 | |
| Clinical data | |
|---|---|
| Other names | Bestrabucil; Busramustine; KM-2210; Kregan; Estradiol 3-benzoate 17β-((4-(4-(bis(2-chloroethyl)amino)phenyl)-1-oxobutoxy)acetate; 3-Benzoyl-17β-((4-(4-(bis(2-chloroethyl)amino)phenyl)-1-oxobutoxy)acetylestradiol [1] |
| Drug class | Chemotherapeutic agent; Estrogen; Estrogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
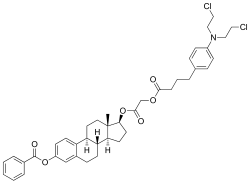
| Formula | C41H47Cl2NO6 |
| Molar mass | 720.73 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Atrimustine ( INN) (developmental code name KM-2210; former tentative brand name Kregan), also known as bestrabucil or busramustine, is a cytostatic antineoplastic agent which was under development in Japan by Kureha Chemicals (now Kureha Corporation) for the treatment of breast cancer and non-Hodgkin's lymphoma as well as for the prevention of graft-versus-host disease in bone marrow transplant recipients. [1] [2] [3] It is the benzoate ester of an ester conjugate of estradiol and chlorambucil, [4] which results in targeted/site-directed cytostatic activity toward estrogen receptor–positive tissues such as breast and bone. [5] [6] It reached preregistration for the treatment of cancer but was ultimately discontinued. [3] Estrogenicic side effects of atrimustine in clinical trials included vaginal bleeding and gynecomastia. [3] The drug was first patented in 1980. [1]
See also
References
- ^ a b c J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 897–898. ISBN 978-1-4757-2085-3.
- ^ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 438–. ISBN 978-0-8155-1856-3.
- ^ a b c "Atrimustine – AdisInsight".
- ^ The Cancer Bulletin. Medical Arts Publishing Foundation. 1987. p. 245.
- ^ Ohsawa N, Yamazaki Z, Wagatsuma T, Isurugi K (1984). "[Bestrabacil: a possible target-oriented anticancer agent]". Gan to Kagaku Ryoho (in Japanese). 11 (10): 2115–2124. PMID 6548354.
- ^ Joji Ishigami (1985). Recent Advances in Chemotherapy: Proceedings of the 14th Internat. Congress of Chemotherapy, Kyoto, 1985. Antimicrobial section; 1. 1 ,1. University of Tokyo Press. p. 52,54,471. ISBN 978-0-86008-385-6.