| CadoganŌĆōSundberg indole synthesis | |
|---|---|
| Named after |
John Cadogan Richard J. Sundberg |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000509 |
The CadoganŌĆōSundberg indole synthesis, or simply Cadogan indole synthesis, is a name reaction in organic chemistry that allows for the generation of indoles from o-nitrostyrenes with the use of trialkyl phosphites, such as triethyl phosphite. [1] [2] [3] [4] [5] [6] [7]
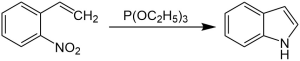
Mechanism
o-nitrostyrene first reacts with triethyl phosphite, and the nitro group is converted to a nitroso group. The nitroso group then reacts with the alkene, and N-hydroxylindole is formed, which reacts again with triethyl phosphite to form the indole. [1] [2] [3] [4]

Application
The CadoganŌĆōSundberg indole synthesis has been used as an intermediate step in the total synthesis of Tjipanazole E, [8] transforming 2-[trans-2-[5-Chloro-2-nitrophenyl)vinyl]-5-chloro-1H-indole to 5,5ŌĆÖ-Dichloro-2,2ŌĆÖ-biindole.
References
- ^ a b Cadogan, John Ivan George; Mackie, R. K. (1974). "Tervalent phosphorus compounds in organic synthesis". Chemical Society Reviews. 3 (1): 87ŌĆō137. doi: 10.1039/CS9740300087.
- ^ a b Wang, Zerong (15 September 2010). Comprehensive Organic Name Reactions and Reagents. doi: 10.1002/9780470638859.conrr128. ISBN 9780470638859.
- ^ a b Sundberg, Richard J.; Yamazaki, Toshio (1 February 1967). "Rearrangements and ring expansions during the deoxygenation of ╬▓,╬▓-disubstituted o-nitrostyrenes". The Journal of Organic Chemistry. 32 (2): 290ŌĆō294. doi: 10.1021/jo01288a009.
- ^ a b Sundberg, Richard J. (1 November 1965). "Deoxygenation of Nitro Groups by Trivalent Phosphorus. Indoles from o-Nitrostyrenes". The Journal of Organic Chemistry. 30 (11): 3604ŌĆō3610. doi: 10.1021/jo01022a006.
- ^ Gribble, Gordon W. (17 June 2016). "CadoganŌĆōSundberg Indole Synthesis". Indole Ring Synthesis: From Natural Products to Drug Discovery. pp. 266ŌĆō277. doi: 10.1002/9781118695692.ch26. ISBN 9781118695692.
- ^ Majgier-Baranowska, Helena; Williams, John D.; Li, Bing; Peet, Norton P. (1 February 1967). "Studies on the mechanism of the CadoganŌĆōSundberg indole synthesis". Tetrahedron Letters. 53 (35): 4785ŌĆō4788. doi: 10.1016/j.tetlet.2012.06.146.
- ^ Li, Jie Jack (4 January 2014). "CadoganŌĆōSundberg indole synthesis". Indole Ring Synthesis: From Natural Products to Drug Discovery. Springer. p. 102-103. doi: 10.1007/978-3-319-03979-4_49. ISBN 978-3-319-03979-4.
- ^ Kuethe, Jeffrey T.; Wong, Audrey; Davies, Iaan W. (3 September 2003). "Effective Strategy for the Preparation of Indolocarbazole Aglycons and Glycosides: Total Synthesis of Tjipanazoles B, D, E, and I". Organic Letters. 5 (20): 3721ŌĆō3723. doi: 10.1021/ol035418r. PMID 14507214.