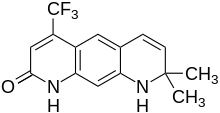
 | |
| Clinical data | |
|---|---|
| Other names | 1,2,3,4-Tetrahydro-2,2-dimethyl-6-trifluoromethyl-8-pyridono[5,6-g]quinoline |
| Drug class | Nonsteroidal antiandrogen |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C15H13F3N2O |
| Molar mass | 294.277 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
LG-120907 is a nonsteroidal antiandrogen (NSAA) of the quinoline group which was developed by Ligand Pharmaceuticals along with selective androgen receptor modulators (SARMs) like LG-121071 and was never marketed. [1] [2] [3] [4] The drug is a high- affinity antagonist of the androgen receptor (AR) with a Ki value of 26 nM and has been found to inhibit growth of the ventral prostate and seminal vesicles in male rats without increasing circulating levels of luteinizing hormone or testosterone. [1] [2] [3] However, this tissue selectivity has not been assessed in humans. [5] LG-120907 is orally active and shows greater oral potency than the aryl propionamide NSAA flutamide. [1] [3] [4]
The 7- fluoro derivative of LG-120907, LG-105, is also a potent NSAA, and appears to possess greater potency in comparison. [3] Conversely, the 6- ethyl, 8-di desmethyl analogue of LG-120907, LG-121071, is a SARM with potent androgenic activity equivalent to that of dihydrotestosterone (DHT). [1] [3]
References
- ^ a b c d Chengalvala M, Oh T, Roy AK (2005). "Selective androgen receptor modulators". Expert Opinion on Therapeutic Patents. 13 (1): 59–66. doi: 10.1517/13543776.13.1.59. ISSN 1354-3776. S2CID 219188263.
- ^ a b Mohler ML, Nair VA, Hwang DJ, Rakov IM, Patil R, Miller DD (2005). "Nonsteroidal tissue selective androgen receptor modulators: a promising class of clinical candidates". Expert Opinion on Therapeutic Patents. 15 (11): 1565–1585. doi: 10.1517/13543776.15.11.1565. ISSN 1354-3776. S2CID 96279138.
- ^ a b c d e Gao W, Kim J, Dalton JT (August 2006). "Pharmacokinetics and pharmacodynamics of nonsteroidal androgen receptor ligands". Pharmaceutical Research. 23 (8): 1641–1658. doi: 10.1007/s11095-006-9024-3. PMC 2072875. PMID 16841196.
- ^ a b Nyrönen TH, Söderholm AA (January 2010). "Structural basis for computational screening of non-steroidal androgen receptor ligands". Expert Opinion on Drug Discovery. 5 (1): 5–20. doi: 10.1517/17460440903468680. PMID 22823968. S2CID 21630468.
- ^ Hoffmann J, Sommer A (30 January 2007). "Anti-hormone therapy: principles of endocrine therapy of cancer.". In Bradbury R (ed.). Cancer. Springer Science & Business Media. pp. 19-82 (67). ISBN 978-3-540-33120-9.