Receptor tyrosine-protein kinase erbB-2 is a protein that normally resides in the membranes of cells and is encoded by the ERBB2 gene. ERBB is abbreviated from erythroblastic oncogene B, a gene originally isolated from the avian genome. The human protein is also frequently referred to as HER2 (human epidermal growth factor receptor 2) or CD340 ( cluster of differentiation 340). [5] [6] [7]
HER2 is a member of the human epidermal growth factor receptor (HER/EGFR/ERBB) family. But contrary to other members of the ERBB family, HER2 does not directly bind ligand. HER2 activation results from heterodimerization with another ERBB member or by homodimerization when HER2 concentration are high, for instance in cancer. [8] Amplification or over-expression of this oncogene has been shown to play an important role in the development and progression of certain aggressive types of breast cancer. In recent years the protein has become an important biomarker and target of therapy for approximately 30% of breast cancer patients. [9]
Name
HER2 is so named because it has a similar structure to human epidermal growth factor receptor, or HER1. Neu is so named because it was derived from a rodent glioblastoma cell line, a type of neural tumor. ErbB-2 was named for its similarity to ErbB (avian erythroblastosis oncogene B), the oncogene later found to code for EGFR. Molecular cloning of the gene showed that HER2, Neu, and ErbB-2 are all encoded by the same orthologs. [10]
Gene
ERBB2, a known proto-oncogene, [11] is located at the long arm of human chromosome 17 (17q12). [12]
Function
The ErbB family consists of four individual plasma membrane-bound receptor tyrosine kinases. One of which is erbB-2, and the other members being erbB-1, erbB-3 (neuregulin-binding; lacks kinase domain), and erbB-4. All four contain an extracellular ligand binding domain, a transmembrane domain, and an intracellular domain that can interact with a multitude of signaling molecules and exhibit both ligand-dependent and ligand-independent activity. Notably, no ligands for HER2 have yet been identified. [13] [14] HER2 can heterodimerise with any of the other three receptors and is considered to be the preferred dimerisation partner of the other ErbB receptors. [15]
Dimerisation results in the autophosphorylation of tyrosine residues within the cytoplasmic domain of the receptors and initiates a variety of signaling pathways.
Signal transduction
Signaling pathways activated by HER2 include: [16]
- mitogen-activated protein kinase (MAPK)
- phosphoinositide 3-kinase (PI3K/Akt)
- phospholipase C γ
- protein kinase C (PKC)
- Signal transducer and activator of transcription (STAT)
In summary, signaling through the ErbB family of receptors promotes cell proliferation and opposes apoptosis, and therefore must be tightly regulated to prevent uncontrolled cell growth from occurring.
Clinical significance
Cancer
Amplification, also known as the over-expression of the ERBB2 gene, occurs in approximately 15-30% of breast cancers. [9] [17] HER2-positive breast cancers are well established as being associated with increased disease recurrence and a poor prognosis compared with other identifiably genetically distinct breast cancers with other known, or lack thereof, genetic markers that are thought to be associated with other breast cancers; however, drug agents targeting HER2 in breast cancer have significantly and positively altered the otherwise poor prognosis of the historically problematic difficulties associated with HER2-positive breast cancer. [18] Over-expression is also known to occur in ovarian, [19] stomach, adenocarcinoma of the lung [20] and aggressive forms of uterine cancer, such as uterine serous endometrial carcinoma, [21] [22] e.g. HER2 is over-expressed in approximately 7-34% of patients with gastric cancer [23] [24] and in 30% of salivary duct carcinomas. [25]
HER2 is colocalised and most of the time, coamplified with the gene GRB7, which is a proto-oncogene associated with breast, testicular germ cell, gastric, and esophageal tumours.
