 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /klɪndəˈmaɪsɪn/ |
| Trade names | Cleocin, Clinacin, Dalacin, others |
| Other names | 7-chloro-lincomycin 7-chloro-7-deoxylincomycin, DARE-BV1 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a682399 |
| License data |
|
|
Pregnancy category |
|
|
Routes of administration | By mouth, topical, intravenous, intravaginal |
| Drug class | Lincosamide antibiotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 90% (by mouth) 4–5% (topical) |
| Protein binding | 95% |
| Metabolism | Liver |
| Elimination half-life | 2–3 hour |
| Excretion | Bile duct and kidney (around 20%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.038.357 |
| Chemical and physical data | |
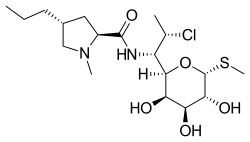
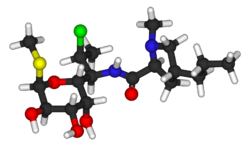
| Formula | C18H33ClN2O5S |
| Molar mass | 424.98 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Clindamycin is a lincosamide antibiotic medication used for the treatment of a number of bacterial infections, including osteomyelitis (bone) or joint infections, pelvic inflammatory disease, strep throat, pneumonia, acute otitis media (middle ear infections), and endocarditis. [5] It can also be used to treat acne, [5] [6] and some cases of methicillin-resistant Staphylococcus aureus (MRSA). [7] In combination with quinine, it can be used to treat malaria. [5] [6] It is available by mouth, by injection into a vein, and as a cream or a gel to be applied to the skin or in the vagina. [4] [5] [6] [8] [9]
Common side effects include nausea and vomiting, diarrhea, skin rashes, and pain at the site of injection. [5] It increases the risk of hospital-acquired Clostridium difficile colitis about fourfold and thus is only recommended for use when other antibiotics are not appropriate. [10] [5] It appears to be generally safe in pregnancy. [5] It is of the lincosamide class and works by blocking bacteria from making protein. [5]
Clindamycin was first made in 1966 from lincomycin. [11] [12] It is on the World Health Organization's List of Essential Medicines. [13] It is available as a generic medication. [14] [15] In 2021, it was the 118th most commonly prescribed medication in the United States, with more than 5 million prescriptions. [16] [17]
Medical uses
Clindamycin is used primarily to treat anaerobic infections caused by susceptible anaerobic bacteria, including dental infections, [18] and infections of the respiratory tract, skin, and soft tissue, and peritonitis. [19] In people with hypersensitivity to penicillins, clindamycin may be used to treat infections caused by susceptible aerobic bacteria, as well. It is also used to treat bone and joint infections, particularly those caused by Staphylococcus aureus. [19] [20] Topical application of clindamycin phosphate can be used to treat mild to moderate acne. [21] [22]
Acne

For the treatment of acne, in the long term, the combined use of topical clindamycin and benzoyl peroxide was similar to salicylic acid plus benzoyl peroxide. [22] [23] Topical clindamycin plus topical benzoyl peroxide is more effective than topical clindamycin alone. [22] [23]
Susceptible bacteria
It is most effective against infections involving the following types of organisms:
- Aerobic Gram-positive cocci, including some members of the Staphylococcus and Streptococcus (e.g. pneumococcus) genera, but not enterococci. [24]
- Anaerobic, Gram-negative rod-shaped bacteria, including some Bacteroides, Fusobacterium, and Prevotella, although resistance is increasing in Bacteroides fragilis. [25]
Most aerobic Gram-negative bacteria (such as Pseudomonas, Legionella, Haemophilus influenzae and Moraxella) are resistant to clindamycin, [24] [26] as are the facultative anaerobic Enterobacteriaceae. [27] A notable exception is Capnocytophaga canimorsus, for which clindamycin is a first-line drug of choice. [28]
The following represents MIC susceptibility data for a few medically significant pathogens. [29]
- Staphylococcus aureus: 0.016 μg/mL – >256 μg/mL
- Streptococcus pneumoniae: 0.002 μg/mL – >256 μg/mL
- Streptococcus pyogenes: <0.015 μg/mL – >64 μg/mL
D-test
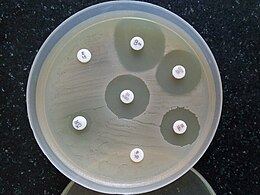
When testing a gram-positive culture for sensitivity to clindamycin, it is common to perform a "D-test" to determine if there is a sub-population of bacteria present with the phenotype known as iMLSB. This phenotype of bacteria are resistant to the macrolide- lincosamide- streptogramin B group of antibiotics, however, the resistance mechanism is only induced by the presence of 14-membered ring macrolides, such as erythromycin. During a D-test, bacteria of the iMLSB phenotype demonstrate in vitro erythromycin-induced in vitro resistance to clindamycin. This is because of the activity of the macrolide-inducible plasmid-encoded erm gene. [30]
