| |||
| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
Xenon tetrafluoride
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.033.858 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| XeF 4 | |||
| Molar mass | 207.2836 g mol−1 | ||
| Appearance | White solid | ||
| Density | 4.040 g cm−3, solid | ||
| Melting point | 117 °C (243 °F; 390 K) sublimes [1] | ||
| Reacts | |||
| Structure | |||
| D4h | |||
| square planar | |||
| 0 D | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
146 J·mol−1·K−1 [2] | ||
Std enthalpy of
formation (ΔfH⦵298) |
−251 kJ·mol−1 [2] | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Xenon tetrafluoride is a
chemical compound with
chemical formula XeF
4. It was the first discovered
binary compound of a
noble gas.
[3] It is produced by the
chemical reaction of
xenon with
fluorine:
[4]
[5]
- Xe + 2 F
2 → XeF
4
This reaction is exothermic, releasing an energy of 251 kJ/mol. [3]
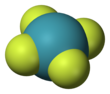
Xenon tetrafluoride is a colorless crystalline solid that sublimes at 117 °C. Its structure was determined by both NMR spectroscopy and X-ray crystallography in 1963. [6] [7] The structure is square planar, as has been confirmed by neutron diffraction studies. [8] According to VSEPR theory, in addition to four fluoride ligands, the xenon center has two lone pairs of electrons. These lone pairs are mutually trans.
Synthesis
Xenon tetrafluoride is produced by heating a mixture of xenon and fluorine in a 1:5 molar ratio in a
nickel container to 400 °C. Some
xenon difluoride (XeF
2) and
xenon hexafluoride (XeF
6) is also produced, where increased temperature or decreased fluorine concentration in the input mixture favors XeF
2 production, and decreased temperature or increased fluorine concentration favors XeF
6.
[9]
[10] The nickel is not a
catalyst for this reaction; nickel containers are used because they react with fluorine to form a protective, non-peeling
passivation layer of
nickel(II) fluoride NiF
2 on their interior surfaces. The low volatility of XeF
4 compared to XeF
2 and XeF
6 allows it to be purified by fractional sublimation.
[9]
Reactions
Xenon tetrafluoride hydrolyzes at low temperatures to form elemental xenon, oxygen, hydrofluoric acid, and aqueous xenon trioxide: [11]
It is used as a precursor for synthesis of all tetravalent Xe compounds.
[9] Reaction with
tetramethylammonium fluoride gives
tetramethylammonium pentafluoroxenate, which contains the pentagonal XeF−
5 anion. The XeF−
5 anion is also formed by reaction with
cesium fluoride:
[12]
- CsF + XeF
4 → CsXeF
5
Reaction with
bismuth pentafluoride (BiF
5) forms the XeF+
3 cation:
[13]
- BiF
5 + XeF
4 → XeF3BiF6
The XeF+
3 cation in the salt XeF3Sb2F11 has been characterized by NMR spectroscopy.
[14]
At 400 °C, XeF
4 reacts with xenon to form
XeF
2:
[10]
- XeF4 + Xe → 2 XeF2
The reaction of xenon tetrafluoride with platinum yields platinum tetrafluoride and xenon: [10]
- XeF4 + Pt → PtF4 + Xe
Applications
Xenon tetrafluoride has few applications. It has been shown to degrade
silicone rubber for analyzing trace metal impurities in the rubber. XeF
4 reacts with the silicone to form simple gaseous products, leaving a residue of metal impurities.
[15]
References
- ^ Holleman, Arnold F.; Wiberg, Egon (2001). Wiberg, Nils (ed.). Inorganic Chemistry. Translated by Eagleson, Mary; Brewer, William. Academic Press. p. 394. ISBN 0-12-352651-5.
- ^ a b Zumdahl, Steven S. (2009). Chemical Principles (6th ed.). Houghton Mifflin Company. p. A23. ISBN 978-0-618-94690-7.
- ^ a b Zumdahl (2007). Chemistry. Boston: Houghton Mifflin. p. 243. ISBN 978-0-618-52844-8.
