 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Mepron |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a693003 |
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 2.2–3.2 days |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.158.738 |
| Chemical and physical data | |
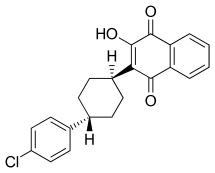
| Formula | C22H19ClO3 |
| Molar mass | 366.84 g·mol−1 |
| 3D model ( JSmol) | |
| Melting point | 216 to 219 °C (421 to 426 °F) |
| |
| |
| | |
Atovaquone, sold under the brand name Mepron, is an antimicrobial medication for the prevention and treatment of Pneumocystis jirovecii pneumonia (PCP). [2]
Atovaquone is a chemical compound that belongs to the class of naphthoquinones. Atovaquone is a hydroxy-1,4-naphthoquinone, an analog of both ubiquinone and lawsone.
Medical uses
Atovaquone is a medication used to treat or prevent:
- For pneumocystis pneumonia (PCP), [3] [4] it is used in mild cases, although it is not approved for treatment of severe cases.
- For toxoplasmosis, [5] the medication has antiparasitic and therapeutic effects.
- For malaria, it is one of the two components (along with proguanil) in the drug Malarone. Malarone has fewer side effects and is more expensive than mefloquine. [6] Resistance has been observed. [7]
- For babesia, it is often used in conjunction with oral azithromycin. [8]
Trimethoprim/sulfamethoxazole (TMP-SMX, Bactrim) is generally considered first-line therapy for PCP (not to be confused with sulfadiazine and pyrimethamine, which is first line for toxoplasmosis). However, atovaquone may be used in patients who cannot tolerate, or are allergic to, sulfonamide medications such as TMP-SMX. In addition, atovaquone has the advantage of not causing myelosuppression, which is an important issue in patients who have undergone bone marrow transplantation.[ citation needed]
Atovaquone is given prophylactically to kidney transplant patients to prevent PCP in cases where Bactrim is contraindicated for the patient.[ medical citation needed]
Malaria
Atovaquone, as a combination preparation with proguanil, has been commercially available from GlaxoSmithKline since 2000 as Malarone for the treatment and prevention of malaria.
Research
COVID-19
This section needs to be updated. (November 2022) |
Preliminary research found that atovaquone could inhibit the replication of SARS-CoV-2 in vitro. [9] Clinical trials of atovaquone for the treatment of COVID-19 are planned, [10] [11] and ongoing in United States in December 2021. [12][ needs update]
Atovaquone has also been found to inhibit human coronavirus OC43 and feline coronavirus in vitro. [13]
In newer researches, atovaquone did not demonstrate evidence of enhanced SARS-CoV-2 viral clearance compared with placebo. [14]
Veterinary use
Atovaquone is used in livestock veterinary cases of babesiosis in cattle, especially if imidocarb resistance is a concern. [15]
References
- ^ "Wellvone 750mg/5ml oral suspension - Summary of Product Characteristics (SmPC)". (emc). 28 November 2019. Retrieved 18 September 2020.
- ^ "Atovaquone Oral SUSPENSION- atovaquone suspension". DailyMed. 10 December 2019. Retrieved 18 September 2020.
- ^ Hughes W, Leoung G, Kramer F, Bozzette SA, Safrin S, Frame P, et al. (May 1993). "Comparison of atovaquone (566C80) with trimethoprim-sulfamethoxazole to treat Pneumocystis carinii pneumonia in patients with AIDS". The New England Journal of Medicine. 328 (21): 1521–1527. doi: 10.1056/NEJM199305273282103. PMID 8479489.
- ^ Dohn MN, Weinberg WG, Torres RA, Follansbee SE, Caldwell PT, Scott JD, et al. (August 1994). "Oral atovaquone compared with intravenous pentamidine for Pneumocystis carinii pneumonia in patients with AIDS. Atovaquone Study Group". Annals of Internal Medicine. 121 (3): 174–180. doi: 10.7326/0003-4819-121-3-199408010-00003. PMID 7880228. S2CID 24263604.
- ^ Djurković-Djaković O, Milenković V, Nikolić A, Bobić B, Grujić J (December 2002). "Efficacy of atovaquone combined with clindamycin against murine infection with a cystogenic (Me49) strain of Toxoplasma gondii". The Journal of Antimicrobial Chemotherapy. 50 (6): 981–987. doi: 10.1093/jac/dkf251. PMID 12461021.
- ^ Malarone: New Malaria Medication With Fewer Side-effects Archived May 14, 2006, at the Wayback Machine
- ^ Färnert A, Lindberg J, Gil P, Swedberg G, Berqvist Y, Thapar MM, et al. (March 2003). "Evidence of Plasmodium falciparum malaria resistant to atovaquone and proguanil hydrochloride: case reports". BMJ. 326 (7390): 628–629. doi: 10.1136/bmj.326.7390.628. PMC 151974. PMID 12649236.
- ^ Krause PJ, Lepore T, Sikand VK, Gadbaw J, Burke G, Telford SR, et al. (November 2000). "Atovaquone and azithromycin for the treatment of babesiosis". The New England Journal of Medicine. 343 (20): 1454–1458. doi: 10.1056/NEJM200011163432004. PMID 11078770.
- ^ Farag A, Wang P, et al. (May 2020). "Identification of Atovaquone, Ouabain and Mebendazole as FDA-Approved Drugs Targeting SARS-CoV-2". chemRxiv (preprint). doi: 10.26434/chemrxiv.12003930.v4. S2CID 219428383. Retrieved 20 June 2020.
- ^ "Atovaquone and Azithromycin Combination for Confirmed COVID-19 Infection". ClinicalTrials.gov. Retrieved 22 October 2020.
- ^ "Atovaquone for Treatment of COVID-19". ClinicalTrials.gov. Retrieved 22 October 2020.
- ^ KATHRINE EMILIE KRISTENSEN (10 December 2021). "Ny forskning finder lægemiddel mod corona: 'Kan redde menneskeliv'". B.T. (in Danish). Retrieved 10 December 2021.
- ^ Yang CW, Peng TT, Hsu HY, Lee YZ, Wu SH, Lin WH, et al. (August 2020). "Repurposing old drugs as antiviral agents for coronaviruses". Biomedical Journal. 43 (4): 368–374. doi: 10.1016/j.bj.2020.05.003. PMC 7245249. PMID 32563698.
- ^ Jain MK, De Lemos JA, McGuire DK, Ayers C, Eitson JL, Sanchez CL, et al. (2022). "Atovaquone for treatment of COVID-19: A prospective randomized, double-blind, placebo-controlled clinical trial". Frontiers in Pharmacology. 13: 1020123. doi: 10.3389/fphar.2022.1020123. PMC 9561237. PMID 36249792.
- ^ Vial HJ, Gorenflot A (May 2006). "Chemotherapy against babesiosis". Veterinary Parasitology. 138 (1–2): 147–160. doi: 10.1016/j.vetpar.2006.01.048. PMID 16504402.
Further reading
- Kessl JJ, Hill P, Lange BB, Meshnick SR, Meunier B, Trumpower BL (January 2004). "Molecular basis for atovaquone resistance in Pneumocystis jirovecii modeled in the cytochrome bc(1) complex of Saccharomyces cerevisiae". The Journal of Biological Chemistry. 279 (4): 2817–2824. doi: 10.1074/jbc.M309984200. PMID 14576156.
External links
- "Atovaquone". Drug Information Portal. U.S. National Library of Medicine.