To perform a D-test, an agar plate is inoculated with the bacteria in question and two drug-impregnated disks (one with erythromycin, one with clindamycin) are placed 15–20 mm apart on the plate. If the area of inhibition around the clindamycin disk is D-shaped, the test result is positive. Despite the apparent susceptibility to clindamycin in the absence of erythromycin, a positive D-test precludes therapeutic use of clindamycin. This is because the erythromycin-inducible erm gene is prone to mutations causing the inducible activity to switch to constitutive (permanently switched on). [31] This in turn, may lead to the therapeutic failure of clindamycin.
If the area of inhibition around the clindamycin disk is circular, the test result is negative and clindamycin can be used. [31]
Malaria
Given with chloroquine or quinine, clindamycin is effective and well tolerated in treating Plasmodium falciparum malaria; the latter combination is particularly useful for children, and is the treatment of choice for pregnant women who become infected in areas where resistance to chloroquine is common. [32] [33] Clindamycin should not be used as an antimalarial by itself, although it appears to be very effective as such, because of its slow action. [32] [33] Patient-derived isolates of Plasmodium falciparum from the Peruvian Amazon have been reported to be resistant to clindamycin as evidenced by in vitro drug susceptibility testing. [34]
Other
Clindamycin may be useful in skin and soft tissue infections caused by methicillin-resistant Staphylococcus aureus (MRSA). [7] Many strains of MRSA are still susceptible to clindamycin; however, in the United States spreading from the West Coast eastwards, MRSA is becoming increasingly resistant.[ medical citation needed]
While it has been used in intraabdominal infections, such use is generally not recommended due to resistance. [5]
Clindamycin is used in cases of suspected toxic shock syndrome, [35] often in combination with a bactericidal agent such as vancomycin. The rationale for this approach is a presumed synergy between vancomycin, which causes the death of the bacteria by breakdown of the cell wall, and clindamycin, which is a powerful inhibitor of toxin synthesis. Both in vitro and in vivo studies have shown clindamycin reduces the production of exotoxins by staphylococci; [36] it may also induce changes in the surface structure of bacteria that make them more sensitive to immune system attack ( opsonization and phagocytosis). [37] [38]
Clindamycin has been proven to decrease the risk of premature births in women diagnosed with bacterial vaginosis during early pregnancy to about a third of the risk of untreated women. [39]
The combination of clindamycin and quinine is the standard treatment for severe babesiosis. [40]
Clindamycin may also be used to treat toxoplasmosis, [24] [41] [42] and, in combination with primaquine, is effective in treating mild to moderate Pneumocystis jirovecii pneumonia. [43]
Clindamycin, either applied to skin or taken by mouth, may also be used in hidradenitis suppurativa. [44]
Side effects
Common adverse drug reactions associated with systemic clindamycin therapy – found in over 1% of people – include: diarrhea, pseudomembranous colitis, nausea, vomiting, abdominal pain or cramps and/or rash. High doses (both intravenous and oral) may cause a metallic taste. Common adverse drug reactions associated with topical formulations – found in over 10% of people – include: dryness, burning, itching, scaliness, or peeling of skin (lotion, solution); erythema (foam, lotion, solution); oiliness (gel, lotion). Additional side effects include contact dermatitis. [45] [46] Common side effects – found in over 10% of people – in vaginal applications include fungal infection.[ medical citation needed]
Rarely – in less than 0.1% of people – clindamycin therapy has been associated with anaphylaxis, blood dyscrasias, polyarthritis, jaundice, raised liver enzyme levels, renal dysfunction, cardiac arrest, and/or hepatotoxicity. [45]
Clostridioides difficile