- ^ Claassen, H. H.; Selig, H.; Malm, J. G. (1962). "Xenon Tetrafluoride". J. Am. Chem. Soc. 84 (18): 3593. doi: 10.1021/ja00877a042.
- ^ Chernick, C. L.; Claassen, H. H.; Fields, P. R.; Hyman, H. H.; Malm, J. G.; Manning, W. M.; Matheson, M. S.; Quarterman, L. A.; Schreiner, F.; Selig, H. H.; Sheft, I.; Siegel, S.; Sloth, E. N.; Stein, L.; Studier, M. H.; Weeks, J. L.; Zirin, M. H. (1962). "Fluorine Compounds of Xenon and Radon". Science. 138 (3537): 136–138. Bibcode: 1962Sci...138..136C. doi: 10.1126/science.138.3537.136. PMID 17818399. S2CID 10330125.
- ^ Brown, Thomas H.; Whipple, E. B.; Verdier, Peter H. (1963). "Xenon Tetrafluoride: Fluorine-19 High-Resolution Magnetic Resonance Spectrum". Science. 140 (3563): 178. Bibcode: 1963Sci...140..178B. doi: 10.1126/science.140.3563.178. PMID 17819836. S2CID 35981023.
- ^ Ibers, James A.; Hamilton, Walter C. (1963). "Xenon Tetrafluoride: Crystal Structure". Science. 139 (3550): 106–107. Bibcode: 1963Sci...139..106I. doi: 10.1126/science.139.3550.106. PMID 17798707. S2CID 42119788.
- ^ Burns, John H.; Agron, P. A.; Levy, Henri A (1963). "Xenon Tetrafluoride Molecule and Its Thermal Motion: A Neutron Diffraction Study". Science. 139 (3560): 1208–1209. Bibcode: 1963Sci...139.1208B. doi: 10.1126/science.139.3560.1208. PMID 17757912. S2CID 35858682.
- ^ a b c Haner, Jamie; Schrobilgen, Gary J. (2015). "The Chemistry of Xenon(IV)". Chem. Rev. 115 (2): 1255–1295. doi: 10.1021/cr500427p. ISSN 0009-2665. PMID 25559700.
- ^ a b c Bard, Allen J.; Parsons, Roger; Jordan, Joseph; International Union of Pure and Applied Chemistry (1985). Standard Potentials in Aqueous Solution. CRC Press. pp. 767–768. ISBN 0-8247-7291-1.
- ^ Williamson; Koch, C. W. (Mar 1963). "Xenon Tetrafluoride: Reaction with Aqueous Solutions". Science. 139 (3559): 1046–1047. Bibcode: 1963Sci...139.1046W. doi: 10.1126/science.139.3559.1046. ISSN 0036-8075. PMID 17812981. S2CID 33320384.
- ^ Harding, Charlie; Johnson, David Arthur; Janes, Rob (2002). Elements of the p Block. Molecular World. Vol. 9. Royal Society of Chemistry. p. 93. ISBN 0-85404-690-9.
- ^ Suzuki, Hitomi; Matano, Yoshihiro (2001). Organobismuth chemistry. Elsevier. p. 8. ISBN 0-444-20528-4.
-
^ Gillespie, R. J.; Landa, B.; Schrobilgen, G. J. (1971). "Trifluoroxenon(IV) µ-fluoro-bispentafluoroantimonate(V): the XeF+
3 cation". Journal of the Chemical Society D: Chemical Communications (23): 1543–1544. doi: 10.1039/C29710001543. - ^ Rigin, V.; Skvortsov, N. K.; Rigin, V. V. (March 1997). "Xenon tetrafluoride as a decomposition agent for silicone rubber for isolation and atomic emission spectrometric determination of trace metals". Analytica Chimica Acta. 340 (1–3): 1–3. Bibcode: 1997AcAC..340....1R. doi: 10.1016/S0003-2670(96)00563-6.