From Wikipedia, the free encyclopedia
Chemical compound
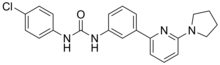
PSNCBAM-1
 |
|
1-(4-chlorophenyl)-3-(3-(6-(pyrrolidin-1-yl)pyridin-2-yl)phenyl)urea
|
|
CAS Number | |
|---|
|
PubChem
CID | |
|---|
|
ChemSpider | |
|---|
|
UNII | |
|---|
|
CompTox Dashboard (
EPA) | |
|---|
|
|
Formula | C22H21ClN4O |
|---|
|
Molar mass | 392.89 g·mol−1 |
|---|
| 3D model (
JSmol) | |
|---|
Clc1ccc(NC(=O)Nc2cccc(c2)c3cccc(n3)N4CCCC4)cc1
|
InChI=1S/C22H21ClN4O/c23-17-9-11-18(12-10-17)24-22(28)25-19-6-3-5-16(15-19)20-7-4-8-21(26-20)27-13-1-2-14-27/h3-12,15H,1-2,13-14H2,(H2,24,25,28) Key:HDAYFSFWIPRJSO-UHFFFAOYSA-N
|
PSNCBAM-1 is a negative
allosteric modulator of the
cannabinoid
CB1
receptor.
[1]
[2]
See also
References
-
^ German N, Decker AM, Gilmour BP, Gay EA, Wiley JL, Thomas BF, Zhang Y (September 2014).
"Diarylureas as Allosteric Modulators of the Cannabinoid CB1 Receptor: Structure–Activity Relationship Studies on 1-(4-Chlorophenyl)-3-{3-[6-(pyrrolidin-1-yl)pyridin-2-yl]phenyl}urea (PSNCBAM-1)". Journal of Medicinal Chemistry. 57 (18): 7758–7769.
doi:
10.1021/jm501042u.
PMC
4175001.
PMID
25162172.
-
^ Horswill JG, Bali U, Shaaban S, Keily JF, Jeevaratnam P, Babbs AJ, Reynet C, Wong Kai In P (November 2007).
"PSNCBAM-1, a novel allosteric antagonist at cannabinoid CB1 receptors with hypophagic effects in rats". British Journal of Pharmacology. 152 (5): 805–814.
doi:
10.1038/sj.bjp.0707347.
PMC
2190018.
PMID
17592509.
|
|---|
Phytocannabinoids
(
comparison) | | Cannabibutols | |
|---|
| Cannabichromenes | |
|---|
| Cannabicyclols | |
|---|
| Cannabidiols | |
|---|
| Cannabielsoins | |
|---|
| Cannabigerols | |
|---|
| Cannabiphorols | |
|---|
| Cannabinols | |
|---|
| Cannabitriols | |
|---|
| Cannabivarins | |
|---|
| Delta-8-tetrahydrocannabinols | |
|---|
| Delta-9-tetrahydrocannabinols | |
|---|
| Delta-10-Tetrahydrocannabinols | |
|---|
| Miscellaneous cannabinoids | |
|---|
| Active metabolites | |
|---|
|
|---|
|
Endocannabinoids | |
|---|
Synthetic
cannabinoid
receptor
agonists /
neocannabinoids | Classical cannabinoids
(dibenzopyrans) | |
|---|
Non-classical
cannabinoids | |
|---|
| Adamantoylindoles | |
|---|
| Benzimidazoles | |
|---|
| Benzoylindoles | |
|---|
| Cyclohexylphenols | |
|---|
|
Eicosanoids | |
|---|
|
Hydrocarbons | |
|---|
| Indazole carboxamides | |
|---|
Indazole-3-
carboxamides | |
|---|
| Indole-3-carboxamides | |
|---|
| Indole-3-carboxylates | |
|---|
| Naphthoylindazoles | |
|---|
|
Naphthoylindoles | |
|---|
| Naphthoylpyrroles | |
|---|
| Naphthylmethylindenes | |
|---|
| Naphthylmethylindoles | |
|---|
| Phenylacetylindoles | |
|---|
| Pyrazolecarboxamides | |
|---|
| Pyrrolobenzoxazines | |
|---|
| Quinolinyl esters | |
|---|
Tetramethylcyclo-
propanoylindazoles | |
|---|
Tetramethylcyclo-
propanoylindoles | |
|---|
Tetramethylcyclo-
propylindoles | |
|---|
| Others | |
|---|
|
|---|
|
Allosteric
CBRTooltip Cannabinoid receptor
ligands | |
|---|
Endocannabinoid
enhancers
(inactivation inhibitors) | |
|---|
Anticannabinoids
(antagonists/inverse
agonists/antibodies) | |
|---|
|
|
|---|
Receptor
(
ligands) | |
CB1Tooltip Cannabinoid receptor type 1 | Agonists
(abridged,
full list) | |
|---|
| Inverse agonists | |
|---|
| Antagonists | |
|---|
|
|---|
|
CB2Tooltip Cannabinoid receptor type 2 | | Agonists |
-
2-AG
-
2-AGE (noladin ether)
-
3,3'-Diindolylmethane
-
4-O-Methylhonokiol
-
α-Amyrin · β-Amyrin
-
A-796,260
-
A-834,735
-
A-836,339
-
AM-1172
-
AM-1221
-
AM-1235
-
AM-1241
-
AM-2232
-
Anandamide
-
AZ-11713908
-
Cannabinol
-
Caryophyllene
-
CB-13
-
CBS-0550
-
CP 55,940
-
GW-405,833 (L-768,242)
-
GW-842,166X
-
HU-308
-
JTE 7-31
-
JWH-007
-
JWH-015
-
JWH-018
-
JWH-73
-
JWH-133
-
L-759,633
-
L-759,656
-
Lenabasum (anabasum)
-
Magnolol
-
MDA-19
-
Nabitan
-
NADA
-
Olorinab (APD-371)
-
PF-03550096
-
S-444,823
-
SER-601
-
Serinolamide A
-
UR-144
-
Tedalinab
-
THC (dronabinol)
-
THCV
-
Tetrahydromagnolol
-
Virodhamine
|
|---|
| Antagonists | |
|---|
|
|---|
NAGly
(
GPR18) | |
|---|
|
GPR55 | |
|---|
|
GPR119 | |
|---|
|
|---|
Transporter
(
modulators) | |
eCBTsTooltip Endocannabinoid transporter | |
|---|
|
|---|
Enzyme
(
modulators) | |
|---|
| Others |
- Others:
2-PG (directly potentiates activity of 2-AG at CB1 receptor)
-
ARN-272 (FAAH-like anandamide transporter inhibitor)
|
|---|
|