Pseudomembranous colitis is a potentially lethal condition commonly associated with clindamycin, but which also occurs with other antibiotics. [10] [47] Overgrowth of Clostridioides difficile, which is inherently resistant to clindamycin, results in the production of a toxin that causes a range of adverse effects, from diarrhea to colitis and toxic megacolon. [45] [48]
Pregnancy and breastfeeding
Use of clindamycin during pregnancy is generally considered safe. [49]
Clindamycin is classified as compatible with breastfeeding by the American Academy of Pediatrics, [50] however, the WHO categorizes it as "avoid if possible". [51] It is classified as L2 probably compatible with breastfeeding according to Medications and Mothers' Milk. [52] A 2009 review found it was likely safe in breastfeeding mothers, but did find one complication ( hematochezia) in a breastfed infant which might be attributable to clindamycin. [53] LactMed lists potentially negative gastrointestinal effects in babies whose mothers take it while breastfeeding but did not see that as justification to stop breastfeeding. [54]
Interactions
Clindamycin may prolong the effects of neuromuscular-blocking drugs, such as succinylcholine and vecuronium. [55] [56] [57] Its similarity to the mechanism of action of macrolides and chloramphenicol means they should not be given simultaneously, as this causes antagonism [26] and possible cross-resistance.[ medical citation needed]
Chemistry
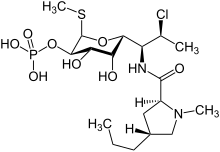
Clindamycin is a semisynthetic derivative of lincomycin, a natural antibiotic produced by the actinobacterium Streptomyces lincolnensis. It is obtained by 7(S)- chloro- substitution of the 7(R)- hydroxyl group of lincomycin. [59] [60] The synthesis of clindamycin was first announced by BJ Magerlein, RD Birkenmeyer, and F Kagan on the fifth Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC) in 1966. [61] It has been on the market since 1968. [46]
Clindamycin is white or yellow powder that is very soluble in water. [62] The topically used clindamycin phosphate is a phosphate- ester prodrug of clindamycin. [58]
Mechanism of action

Clindamycin has a primarily bacteriostatic effect. At higher concentrations, it may be bactericidal. [62] It is a bacterial protein synthesis inhibitor by inhibiting ribosomal translocation, [63] in a similar way to macrolides. It does so by binding to the rRNA of the bacterial 50S ribosome subunit, overlapping with the binding sites of the oxazolidinone, pleuromutilin, and macrolide antibiotics, among others. [24] [64] The binding is reversible. [65] Clindamycin is more effective than lincomycin. [62]
The X-ray crystal structures of clindamycin bound to ribosomes (or ribosomal subunits) derived from Escherichia coli, [66] Deinococcus radiodurans, [67] and Haloarcura marismortui [68] have been determined; the structure of the closely related antibiotic lincomycin bound to the 50S ribosomal subunit of Staphylococcus aureus has also been reported. [69]
Society and culture
Economics
Clindamycin is available as a generic medication and is relatively inexpensive. [14] [70]
Available forms

Clindamycin preparations that are taken by mouth include capsules (containing clindamycin hydrochloride) and oral suspensions (containing clindamycin palmitate hydrochloride). [32] Oral suspension is not favored for administration of clindamycin to children, due to its extremely foul taste and odor. Clindamycin is formulated in a vaginal cream and as vaginal ovules for treatment of bacterial vaginosis. [39] It is also available for topical administration in gel form, as a lotion, and in a foam delivery system (each containing clindamycin phosphate) and a solution in ethanol (containing clindamycin hydrochloride) and is used primarily as a prescription acne treatment. [71]
Several combination acne treatments containing clindamycin are also marketed, such as single-product formulations of clindamycin with benzoyl peroxide—sold as BenzaClin ( Sanofi-Aventis), Duac (a gel form made by Stiefel), and Acanya, among other trade names—and, in the United States, a combination of clindamycin and tretinoin, sold as Ziana. [72] In India, vaginal suppositories containing clindamycin in combination with clotrimazole are manufactured by Olive Health Care and sold as Clinsup-V. In Egypt, vaginal cream containing clindamycin produced by Biopharmgroup sold as Vagiclind indicated for vaginosis.[ citation needed]
Clindamycin is available as a generic drug, for both systemic (oral and intravenous) and topical use. [32] (The exception is the vaginal suppository, which is not available as a generic in the US [73]).
