From Wikipedia, the free encyclopedia
Chemical compound
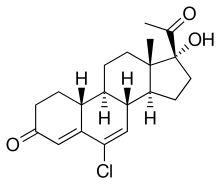
Amadinone
Other names 6-Chloro-17α-hydroxy-19-norpregna-4,6-dione
(1S,2R,10R,11S,14R,15S)-14-acetyl-8-chloro-14-hydroxy-15-methyltetracyclo[8.7.0.02,7 .011,15 ]heptadeca-6,8-dien-5-one
CAS Number
PubChem
CID
ChemSpider
UNII
CompTox Dashboard (
EPA )
Formula C 20 H 25 Cl O 3
Molar mass −1 3D model (
JSmol )
CC(=O)[C@]1(CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2C=C(C4=CC(=O)CC[C@H]34)Cl)C)O
InChI=1S/C20H25ClO3/c1-11(22)20(24)8-6-17-15-10-18(21)16-9-12(23)3-4-13(16)14(15)5-7-19(17,20)2/h9-10,13-15,17,24H,3-8H2,1-2H3/t13-,14-,15-,17+,19+,20+/m1/s1
Key:ANJGIFXUFSBZPX-WLCXVKOPSA-N
Amadinone (
INN ), also known as 19-norchlormadinone , is a
steroidal
progestin of the
19-norprogesterone and
17α-hydroxyprogesterone groups that was synthesized and characterized in 1968 but was never marketed.
[1]
[2] It has
antigonadotropic properties, and for this reason, is a functional
antiandrogen .
[3]
[4] An
acetate
ester ,
amadinone acetate , also exists, but similarly was never marketed.
[1]
See also References
^
a b Elks J (14 November 2014).
The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies ISBN
978-1-4757-2085-3
^ William Andrew Publishing (22 October 2013).
Pharmaceutical Manufacturing Encyclopedia, 3rd Edition ISBN
978-0-8155-1856-3
^ Hughes A, Hasan SH, Oertel GW, Voss HE, Bahner F, Neumann G, Steinbeck H, Gräf KJ, Brotherton J, Horn HJ, Wagner RK (27 November 2013).
Androgens II and Antiandrogens / Androgene II und Antiandrogene ISBN
978-3-642-80859-3
^ Kent JR, Hill M, Huix FJ, Segre EJ (1972). "Seminal acid phosphatase content in the clinical bioassay of androgens and antiandrogens". Clinical Pharmacology and Therapeutics . 13 (2): 205–11.
doi :
10.1002/cpt1972132205 .
PMID
5017374 .
S2CID
40886901 .
PR Tooltip Progesterone receptor
Agonists
Testosterone derivatives: Progestins:
6,6-Difluoronorethisterone
6,6-Difluoronorethisterone acetate
17α-Allyl-19-nortestosterone
Allylestrenol
Altrenogest
Chloroethynylnorgestrel
Cingestol
Danazol
Desogestrel
Dienogest
Ethinylandrostenediol
Ethisterone
Ethynerone
Etonogestrel
Etynodiol
Etynodiol diacetate
Gestodene
Gestrinone
Levonorgestrel
Levonorgestrel esters (e.g.,
levonorgestrel butanoate )
Lynestrenol
Lynestrenol phenylpropionate
Metynodiol
Metynodiol diacetate
Norelgestromin
Norethisterone (norethindrone)
Norethisterone esters (e.g.,
norethisterone acetate ,
norethisterone enanthate )
Noretynodrel
Norgesterone
Norgestimate
Norgestrel
Norgestrienone
Norvinisterone
Oxendolone
Quingestanol
Quingestanol acetate
Tibolone
Tigestol
Tosagestin ; Anabolic–androgenic steroids:
11β-Methyl-19-nortestosterone
11β-Methyl-19-nortestosterone dodecylcarbonate
19-Nor-5-androstenediol
19-Nor-5-androstenedione
19-Nordehydroepiandrosterone
Bolandiol
Bolandiol dipropionate
Bolandione
Dimethisterone
Dienedione
Dienolone
Dimethandrolone
Dimethandrolone buciclate
Dimethandrolone dodecylcarbonate
Dimethandrolone undecanoate
Dimethyldienolone
Dimethyltrienolone
Ethyldienolone
Ethylestrenol (ethylnandrol)
Methyldienolone
Metribolone (R-1881)
Methoxydienone (methoxygonadiene)
Mibolerone
Nandrolone
Nandrolone esters (e.g.,
nandrolone decanoate ,
nandrolone phenylpropionate )
Norethandrolone
Normethandrone (methylestrenolone, normethandrolone, normethisterone)
RU-2309
Tetrahydrogestrinone
Trenbolone (trienolone)
Trenbolone esters (e.g.,
trenbolone acetate ,
trenbolone enanthate )
Trendione
Trestolone
Trestolone acetate Mixed
SPRMs Tooltip Selective progesterone receptor modulators ) Antagonists
mPR Tooltip Membrane progesterone receptor
PAQR Tooltip Progestin and adipoQ receptor )