 | |
| Clinical data | |
|---|---|
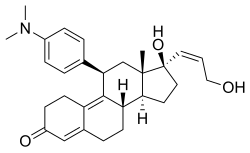
| Other names | ZK-98734; ZK-734; 11β-(4-(Dimethylamino)phenyl)-17β-hydroxy-17α-((Z)-3-hydroxypropenyl)estra-4,9-dien-3-one |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C29H37NO3 |
| Molar mass | 447.619 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Lilopristone ( INN) (developmental code names ZK-98734, ZK-734) is a synthetic, steroidal antiprogestogen with additional antiglucocorticoid activity which was developed by Schering and was patented in 1985. [1] [2] [3] [4] It is described as an abortifacient and endometrial contraceptive. [1] [4] [5] The drug differs from mifepristone only in the structure of its C17α side chain, and is said to have much reduced antiglucocorticoid activity in comparison. [6]
See also
References
- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 733–. ISBN 978-1-4757-2085-3.
- ^ Rao KA (November 2009). Textbook of Gynaecology. Elsevier India. pp. 187–. ISBN 978-81-312-1526-5.
- ^ Baird DT, Schütz G, Krattenmacher R (9 March 2013). Organ-Selective Actions of Steroid Hormones. Springer Science & Business Media. pp. 108–. ISBN 978-3-662-09153-1.
- ^ a b Milne GW (8 May 2018). Drugs: Synonyms and Properties: Synonyms and Properties. Taylor & Francis. pp. 23–. ISBN 978-1-351-78989-9.
- ^ Deshpande H (12 February 2016). Practical Management of Ovulation Induction. JP Medical Ltd. pp. 29–. ISBN 978-93-5250-028-4.
-
^ Van Look PF, Pérez-Palacios G, World Health Organization (1994).
Contraceptive research and development, 1984 to 1994: the road from Mexico City to Cairo and beyond. Oxford University Press. p. 169.
ISBN
978-0-19-563630-7.
[...] lilopristone, which differs from mifepristone only in the structure of the 17a side chain, is said to have a much reduced antiglucocorticoid activity (Neef et al., 1984).
Further reading
- Puri CP, Katkam RR, D'Souza A, Elger WA, Patil RK (September 1990). "Effects of progesterone antagonist, lilopristone (ZK 98.734), on induction of menstruation, inhibition of nidation, and termination of pregnancy in bonnet monkeys". Biology of Reproduction. 43 (3): 437–43. doi: 10.1095/biolreprod43.3.437. PMID 2271724.
- Puri CP, Patil RK, Kholkute SD, Elger WA, Swamy XR (July 1989). "Progesterone antagonist lilopristone: a potent abortifacient in the common marmoset". American Journal of Obstetrics and Gynecology. 161 (1): 248–53. doi: 10.1016/0002-9378(89)90274-3. PMID 2502015.