Veterinary use
The veterinary uses of clindamycin are quite similar to its human indications, and include treatment of osteomyelitis, [74] skin infections, and toxoplasmosis, for which it is the preferred drug in dogs and cats. [75] They can be used both by mouth and topically. [62] A disadvantage is that bacterial resistance can develop fairly quickly. [62] Gastrointestinal upset may also occur. [62] Toxoplasmosis rarely causes symptoms in cats, but can do so in very young or immunocompromised kittens and cats.[ citation needed]
References
- ^ Use During Pregnancy and Breastfeeding
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Product monograph brand safety updates". Health Canada. 6 June 2024. Retrieved 8 June 2024.
- ^ a b "Xaciato- clindamycin phosphate gel". DailyMed. Retrieved 24 December 2021.
- ^ a b c d e f g h i "Clindamycin (Systemic)". The American Society of Health-System Pharmacists. Archived from the original on 12 August 2021. Retrieved 19 December 2021.
- ^ a b c Leyden JJ (2006). Hidradenitis suppurativa. Berlin: Springer. p. 152. ISBN 9783540331018. Archived from the original on 8 September 2017.
- ^ a b Daum RS (July 2007). "Clinical practice. Skin and soft-tissue infections caused by methicillin-resistant Staphylococcus aureus". N. Engl. J. Med. 357 (4): 380–90. doi: 10.1056/NEJMcp070747. PMID 17652653.
- ^ "Clindamycin phosphate- clindamycin phosphate gel usp, 1% gel". DailyMed. Retrieved 19 December 2021.
- ^ "Daré Announces FDA Approval of Xaciato (clindamycin phosphate) Vaginal Gel as a Treatment for Bacterial Vaginosis". Daré Bioscience (Press release). 7 December 2021. Retrieved 19 December 2021.
- ^ a b Thomas C, Stevenson M, Riley TV (2003). "Antibiotics and hospital-acquired Clostridium difficile-associated diarrhoea: a systematic review". J Antimicrob Chemother. 51 (6): 1339–50. doi: 10.1093/jac/dkg254. PMID 12746372.
- ^ Smieja M (January 1998). "Current indications for the use of clindamycin: A critical review". The Canadian Journal of Infectious Diseases. 9 (1): 22–8. doi: 10.1155/1998/538090. PMC 3250868. PMID 22346533.
- ^ Neonatal Formulary: Drug Use in Pregnancy and the First Year of Life (7 ed.). John Wiley & Sons. 2014. p. 162. ISBN 9781118819517. Archived from the original on 8 September 2017.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl: 10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ a b Hamilton R (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 108. ISBN 9781284057560.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Clindamycin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ Brook I, Lewis MA, Sándor GK, Jeffcoat M, Samaranayake LP, Vera Rojas J (November 2005). "Clindamycin in dentistry: more than just effective prophylaxis for endocarditis?". Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 100 (5): 550–8. doi: 10.1016/j.tripleo.2005.02.086. PMID 16243239.