HER2 proteins have been shown to form clusters in cell membranes that may play a role in tumorigenesis. [26] [27]
Evidence has also implicated HER2 signaling in resistance to the EGFR-targeted cancer drug cetuximab. [28]
The high expression of HER2 correlates with better survival in esophageal adenocarcinoma. [29]
The high amplification of HER2 copy number positively contributes to the survival time of gastric cardia adenocarcinoma patients. [30]
Mutations
Furthermore, diverse structural alterations have been identified that cause ligand-independent firing of this receptor, doing so in the absence of receptor over-expression. HER2 is found in a variety of tumours and some of these tumours carry point mutations in the sequence specifying the transmembrane domain of HER2. Substitution of a valine for a glutamic acid or a glutamine in the transmembrane domain can result in the constitutive dimerisation of this protein in the absence of a ligand. [31]
HER2 mutations have been found in non-small-cell lung cancers (NSCLC) and can direct treatment. [32]
As a drug target
HER2 is the target of the monoclonal antibody trastuzumab (marketed as Herceptin). Trastuzumab is effective only in cancers where HER2 is over-expressed. One year of trastuzumab therapy is recommended for all patients with HER2-positive breast cancer who are also receiving chemotherapy. [33] Twelve months of trastuzumab therapy is optimal. Randomized trials have demonstrated no additional benefit beyond 12 months, whereas 6 months has been shown to be inferior to 12. Trastuzumab is administered intravenously weekly or every 3 weeks. [34]
An important downstream effect of trastuzumab binding to HER2 is an increase in p27, a protein that halts cell proliferation. [35] Another monoclonal antibody, Pertuzumab, which inhibits dimerisation of HER2 and HER3 receptors, was approved by the FDA for use in combination with trastuzumab in June 2012.
As of November 2015, there are a number of ongoing and recently completed clinical trials of novel targeted agents for HER2+ metastatic breast cancer, e.g. margetuximab. [36]
Additionally, NeuVax ( Galena Biopharma) is a peptide-based immunotherapy that directs "killer" T cells to target and destroy cancer cells that express HER2. It has entered phase 3 clinical trials.
It has been found that patients with ER+ ( Estrogen receptor positive)/HER2+ compared with ER-/HER2+ breast cancers may actually benefit more from drugs that inhibit the PI3K/AKT molecular pathway. [37]
Over-expression of HER2 can also be suppressed by the amplification of other genes. Research is currently being conducted to discover which genes may have this desired effect.
The expression of HER2 is regulated by signaling through estrogen receptors. Normally, estradiol and tamoxifen acting through the estrogen receptor down-regulate the expression of HER2. However, when the ratio of the coactivator AIB-3 exceeds that of the corepressor PAX2, the expression of HER2 is upregulated in the presence of tamoxifen, leading to tamoxifen-resistant breast cancer. [38] [39]
Among approved anti-HER2 therapeutics are also tyrosine kinase inhibitors ( Lapatinib, Neratinib, and Tucatinib) and antibody-drug conjugates ( ado-trastuzumab emtansine and trastuzumab deruxtecan). [40]

Diagnostics
HER2 testing is performed on breast biopsy of breast cancer patients to assess prognosis and to determine suitability for trastuzumab therapy. It is important that trastuzumab is restricted to HER2-positive individuals as it is expensive and has been associated with cardiac toxicity. [41] For HER2-positive tumors, the benefits of trastuzumab clearly outweigh the risks.
Tests are usually performed on breast biopsy samples obtained by either fine-needle aspiration, core needle biopsy, vacuum-assisted breast biopsy, or surgical excision.
Immunohistochemistry (IHC) is generally used to measure the amount of HER2 protein present in the sample, with fluorescence in situ hybridisation (FISH) being used on samples that are equivocal in IHC. However, in several locations, FISH is used initially, followed by IHC in equivocal cases. [42]
Immunohistochemistry
By immunohistochemistry, the sample is given a score based on the cell membrane staining pattern.
| Score [43] [44] | Pattern [45] | Status [43] [44] |
|---|---|---|
| 0 | Either:
[45]
|
HER2 negative (not present) |
| 1+ | Incomplete membrane staining that is faint or barely perceptible and within >10% of the invasive tumor cells. [45] | |
| 2+ | Weak to moderate complete membrane staining observed in >10% of tumor cells. [45] | Borderline/Equivocal |
| 3+ | Circumferential membrane staining that is complete, intense, and in >10% of tumor cells. [45] | HER2 positive |
Micrographs showing each score: [46]
-
0
-
1+
-
2+
-
3+
Fluorescence in situ hybridisation
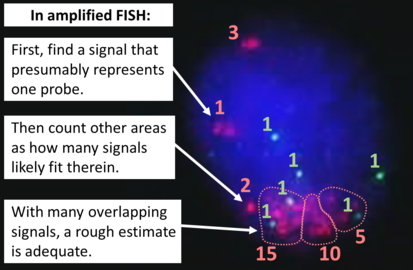
FISH can be used to measure the number of copies of the gene which are present and is thought to be more reliable than immunohistochemistry. [47] It usually uses chromosome enumeration probe 17 (CEP17) to count the amount of chromosomes. Hence, the HER2/CEP17 ratio reflects any amplification of HER2 as compared to the number of chromosomes. The signals of 20 cells are usually counted.
