| |
| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| HgF4 | |
| Molar mass | 276.58 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
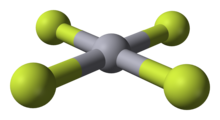
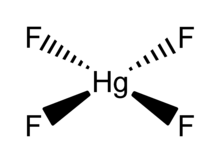
Mercury(IV) fluoride, HgF4, is the first mercury compound to be reported with mercury in the +4 oxidation state. Mercury, like the other group 12 elements ( cadmium and zinc), has an s2d10 electron configuration and generally only forms bonds involving its 6s orbital. This means that the highest oxidation state mercury normally attains is +2, and for this reason it is sometimes considered a post-transition metal instead of a transition metal. HgF4 was first reported from experiments in 2007, but its existence remains disputed; experiments conducted in 2008 could not replicate the compound. [1] [2]
History
Speculation about higher oxidation states for mercury had existed since the 1970s, and theoretical calculations in the 1990s predicted that it should be stable in the gas phase, with a square-planar geometry consistent with a formal d8 configuration. However, experimental proof remained elusive until 2007, when HgF4 was first prepared using solid neon and argon for matrix isolation at a temperature of 4 K. The compound was detected using infrared spectroscopy. [3] [4] Analysis of density functional theory and coupled cluster calculations showed that the d orbitals are involved in bonding. This has led to the suggestion that mercury should be considered a transition metal after all (the group 12 metals are sometimes not included as transition metals because they do not have oxidation states beyond +2). [5] However, that conclusion has been challenged by William B. Jensen with the argument that HgF4 only exists under highly atypical non-equilibrium conditions and should best be considered as an exception. [6]
Explanation
Theoretical studies suggest that mercury is unique among the natural elements of group 12 in forming a tetrafluoride, and attribute this observation to relativistic effects. According to calculations, the tetrafluorides of the "less relativistic" elements cadmium and zinc are unstable and eliminate a fluorine molecule, F2, to form the metal difluoride complex. On the other hand, the tetrafluoride of the "more relativistic" synthetic element 112, copernicium, is predicted to be more stable. [7] However, more recent theoretical studies cast doubt on the possible existence of mercury(IV) and even copernicium(IV) fluoride. [8]
Synthesis and properties
HgF4 is produced by the reaction of elemental mercury with fluorine:
- Hg + 2 F2 → HgF4
HgF4 is only stable in matrix isolation at 4 K (−269 °C); upon heating, or if the HgF4 molecules touch each other, it decomposes to mercury(II) fluoride and fluorine:
- HgF4 → HgF2 + F2
HgF4 is a
diamagnetic, square planar molecule. The mercury atom has a formal 6s25d86p6 electron configuration, and as such obeys the
octet rule but not the
18-electron rule. HgF4 is
isoelectronic with the
tetrafluoroaurate anion, AuF−
4, and is valence isoelectronic with the
tetrachloroaurate (AuCl−
4),
tetrabromoaurate (AuBr−
4), and
tetrachloroplatinate (PtCl2−
4) anions.
References
- ^ Is mercury a transition metal? Archived 2016-10-12 at the Wayback Machine
- ^ Rooms, John F.; Wilson, Antony V.; Harvey, Ian; Bridgeman, Adam J.; Young, Nigel A. (2008). "Mercury–fluorine interactions: a matrix isolation investigation of Hg···F2, HgF2 and HgF4 in argon matrices". Phys. Chem. Chem. Phys. 10 (31): 4594–4605. doi: 10.1039/B805608K. PMID 18665309.
- ^ "High Oxidation States: Mercury tetrafluoride synthesized". Archived from the original on 2011-07-19. Retrieved 2008-06-26.
- ^ "Elusive Hg(IV) species has been synthesized under cryogenic conditions". 2007-10-12.
- ^ Wang, Xuefang; Andrews, Lester; Riedel, Sebastian; Kaupp, Martin (2007). "Mercury Is a Transition Metal: The First Experimental Evidence for HgF4". Angew. Chem. Int. Ed. 46 (44): 8371–8375. doi: 10.1002/anie.200703710. PMID 17899620.
- ^ Jensen, William B. (2008). "Is Mercury Now a Transition Element?". J. Chem. Educ. 85 (9): 1182–1183. Bibcode: 2008JChEd..85.1182J. doi: 10.1021/ed085p1182.
- ^ Hoffman, Darleane C.; Lee, Diana M.; Pershina, Valeria (2006). "Transactinides and the future elements". In Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. ISBN 1-4020-3555-1.
- ^ Brändas, Erkki J.; Kryachko, Eugene S. (2013-03-09). Fundamental World of Quantum Chemistry: A Tribute to the Memory of Per-Olov Löwdin. Springer. ISBN 9789401704489.