 | |
| Clinical data | |
|---|---|
| Other names | RU-40555; 17β-Hydroxy-11β-[4-[methyl(1-methylethyl)amino]phenyl]-17α-(1-propyn-1-yl)estra-4,9-dien-3-one |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
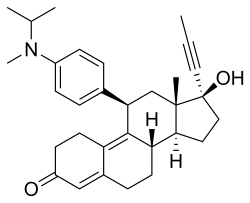
| Formula | C31H39NO2 |
| Molar mass | 457.658 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Toripristone ( INN; developmental code name RU-40555) is a synthetic, steroidal antiglucocorticoid as well as antiprogestogen which was never marketed. [1] [2] It is reported as a potent and highly selective antagonist of the glucocorticoid receptor (GR) (Ki = 2.4 nM), [3] [4] though it also acts as an antagonist of the progesterone receptor (PR). [5] [6] The pharmacological profile of toripristone is said to be very similar to that of mifepristone, except that toripristone does not bind to orosomucoid (α1-acid glycoprotein). [5] The drug has been used to study the hypothalamic-pituitary-adrenal axis and has been used as a radiotracer for the GR. [4] [3] Its INN was given in 1990. [2]
See also
References
- ^ Hill RA, Makin HL, Kirk DN, Murphy GM (23 May 1991). "Toripristone". Dictionary of Steroids. CRC Press. pp. 101–. ISBN 978-0-412-27060-4.
- ^ a b "International Nonproprietary Names for Pharmaceutical Substances" (PDF). WHO Drug Information. 3 (2): 18. 1989. Archived from the original (PDF) on 4 March 2016.
- ^ a b Steiniger B, Kniess T, Bergmann R, Pietzsch J, Wuest FR (June 2008). "Radiolabeled glucocorticoids as molecular probes for imaging brain glucocorticoid receptors by means of positron emission tomography (PET)". Mini Reviews in Medicinal Chemistry. 8 (7): 728–739. doi: 10.2174/138955708784567403. PMID 18537728.
- ^ a b Bachmann CG, Linthorst AC, Holsboer F, Reul JM (June 2003). "Effect of chronic administration of selective glucocorticoid receptor antagonists on the rat hypothalamic-pituitary-adrenocortical axis". Neuropsychopharmacology. 28 (6): 1056–1067. doi: 10.1038/sj.npp.1300158. PMID 12700716.
- ^ a b Philibert D, Costerousse G, Gaillard-Moguilewsky M, Nedelec L, Nique F, Tournemine C, Teutsch G (1991). "From RU 38486 towards Dissociated Antiglucocorticoid and Antiprogesterone". Antihormones in Health and Disease. Frontiers of Hormone Research. Vol. 19. pp. 1–17. doi: 10.1159/000419634. ISBN 978-3-8055-5297-4. ISSN 0301-3073.
- ^ Fu J, Hu L, Huang W, Zhu H, Wang Q, He F, Xie L, Gan, Huang W (2012). Huang W (ed.). "Progesterone receptor antagonists and progesterone receptor modulators for endometriosis". Cochrane Database of Systematic Reviews (5): CD009881. doi: 10.1002/14651858.CD009881.