-
This cell displays 2 signals of HER2 (red) and 3 signals of CEP17 (green)
-
Two signals that are closer to each other than the signal diameter count as one.
-
One of these signals is too faint, and is presumably debris.
-
Cells with only one type of signal are excluded from the count.
-
Overlapping cells are also excluded from the count.
-
A yellow signal counts as one red and one green (which are overlapping)
-
Algorithm for the evaluation of HER2 on fluorescence in situ hybridization (FISH). [48]
| HER2/CEP17 ratio | |||
|---|---|---|---|
| ≥2.0 | <2.0 | ||
| Average HER2 copy number per cell | ≥4.0 | HER2 positive | Additional work-up required |
| <4.0 | Additional work-up required | HER2 negative | |
If the initial HER2 result is negative for a needle biopsy of a primary breast cancer, a new HER2 test may be performed on the subsequent breast excision. [48]
Serum
The extracellular domain of HER2 can be shed from the surface of tumour cells and enter the circulation. Measurement of serum HER2 by enzyme-linked immunosorbent assay ( ELISA) offers a far less invasive method of determining HER2 status than a biopsy and consequently has been extensively investigated. Results so far have suggested that changes in serum HER2 concentrations may be useful in predicting response to trastuzumab therapy. [49] However, its ability to determine eligibility for trastuzumab therapy is less clear. [50]
Interactions
HER2/neu has been shown to interact with:
See also
- SkBr3 Cell Line, over-expresses HER2
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000141736 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000062312 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "ERBB2 erb-b2 receptor tyrosine kinase 2 [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2016-06-14.
- ^ "ERBB2". Genetics Home Reference. Retrieved 2016-06-19.
- ^ Barh D, Gunduz M (2015-01-22). Noninvasive Molecular Markers in Gynecologic Cancers. CRC Press. p. 427. ISBN 978-1-4665-6939-3.
- ^ Hsu JL, Hung MC (2016). "The role of HER2, EGFR, and other receptor tyrosine kinases in breast cancer". Cancer and Metastasis Reviews. 35 (4): 575–588. doi: 10.1007/s10555-016-9649-6. PMC 5215954. PMID 27913999.
- ^ a b Mitri Z, Constantine T, O'Regan R (2012). "The HER2 Receptor in Breast Cancer: Pathophysiology, Clinical Use, and New Advances in Therapy". Chemotherapy Research and Practice. 2012: 743193. doi: 10.1155/2012/743193. PMC 3539433. PMID 23320171.
- ^ Coussens L, Yang-Feng TL, Liao YC, Chen E, Gray A, McGrath J, et al. (December 1985). "Tyrosine kinase receptor with extensive homology to EGF receptor shares chromosomal location with neu oncogene". Science. 230 (4730): 1132–1139. Bibcode: 1985Sci...230.1132C. doi: 10.1126/science.2999974. PMID 2999974.
- ^ Bargmann CI, Hung MC, Weinberg RA (January 1986). "The neu oncogene encodes an epidermal growth factor receptor-related protein". Nature. 319 (6050): 226–230. doi: 10.1038/319226a0. ISSN 0028-0836.
- ^ Fukushige SI, Matsubara KI, Yoshida M, Sasaki M, Suzuki T, Semba K, et al. (1986-03-01). "Localization of a Novel v- erb B-Related Gene, c- erb B-2, on Human Chromosome 17 and Its Amplification in a Gastric Cancer Cell Line". Molecular and Cellular Biology. 6 (3): 955–958. doi: 10.1128/mcb.6.3.955-958.1986. ISSN 1098-5549.
- ^ Keshamouni VG, Mattingly RR, Reddy KB (June 2002). "Mechanism of 17-beta-estradiol-induced Erk1/2 activation in breast cancer cells. A role for HER2 AND PKC-delta". The Journal of Biological Chemistry. 277 (25): 22558–22565. doi: 10.1074/jbc.M202351200. PMID 11960991.
- ^ Rusnak DW, Affleck K, Cockerill SG, Stubberfield C, Harris R, Page M, et al. (October 2001). "The characterization of novel, dual ErbB-2/EGFR, tyrosine kinase inhibitors: potential therapy for cancer". Cancer Research. 61 (19): 7196–7203. PMID 11585755.
- ^ Olayioye MA (2001). "Update on HER-2 as a target for cancer therapy: intracellular signaling pathways of ErbB2/HER-2 and family members". Breast Cancer Research. 3 (6): 385–389. doi: 10.1186/bcr327. PMC 138705. PMID 11737890.