- ^ a b "Cleocin I.V. Indications & Dosage". RxList.com. 2007. Archived from the original on 27 November 2007. Retrieved 1 December 2007.
- ^ Darley ES, MacGowan AP (2004). "Antibiotic treatment of gram-positive bone and joint infections". J Antimicrob Chemother. 53 (6): 928–35. doi: 10.1093/jac/dkh191. PMID 15117932.
- ^ Feldman S, Careccia RE, Barham KL, Hancox J (May 2004). "Diagnosis and treatment of acne". Am Fam Physician. 69 (9): 2123–30. PMID 15152959. Archived (PDF) from the original on 27 July 2011.
- ^ a b c Oge' LK, Broussard A, Marshall MD (October 2019). "Acne Vulgaris: Diagnosis and Treatment". Am Fam Physician. 100 (8): 475–84. PMID 31613567.
- ^ a b Seidler EM, Kimball AB (July 2010). "Meta-analysis comparing efficacy of benzoyl peroxide, clindamycin, benzoyl peroxide with salicylic acid, and combination benzoyl peroxide/clindamycin in acne". J. Am. Acad. Dermatol. 63 (1): 52–62. doi: 10.1016/j.jaad.2009.07.052. PMID 20488582.
- ^ a b c d "Lincosamides, Oxazolidinones, and Streptogramins". Merck Manual of Diagnosis and Therapy. Merck & Co. May 2020. Archived from the original on 2 December 2007. Retrieved 19 December 2021.
- ^ Di Bella S, Antonello RM, Sanson G, Maraolo AE, Giacobbe DR, Sepulcri C, et al. (June 2022). "Anaerobic bloodstream infections in Italy (ITANAEROBY): A 5-year retrospective nationwide survey". Anaerobe. 75: 102583. doi: 10.1016/j.anaerobe.2022.102583. hdl: 11368/3020691. PMID 35568274. S2CID 248736289.
- ^ a b Bell EA (January 2005). "Clindamycin: new look at an old drug". Infectious Diseases in Children. Archived from the original on 8 October 2011. Retrieved 1 December 2007.
- ^ Gold HS, Moellering RC (1999). "Macrolides and clindamycin". In Root RE, Waldvogel F, Corey L, Stamm WE (eds.). Clinical infectious diseases: a practical approach. Oxford: Oxford University Press. pp. 291–7. ISBN 978-0-19-508103-9. Archived from the original on 13 May 2018. Retrieved 19 January 2009.through Google Book Search.
- ^ Jolivet-Gougeon A, Sixou JL, Tamanai-Shacoori Z, Bonnaure-Mallet M (April 2007). "Antimicrobial treatment of Capnocytophaga infections". Int J Antimicrob Agents. 29 (4): 367–73. doi: 10.1016/j.ijantimicag.2006.10.005. PMID 17250994.
- ^ "Clindamycin Phosphate Susceptibility and Minimum Inhibitory Concentration (MIC) Data" (PDF). toku-e.com. 1 June 2020.
- ^ Leclerq R (February 2002). "Mechanisms of Resistance to Macrolides and Lincosamides: Nature of the Resistance Elements and Their Clinical Implications". Clinical Infectious Diseases. 34 (4): 482–92. doi: 10.1086/324626. PMID 11797175.
- ^ a b Woods CR (December 2009). "Macrolide-inducible resistance to clindamycin and the D-test". The Pediatric Infectious Disease Journal. 28 (12): 1115–8. doi: 10.1097/INF.0b013e3181c35cc5. PMID 19935273.
- ^ a b c d Lell B, Kremsner PG (2002). "Clindamycin as an antimalarial drug: review of clinical trials". Antimicrobial Agents and Chemotherapy. 46 (8): 2315–20. doi: 10.1128/AAC.46.8.2315-2320.2002. ISSN 0066-4804. PMC 127356. PMID 12121898.
