyl-17β-hydroxy-4-[4-14C] estren-3-one (14C-oxendolone; 300 mg) to 3 human subjects, [...]
- ^ a b c Iga K (7 November 2002). "Slowly Disintegrating Buccal Mucoadhesive Pain-Tablet (S-DBMP-T) and Buccal Covered=Tablet System (BCTS)". In Rathbone MJ, Hadgraft J, Roberts MS (eds.). Modified-Release Drug Delivery Technology. CRC Press. pp. 368–. ISBN 978-0-8247-0869-6.
- ^ a b Gao W, Bohl CE, Dalton JT (September 2005). "Chemistry and structural biology of androgen receptor". Chemical Reviews. 105 (9): 3352–3370. doi: 10.1021/cr020456u. PMC 2096617. PMID 16159155.
- ^ a b c d e Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 914–. ISBN 978-1-4757-2085-3.
- ^ a b c d e f William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 2935–. ISBN 978-0-8155-1856-3.
- ^ Negwer M, Scharnow HG (2001). Organic-chemical drugs and their synonyms: (an international survey). Wiley-VCH. p. 2023. ISBN 978-3-527-30247-5.
- ^ Tan MH, Li J, Xu HE, Melcher K, Yong EL (January 2015). "Androgen receptor: structure, role in prostate cancer and drug discovery". Acta Pharmacologica Sinica. 36 (1): 3–23. doi: 10.1038/aps.2014.18. PMC 4571323. PMID 24909511.
- ^ a b c d e Ishizuka O, Nishizawa O, Hirao Y, Ohshima S (November 2002). "Evidence-based meta-analysis of pharmacotherapy for benign prostatic hypertrophy". International Journal of Urology. 9 (11): 607–612. doi: 10.1046/j.1442-2042.2002.00539.x. PMID 12534901.
- ^ a b c Mallamo JP, Juniewicz PE (8 September 1989). "New horizons in the treatment of proliferative prostatic disease". In Johns WS (ed.). Annual Reports in Medicinal Chemistry. Vol. 24. Academic Press. pp. 199–. doi: 10.1016/S0065-7743(08)60543-6. ISBN 978-0-08-058368-6.
- ^ a b c d Annual report of Shionogi Research Laboratories. 1991. pp. 76–77.
- ^ a b c Kirby RS, Christmas T (1991). "The potential value of 5-alpha-reductase inhibition in the treatment of bladder outflow obstruction due to benign prostatic hyperplasia". World Journal of Urology. 9 (1). doi: 10.1007/BF00184713. ISSN 0724-4983. S2CID 38790542.
- ^ a b c Bashirelahi N, Ganesan S, Ekiko DB, Young JD, Shida K, Yamanaka H, Takahashi E (September 1986). "Effect of 16 beta-ethyl-17 beta-hydroxy-4-estren-3-one (TSAA-291) on the binding of promegestone (R5020) and methyltrienolone (R1881) to hyperplastic and neoplastic human prostate". Journal of Steroid Biochemistry. 25 (3): 367–374. doi: 10.1016/0022-4731(86)90249-9. PMID 2430141.
- ^ a b Sudo K, Yamazaki I, Masuoka M, Nakayama R (1979). "Anti-androgen TSAA-291. IV. Effects of the anti-androgen TSAA-291 (16 beta-ethyl-17 beta-hydroxy-4-oestren-3-one) on the secretion of gonadotrophins". Acta Endocrinologica. Supplementum. 229 (3 Supplb): 53–66. doi: 10.1530/acta.0.092S053. PMID 294107.
- ^ a b c Katayama T, Umeda K, Kazama T (November 1986). "[Hormonal environment and antiandrogenic treatment in benign prostatic hypertrophy]". Hinyokika Kiyo. Acta Urologica Japonica (in Japanese). 32 (11): 1584–1589. PMID 2435122.
- ^ Dalton J, Gao W (2010). "Androgen Receptor". Nuclear Receptors: Current Concepts and Future Challenges. Proteins and Cell Regulation. Springer. pp. 143–182. doi: 10.1007/978-90-481-3303-1_6. ISBN 978-90-481-3302-4.
- ^ Wakeling AE, Furr BJ, Glen AT, Hughes LR (December 1981). "Receptor binding and biological activity of steroidal and nonsteroidal antiandrogens". Journal of Steroid Biochemistry. 15: 355–359. doi: 10.1016/0022-4731(81)90297-1. PMID 7339263.
- ^ Hikichi Y, Yamaoka M, Kusaka M, Hara T (October 2015). "Selective androgen receptor modulator activity of a steroidal antiandrogen TSAA-291 and its cofactor recruitment profile". European Journal of Pharmacology. 765: 322–331. doi: 10.1016/j.ejphar.2015.08.052. PMID 26335395.
- ^ a b Masuoka M, Masaki T, Yamazaki I, Hori T, Nakayama R (1979). "Anti-androgen TSAA-291. III. Hormonal spectra of anti-androgen TSAA-291 (16 beta-ethyl-17 beta-hydroxy-4-oestren-3-one) and its derivatives". Acta Endocrinologica. Supplementum. 229: 36–52. doi: 10.1530/acta.0.092s036. PMID 294106.
- ^ a b c d "Oxandrolone Uses, Side Effects & Warnings". Drugs.com.