- ^ Roy V, Perez EA (November 2009). "Beyond trastuzumab: small molecule tyrosine kinase inhibitors in HER-2-positive breast cancer". The Oncologist. 14 (11): 1061–1069. doi: 10.1634/theoncologist.2009-0142. PMID 19887469. S2CID 207242039.
- ^ Burstein HJ (October 2005). "The distinctive nature of HER2-positive breast cancers". The New England Journal of Medicine. 353 (16): 1652–1654. doi: 10.1056/NEJMp058197. PMID 16236735. S2CID 26675265.
- ^ Tan M, Yu D (2007). "Molecular Mechanisms of ErbB2-Mediated Breast Cancer Chemoresistance". Breast Cancer Chemosensitivity. Advances in Experimental Medicine and Biology. Vol. 608. pp. 119–29. doi: 10.1007/978-0-387-74039-3_9. ISBN 978-0-387-74037-9. PMID 17993237.
- ^ Kumar V, Abbas A, Aster J (2013). Robbins basic pathology. Philadelphia: Elsevier/Saunders. p. 697. ISBN 978-1-4377-1781-5.
- ^ Kumar V, Abbas A, Aster J (2013). Robbins basic pathology. Philadelphia: Elsevier/Saunders. p. 179. ISBN 978-1-4377-1781-5.
- ^ Santin AD, Bellone S, Roman JJ, McKenney JK, Pecorelli S (August 2008). "Trastuzumab treatment in patients with advanced or recurrent endometrial carcinoma overexpressing HER2/neu". International Journal of Gynaecology and Obstetrics. 102 (2): 128–131. doi: 10.1016/j.ijgo.2008.04.008. PMID 18555254. S2CID 25674060.
- ^ Buza N, Roque DM, Santin AD (March 2014). "HER2/neu in Endometrial Cancer: A Promising Therapeutic Target With Diagnostic Challenges". Archives of Pathology & Laboratory Medicine. 138 (3): 343–350. doi: 10.5858/arpa.2012-0416-RA. PMID 24576030.
- ^ Rüschoff J, Hanna W, Bilous M, Hofmann M, Osamura RY, Penault-Llorca F, et al. (May 2012). "HER2 testing in gastric cancer: a practical approach". Modern Pathology. 25 (5): 637–650. doi: 10.1038/modpathol.2011.198. PMID 22222640.
- ^ Meza-Junco J, Au HJ, Sawyer MB (March 2011). "Critical appraisal of trastuzumab in treatment of advanced stomach cancer". Cancer Management and Research. 3 (3): 57–64. doi: 10.2147/CMAR.S12698. PMC 3085240. PMID 21556317.
- ^ Chiosea SI, Williams L, Griffith CC, Thompson LD, Weinreb I, Bauman JE, et al. (June 2015). "Molecular characterization of apocrine salivary duct carcinoma". The American Journal of Surgical Pathology. 39 (6): 744–752. doi: 10.1097/PAS.0000000000000410. PMID 25723113. S2CID 34106002.
- ^ Nagy P, Jenei A, Kirsch AK, Szöllosi J, Damjanovich S, Jovin TM (June 1999). "Activation-dependent clustering of the erbB2 receptor tyrosine kinase detected by scanning near-field optical microscopy". Journal of Cell Science. 112 ( Pt 11) (11): 1733–1741. doi: 10.1242/jcs.112.11.1733. hdl: 2437/166028. PMID 10318765.
- ^ Kaufmann R, Müller P, Hildenbrand G, Hausmann M, Cremer C (April 2011). "Analysis of Her2/neu membrane protein clusters in different types of breast cancer cells using localization microscopy". Journal of Microscopy. 242 (1): 46–54. doi: 10.1111/j.1365-2818.2010.03436.x. PMID 21118230. S2CID 2119158.
- ^ Yonesaka K, Zejnullahu K, Okamoto I, Satoh T, Cappuzzo F, Souglakos J, et al. (September 2011). "Activation of ERBB2 signaling causes resistance to the EGFR-directed therapeutic antibody cetuximab". Science Translational Medicine. 3 (99): 99ra86. doi: 10.1126/scitranslmed.3002442. PMC 3268675. PMID 21900593.
- ^ Plum PS, Gebauer F, Krämer M, Alakus H, Berlth F, Chon SH, et al. (January 2019). "HER2/neu (ERBB2) expression and gene amplification correlates with better survival in esophageal adenocarcinoma". BMC Cancer. 19 (1): 38. doi: 10.1186/s12885-018-5242-4. PMC 6325716. PMID 30621632.