- ^ a b Griffith KS, Lewis LS, Mali S, Parise ME (2007). "Treatment of malaria in the United States: a systematic review". JAMA. 297 (20): 2264–77. doi: 10.1001/jama.297.20.2264. PMID 17519416.
- ^ Dharia NV, Plouffe D, Bopp SE, González-Páez GE, Lucas C, Salas C, et al. (2010). "Genome scanning of Amazonian Plasmodium falciparum shows subtelomeric instability and clindamycin-resistant parasites". Genome Research. 20 (11): 1534–44. doi: 10.1101/gr.105163.110. PMC 2963817. PMID 20829224.
- ^ Annane D, Clair B, Salomon J (2004). "Managing toxic shock syndrome with antibiotics". Expert Opin Pharmacother. 5 (8): 1701–10. doi: 10.1517/14656566.5.8.1701. PMID 15264985. S2CID 24494787.
- ^ Coyle EA (May 2003). "Targeting bacterial virulence: the role of protein synthesis inhibitors in severe infections. Insights from the Society of Infectious Diseases Pharmacists". Pharmacotherapy. 23 (5): 638–42. doi: 10.1592/phco.23.5.638.32191. PMID 12741438. S2CID 29061418.
- ^ Gemmell CG, O'Dowd A (1983). "Regulation of protein A biosynthesis in Staphylococcus aureus by certain antibiotics: its effect on phagocytosis by leukocytes". J Antimicrob Chemother. 12 (6): 587–97. doi: 10.1093/jac/12.6.587. PMID 6662837.
- ^ Gemmell CG, Peterson PK, Schmeling D, Kim Y, Mathews J, Wannamaker L, et al. (May 1981). "Potentiation of opsonization and phagocytosis of Streptococcus pyogenes following growth in the presence of clindamycin". J Clin Invest. 67 (5): 1249–56. doi: 10.1172/JCI110152. PMC 370690. PMID 7014632.
- ^ a b Lamont RF (2005). "Can antibiotics prevent preterm birth—the pro and con debate". BJOG. 112 (Suppl 1): 67–73. doi: 10.1111/j.1471-0528.2005.00589.x. PMID 15715599. S2CID 25572794.
- ^ Homer MJ, Aguilar-Delfin I, Telford SR, Krause PJ, Persing DH (July 2000). "Babesiosis". Clin Microbiol Rev. 13 (3): 451–69. doi: 10.1128/CMR.13.3.451-469.2000. PMC 88943. PMID 10885987.
- ^ Pleyer U, Torun N, Liesenfeld O (2007). "Okuläre Toxoplasmose" [Ocular toxoplasmosis]. Ophthalmologe (in German). 104 (7): 603–16. doi: 10.1007/s00347-007-1535-8. PMID 17530262. S2CID 36696180.
- ^ Jeddi A, Azaiez A, Bouguila H, Kaoueche M, Malouche S, Daghfous F, et al. (1997). "Intérêt de la clindamycine dans le traitement de la toxoplasmose oculaire" [Value of clindamycin in the treatment of ocular toxoplasmosis]. Journal Français d'Ophtalmologie (in French). 20 (6): 418–22. ISSN 0181-5512. PMID 9296037.
- ^ Fishman JA (June 1998). "Treatment of infection due to Pneumocystis carinii". Antimicrobial Agents and Chemotherapy. 42 (6): 1309–14. doi: 10.1128/AAC.42.6.1309. PMC 105593. PMID 9624465.
- ^ Saunte DM, Jemec GB (November 2017). "Hidradenitis Suppurativa: Advances in Diagnosis and Treatment". JAMA. 318 (20): 2019–32. doi: 10.1001/jama.2017.16691. PMID 29183082. S2CID 5017318.
- ^ a b c Rossi S, ed. (2006). Australian Medicines Handbook. Adelaide: AMH Pty Ltd.
- ^ a b de Groot MC, van Puijenbroek EP (October 2007). "Clindamycin and taste disorders". British Journal of Clinical Pharmacology. 64 (4): 542–5. doi: 10.1111/j.1365-2125.2007.02908.x. PMC 2048568. PMID 17635503.