- ^ Zhao XK, Xing P, Song X, Zhao M, Zhao L, Dang Y, et al. (November 2021). "Focal amplifications are associated with chromothripsis events and diverse prognoses in gastric cardia adenocarcinoma". Nature Communications. 12 (1): 6489. Bibcode: 2021NatCo..12.6489Z. doi: 10.1038/s41467-021-26745-3. PMC 8586158. PMID 34764264.
- ^ Brandt-Rauf PW, Rackovsky S, Pincus MR (November 1990). "Correlation of the structure of the transmembrane domain of the neu oncogene-encoded p185 protein with its function". Proceedings of the National Academy of Sciences of the United States of America. 87 (21): 8660–8664. Bibcode: 1990PNAS...87.8660B. doi: 10.1073/pnas.87.21.8660. PMC 55017. PMID 1978329.
- ^ Mazières J, Peters S, Lepage B, Cortot AB, Barlesi F, Beau-Faller M, et al. (June 2013). "Lung cancer that harbors an HER2 mutation: epidemiologic characteristics and therapeutic perspectives". Journal of Clinical Oncology. 31 (16): 1997–2003. doi: 10.1200/JCO.2012.45.6095. PMID 23610105. S2CID 37663670.
- ^ Mates M, Fletcher GG, Freedman OC, Eisen A, Gandhi S, Trudeau ME, et al. (March 2015). "Systemic targeted therapy for her2-positive early female breast cancer: a systematic review of the evidence for the 2014 Cancer Care Ontario systemic therapy guideline". Current Oncology. 22 (Suppl 1): S114–S122. doi: 10.3747/co.22.2322. PMC 4381787. PMID 25848335.
- ^ Hayes DF, Lippman ME (2018). "Chapter 75: Breast Cancer". In Kasper DL, Fauci AS, Hauser SL, Longo DL, Jameson JL, Loscalzo J (eds.). Harrison's Principles of Internal Medicine (20th ed.). McGraw-Hill Education. ISBN 978-1-259-64403-0.
- ^ Le XF, Pruefer F, Bast RC (January 2005). "HER2-targeting antibodies modulate the cyclin-dependent kinase inhibitor p27Kip1 via multiple signaling pathways". Cell Cycle. 4 (1): 87–95. doi: 10.4161/cc.4.1.1360. PMID 15611642.
- ^ Jiang H, Rugo HS (November 2015). "Human epidermal growth factor receptor 2 positive (HER2+) metastatic breast cancer: how the latest results are improving therapeutic options". Therapeutic Advances in Medical Oncology. 7 (6): 321–339. doi: 10.1177/1758834015599389. PMC 4622301. PMID 26557900.
- ^ Loi S, Sotiriou C, Haibe-Kains B, Lallemand F, Conus NM, Piccart MJ, et al. (June 2009). "Gene expression profiling identifies activated growth factor signaling in poor prognosis (Luminal-B) estrogen receptor positive breast cancer". BMC Medical Genomics. 2: 37. doi: 10.1186/1755-8794-2-37. PMC 2706265. PMID 19552798. lay-url = https://www.sciencedaily.com/releases/2010/05/100506112557.htm / lay-source = ScienceDaily
- ^ "Study sheds new light on tamoxifen resistance". Cordis News. Cordis. 2008-11-13. Archived from the original on 2009-02-20. Retrieved 2008-11-14.
- ^ Hurtado A, Holmes KA, Geistlinger TR, Hutcheson IR, Nicholson RI, Brown M, et al. (December 2008). "Regulation of ERBB2 by oestrogen receptor-PAX2 determines response to tamoxifen". Nature. 456 (7222): 663–666. Bibcode: 2008Natur.456..663H. doi: 10.1038/nature07483. PMC 2920208. PMID 19005469.
- ^ Vranić S, Bešlija S, Gatalica Z (February 2021). "Targeting HER2 expression in cancer: New drugs and new indications". Bosnian Journal of Basic Medical Sciences. 21 (1): 1–4. doi: 10.17305/bjbms.2020.4908. PMC 7861626. PMID 32530388.
- ^ Telli ML, Hunt SA, Carlson RW, Guardino AE (August 2007). "Trastuzumab-related cardiotoxicity: calling into question the concept of reversibility". Journal of Clinical Oncology. 25 (23): 3525–3533. doi: 10.1200/JCO.2007.11.0106. PMID 17687157.
-
^ Petroni S, Caldarola L, Scamarcio R, Giotta F, Latorre A, Mangia A, et al. (2016).
"FISH testing of HER2 immunohistochemistry 1+ invasive breast cancer with unfavorable characteristics". Oncol Lett. 12 (5): 3115–3122.
doi:
10.3892/ol.2016.5125.
PMC
5103906.
PMID
27899970.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ a b "IHC Tests (ImmunoHistoChemistry)". Breastcancer.org. Retrieved 2019-10-04. Last modified on October 23, 2015
- ^ a b Iqbal N, Iqbal N (2014). "Human Epidermal Growth Factor Receptor 2 (HER2) in Cancers: Overexpression and Therapeutic Implications". Molecular Biology International. 2014: 852748. doi: 10.1155/2014/852748. PMC 4170925. PMID 25276427.
- ^
a
b
c
d
e 2018 ASCO/CAP guidelines:
- "Figure 1. Algorithm for evaluation of human epidermal growth factor receptor 2 (HER2) protein expression by immunohistochemistry (IHC) assay of the invasive component of a breast cancer specimen" (PDF). College of American Pathologists: Homepage. Retrieved 2022-09-12.
- Ahn S, Woo JW, Lee K, Park SY (2020). "HER2 status in breast cancer: changes in guidelines and complicating factors for interpretation". J Pathol Transl Med. 54 (1): 34–44. doi: 10.4132/jptm.2019.11.03. PMC 6986968. PMID 31693827.{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Nitta H, Kelly BD, Padilla M, Wick N, Brunhoeber P, Bai I, et al. (May 2012).
"A gene-protein assay for human epidermal growth factor receptor 2 (HER2): brightfield tricolor visualization of HER2 protein, the HER2 gene, and chromosome 17 centromere (CEN17) in formalin-fixed, paraffin-embedded breast cancer tissue sections". Diagnostic Pathology. 7: 60.
doi:
10.1186/1746-1596-7-60.
PMC
3487810.
PMID
22647525.
- "This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( http://creativecommons.org/licenses/by/2.0)" - ^ Giuliano AE, Hurvitz SA (2019). "Breast Disorder". In Papadakis MA, McPhee SJ, Rabow MW (eds.). Current Medical Diagnosis & Treatment. New York, NY: McGraw-Hill.
- ^
a
b
c Diagram and table by Mikael Häggström, MD. Adapted from: Wolff AC, Hammond MEH, Allison KH, Harvey BE, Mangu PB, Bartlett JMS, et al. (2018).
"Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update". J Clin Oncol. 36 (20): 2105–2122.
doi:
10.1200/JCO.2018.77.8738.
hdl:
1805/18766.
PMID
29846122.
S2CID
44143975.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Ali SM, Carney WP, Esteva FJ, Fornier M, Harris L, Köstler WJ, et al. (September 2008). "Serum HER-2/neu and relative resistance to trastuzumab-based therapy in patients with metastatic breast cancer". Cancer. 113 (6): 1294–1301. doi: 10.1002/cncr.23689. PMID 18661530. S2CID 7307111.
- ^ Lennon S, Barton C, Banken L, Gianni L, Marty M, Baselga J, et al. (April 2009). "Utility of serum HER2 extracellular domain assessment in clinical decision making: pooled analysis of four trials of trastuzumab in metastatic breast cancer". Journal of Clinical Oncology. 27 (10): 1685–1693. doi: 10.1200/JCO.2008.16.8351. PMID 19255335.
- ^ Schroeder JA, Adriance MC, McConnell EJ, Thompson MC, Pockaj B, Gendler SJ (June 2002). "ErbB-beta-catenin complexes are associated with human infiltrating ductal breast and murine mammary tumor virus (MMTV)-Wnt-1 and MMTV-c-Neu transgenic carcinomas". The Journal of Biological Chemistry. 277 (25): 22692–22698. doi: 10.1074/jbc.M201975200. PMID 11950845.
- ^ Bonvini P, An WG, Rosolen A, Nguyen P, Trepel J, Garcia de Herreros A, et al. (February 2001). "Geldanamycin abrogates ErbB2 association with proteasome-resistant beta-catenin in melanoma cells, increases beta-catenin-E-cadherin association, and decreases beta-catenin-sensitive transcription". Cancer Research. 61 (4): 1671–1677. PMID 11245482.