- ^ Starr J (2005). "Clostridium difficile associated diarrhoea: diagnosis and treatment". BMJ. 331 (7515): 498–501. doi: 10.1136/bmj.331.7515.498. PMC 1199032. PMID 16141157.
- ^ Kelly CP, Pothoulakis C, LaMont JT (January 1994). "Clostridium difficile colitis". New England Journal of Medicine. 330 (4): 257–62. doi: 10.1056/NEJM199401273300406. PMID 8043060.
- ^ Lell B, Kremsner PG (August 2002). "Clindamycin as an antimalarial drug: review of clinical trials". Antimicrobial Agents and Chemotherapy. 46 (8): 2315–20. doi: 10.1128/AAC.46.8.2315-2320.2002. PMC 127356. PMID 12121898.
- ^ American Academy of Pediatrics Committee on Drugs (September 2001). "Transfer of drugs and other chemicals into human milk". Pediatrics. 108 (3): 776–89. doi: 10.1542/peds.108.3.776. PMID 11533352.
-
^ Organization WH (2002). Breastfeeding and maternal medication : recommendations for drugs in the Eleventh WHO Model List of Essential Drugs. Geneva, Switzerland: World Health Organization.
hdl:
10665/62435.
{{ cite book}}:|work=ignored ( help) - ^ Hale TW (2017). Medications & mothers' milk. Rowe, Hilary E. (Seventeenth ed.). New York, NY: Springer. ISBN 9780826128584. OCLC 959873270.
- ^ Mitrano JA, Spooner LM, Belliveau P (September 2009). "Excretion of antimicrobials used to treat methicillin-resistant Staphylococcus aureus infections during lactation: safety in breastfeeding infants". Pharmacotherapy. 29 (9): 1103–9. doi: 10.1592/phco.29.9.1103. PMID 19698015. S2CID 2594769.
- ^ "Clindamycin". Drugs and Lactation Database (LactMed). National Library of Medicine (US). 2006. PMID 30000267. Bookshelf ID: NBK501208.
- ^ Fogdall RP, Miller RD (1974). "Prolongation of a pancuronium-induced neuromuscular blockade by clindamycin". Anesthesiology. 41 (4): 407–8. doi: 10.1097/00000542-197410000-00023. PMID 4415332.
- ^ al Ahdal O, Bevan DR (1995). "Clindamycin-induced neuromuscular blockade". Can J Anaesth. 42 (7): 614–7. doi: 10.1007/BF03011880. PMID 7553999.
- ^ Sloan PA, Rasul M (2002). "Prolongation of rapacuronium neuromuscular blockade by clindamycin and magnesium". Anesth Analg. 94 (1): 123–4, table of contents. doi: 10.1097/00000539-200201000-00023. PMID 11772813.
- ^ a b "Clindamycin Phosphate Topical Solution". RxList. Archived from the original on 2 February 2017. Retrieved 27 January 2017.
- ^ Birkenmeyer RD, Kagan F (July 1970). "Lincomycin. XI. Synthesis and structure of clindamycin, a potent antibacterial agent". Journal of Medicinal Chemistry. 13 (4): 616–19. doi: 10.1021/jm00298a007. PMID 4916317.
- ^ Meyers BR, Kaplan K, Weinstein L (1969). "Microbiological and Pharmacological Behavior of 7-Chlorolincomycin". Appl Microbiol. 17 (5): 653–7. doi: 10.1128/AEM.17.5.653-657.1969. PMC 377774. PMID 4389137.
- ^ Magerlein BJ, Birkenmeyer RD, Kagan F (1966). "Chemical modification of lincomycin". Antimicrobial Agents and Chemotherapy. 6: 727–36. PMID 5985307.
- ^ a b c d e f Spízek J, Rezanka T (May 2004). "Lincomycin, clindamycin and their applications". Applied Microbiology and Biotechnology. 64 (4): 455–64. doi: 10.1007/s00253-003-1545-7. PMID 14762701. S2CID 7870760.