- ^ Kanai Y, Ochiai A, Shibata T, Oyama T, Ushijima S, Akimoto S, et al. (March 1995). "c-erbB-2 gene product directly associates with beta-catenin and plakoglobin". Biochemical and Biophysical Research Communications. 208 (3): 1067–1072. doi: 10.1006/bbrc.1995.1443. PMID 7702605.
- ^ Huang YZ, Won S, Ali DW, Wang Q, Tanowitz M, Du QS, et al. (May 2000). "Regulation of neuregulin signaling by PSD-95 interacting with ErbB4 at CNS synapses". Neuron. 26 (2): 443–455. doi: 10.1016/s0896-6273(00)81176-9. PMID 10839362. S2CID 1429113.
- ^ a b Jaulin-Bastard F, Saito H, Le Bivic A, Ollendorff V, Marchetto S, Birnbaum D, et al. (May 2001). "The ERBB2/HER2 receptor differentially interacts with ERBIN and PICK1 PSD-95/DLG/ZO-1 domain proteins". The Journal of Biological Chemistry. 276 (18): 15256–15263. doi: 10.1074/jbc.M010032200. PMID 11278603.
- ^ Bilder D, Birnbaum D, Borg JP, Bryant P, Huigbretse J, Jansen E, et al. (July 2000). "Collective nomenclature for LAP proteins". Nature Cell Biology. 2 (7): E114. doi: 10.1038/35017119. PMID 10878817. S2CID 19749569.
- ^ Huang YZ, Zang M, Xiong WC, Luo Z, Mei L (January 2003). "Erbin suppresses the MAP kinase pathway". The Journal of Biological Chemistry. 278 (2): 1108–1114. doi: 10.1074/jbc.M205413200. PMID 12379659.
- ^ a b Schulze WX, Deng L, Mann M (2005). "Phosphotyrosine interactome of the ErbB-receptor kinase family". Molecular Systems Biology. 1: 2005.0008. doi: 10.1038/msb4100012. PMC 1681463. PMID 16729043.
- ^ Bourguignon LY, Zhu H, Zhou B, Diedrich F, Singleton PA, Hung MC (December 2001). "Hyaluronan promotes CD44v3-Vav2 interaction with Grb2-p185(HER2) and induces Rac1 and Ras signaling during ovarian tumor cell migration and growth". The Journal of Biological Chemistry. 276 (52): 48679–48692. doi: 10.1074/jbc.M106759200. PMID 11606575.
- ^ a b Olayioye MA, Graus-Porta D, Beerli RR, Rohrer J, Gay B, Hynes NE (September 1998). "ErbB-1 and ErbB-2 acquire distinct signaling properties dependent upon their dimerization partner". Molecular and Cellular Biology. 18 (9): 5042–5051. doi: 10.1128/mcb.18.9.5042. PMC 109089. PMID 9710588.
- ^ Xu W, Mimnaugh E, Rosser MF, Nicchitta C, Marcu M, Yarden Y, et al. (February 2001). "Sensitivity of mature Erbb2 to geldanamycin is conferred by its kinase domain and is mediated by the chaperone protein Hsp90". The Journal of Biological Chemistry. 276 (5): 3702–3708. doi: 10.1074/jbc.M006864200. PMID 11071886.
- ^ Jeong JH, An JY, Kwon YT, Li LY, Lee YJ (October 2008). "Quercetin-induced ubiquitination and down-regulation of Her-2/neu". Journal of Cellular Biochemistry. 105 (2): 585–595. doi: 10.1002/jcb.21859. PMC 2575035. PMID 18655187.
- ^ Grant SL, Hammacher A, Douglas AM, Goss GA, Mansfield RK, Heath JK, et al. (January 2002). "An unexpected biochemical and functional interaction between gp130 and the EGF receptor family in breast cancer cells". Oncogene. 21 (3): 460–474. doi: 10.1038/sj.onc.1205100. PMID 11821958. S2CID 19754641.
- ^ Li Y, Yu WH, Ren J, Chen W, Huang L, Kharbanda S, et al. (August 2003). "Heregulin targets gamma-catenin to the nucleolus by a mechanism dependent on the DF3/MUC1 oncoprotein". Molecular Cancer Research. 1 (10): 765–775. PMID 12939402.
- ^ Schroeder JA, Thompson MC, Gardner MM, Gendler SJ (April 2001). "Transgenic MUC1 interacts with epidermal growth factor receptor and correlates with mitogen-activated protein kinase activation in the mouse mammary gland". The Journal of Biological Chemistry. 276 (16): 13057–13064. doi: 10.1074/jbc.M011248200. PMID 11278868.