- ^ Clindamycin University of Michigan. Retrieved 31 July 2009
- ^ Wilson DN (January 2014). "Ribosome-targeting antibiotics and mechanisms of bacterial resistance". Nature Reviews Microbiology. 12 (1): 35–48. doi: 10.1038/nrmicro3155. PMID 24336183. S2CID 9264620.
- ^ Beauduy CE, Winston LG. Tetracyclines, Macrolides, Clindamycin, Chloramphenicol, Streptogramins, & Oxazolidinones. In: Katzung BG. eds. Basic & Clinical Pharmacology, 14e New York, NY: McGraw-Hill; .
- ^ Dunkle JA, Xiong L, Mankin AS, Cate JH (October 2010). "Structures of the Escherichia coli ribosome with antibiotics bound near the peptidyl transferase center explain spectra of drug action". Proceedings of the National Academy of Sciences. 107 (40): 17152–57. Bibcode: 2010PNAS..10717152D. doi: 10.1073/pnas.1007988107. PMC 2951456. PMID 20876128.
- ^ Schlünzen F, Zarivach R, Harms J, Bashan A, Tocilj A, Albrecht R, et al. (October 2001). "Structural basis for the interaction of antibiotics with the peptidyl transferase centre in eubacteria". Nature. 413 (6858): 814–21. Bibcode: 2001Natur.413..814S. doi: 10.1038/35101544. PMID 11677599. S2CID 205022511.
- ^ Tu D, Blaha G, Moore PB, Steitz TA (April 2005). "Structures of MLSBK antibiotics bound to mutated large ribosomal subunits provide a structural explanation for resistance". Cell. 121 (2): 257–70. doi: 10.1016/j.cell.2005.02.005. PMID 15851032. S2CID 7086043.
- ^ Matzov D, Eyal Z, Benhamou RI, Shalev-Benami M, Halfon Y, Krupkin M, et al. (September 2017). "Structural insights of lincosamides targeting the ribosome of Staphylococcus aureus". Nucleic Acids Research. 45 (17): 10284–92. doi: 10.1093/nar/gkx658. PMC 5622323. PMID 28973455.
- ^ Cunha BA (2009). Infectious Diseases in Critical Care Medicine. CRC Press. p. 506. ISBN 978-1-4200-9241-7.
- ^ Cunliffe WJ, Holland KT, Bojar R, Levy SF (2002). "A randomized, double-blind comparison of a clindamycin phosphate/benzoyl peroxide gel formulation and a matching clindamycin gel with respect to microbiologic activity and clinical efficacy in the topical treatment of acne vulgaris". Clin Ther. 24 (7): 1117–33. doi: 10.1016/S0149-2918(02)80023-6. PMID 12182256.
- ^ Waknine Y (1 December 2006). "FDA Approvals: Ziana, Kadian, Polyphenon E". Medscape Medical News. Archived from the original on 6 February 2011. Retrieved 1 December 2007.
- ^ "Generic Cleocin Vaginal Availability". Drugs.com. Retrieved 13 October 2019.
- ^ (8 February 2005) "Osteomyelitis" Archived 7 January 2008 at the Wayback Machine, in Kahn, Cynthia M., Line, Scott, Aiello, Susan E. (ed.): The Merck Veterinary Manual, 9th ed., John Wiley & Sons. ISBN 0-911910-50-6. Retrieved 14 December 2007.
- ^ (8 February 2005) "Toxoplasmosis: Introduction" Archived 20 December 2007 at the Wayback Machine, in Kahn, Cynthia M., Line, Scott, Aiello, Susan E. (ed.): The Merck Veterinary Manual, 9th ed., John Wiley & Sons. ISBN 0-911910-50-6. Retrieved 14 December 2007.
External links
- Clinical trial number NCT04370548 for "DARE-BV1 in the Treatment of Bacterial Vaginosis (DARE-BVFREE)" at ClinicalTrials.gov