- ^ Gout I, Dhand R, Panayotou G, Fry MJ, Hiles I, Otsu M, et al. (December 1992). "Expression and characterization of the p85 subunit of the phosphatidylinositol 3-kinase complex and a related p85 beta protein by using the baculovirus expression system". The Biochemical Journal. 288 ( Pt 2) (2): 395–405. doi: 10.1042/bj2880395. PMC 1132024. PMID 1334406.
- ^ Peles E, Levy RB, Or E, Ullrich A, Yarden Y (August 1991). "Oncogenic forms of the neu/HER2 tyrosine kinase are permanently coupled to phospholipase C gamma". The EMBO Journal. 10 (8): 2077–2086. doi: 10.1002/j.1460-2075.1991.tb07739.x. PMC 452891. PMID 1676673.
- ^ Arteaga CL, Johnson MD, Todderud G, Coffey RJ, Carpenter G, Page DL (December 1991). "Elevated content of the tyrosine kinase substrate phospholipase C-gamma 1 in primary human breast carcinomas". Proceedings of the National Academy of Sciences of the United States of America. 88 (23): 10435–10439. Bibcode: 1991PNAS...8810435A. doi: 10.1073/pnas.88.23.10435. PMC 52943. PMID 1683701.
- ^ Wong L, Deb TB, Thompson SA, Wells A, Johnson GR (March 1999). "A differential requirement for the COOH-terminal region of the epidermal growth factor (EGF) receptor in amphiregulin and EGF mitogenic signaling". The Journal of Biological Chemistry. 274 (13): 8900–8909. doi: 10.1074/jbc.274.13.8900. PMID 10085134.
Further reading
- Ross JS, Fletcher JA, Linette GP, Stec J, Clark E, Ayers M, et al. (2003). "The Her-2/neu gene and protein in breast cancer 2003: biomarker and target of therapy". The Oncologist. 8 (4): 307–325. doi: 10.1634/theoncologist.8-4-307. PMID 12897328. S2CID 1055491. (This paper currently has an expression of concern, see doi: 10.1634/theoncologist.2016-3001, PMID 26966227, Retraction Watch)
- Zhou BP, Hung MC (October 2003). "Dysregulation of cellular signaling by HER2/neu in breast cancer". Seminars in Oncology. 30 (5 Suppl 16): 38–48. doi: 10.1053/j.seminoncol.2003.08.006. PMID 14613025.
- Ménard S, Casalini P, Campiglio M, Pupa SM, Tagliabue E (December 2004). "Role of HER2/neu in tumor progression and therapy". Cellular and Molecular Life Sciences. 61 (23): 2965–2978. doi: 10.1007/s00018-004-4277-7. PMID 15583858. S2CID 37611938.
- Becker JC, Muller-Tidow C, Serve H, Domschke W, Pohle T (June 2006). "Role of receptor tyrosine kinases in gastric cancer: new targets for a selective therapy". World Journal of Gastroenterology. 12 (21): 3297–3305. doi: 10.3748/wjg.v12.i21.3297. PMC 4087885. PMID 16733844.
- Laudadio J, Quigley DI, Tubbs R, Wolff DJ (January 2007). "HER2 testing: a review of detection methodologies and their clinical performance". Expert Review of Molecular Diagnostics. 7 (1): 53–64. doi: 10.1586/14737159.7.1.53. PMID 17187484. S2CID 9971746.
- Bianchi F, Tagliabue E, Ménard S, Campiglio M (March 2007). "Fhit expression protects against HER2-driven breast tumor development: unraveling the molecular interconnections". Cell Cycle. 6 (6): 643–646. doi: 10.4161/cc.6.6.4033. hdl: 2434/810034. PMID 17374991.
External links
- AACR Cancer Concepts Factsheet on HER2
- Breast Friends for Life Network - A South African Breast Cancer Support Forum for HER2 Positive Women
- HerceptinR : Herceptin Resistance Database for Understanding Mechanism of Resistance in Breast Cancer Patients. Sci. Rep. 4:4483
- Receptor,+erbB-2 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Receptor tyrosine-protein kinase erbB-2
















![Algorithm for the evaluation of HER2 on fluorescence in situ hybridization (FISH).[48]](https://upload.wikimedia.org/wikipedia/commons/thumb/d/d2/HER2_FISH_algorithm.svg/579px-HER2_FISH_algorithm.svg.png)


