| |

| |
| Names | |
|---|---|
|
IUPAC name
Tetrabromophosphanium tribromide
| |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| PBr7 | |
| Molar mass | 590.302 g·mol−1 |
| Appearance | Red prismatic crystals |
| Structure [1] | |
| Orthorhombic | |
| Pnma, No. 64 | |
a = 9.35 Å, b = 7.94 Å, c = 14.69 Å
| |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
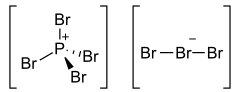
Phosphorus heptabromide is an inorganic compound with the chemical formula P Br7. It is one of the phosphorus bromides. At normal conditions, it forms red prismatic crystals. PBr7 can be prepared by the sublimation of a mixture of phosphorus pentabromide and bromine. [2]
- PBr5 + Br2 → PBr7
The structure of PBr7 consists of a tetrabromophosphonium cation [PBr4+, paired with a tribromide anion [Br3−, and the tribromide anion is non-symmetric. [1]
See also
References
- ^ a b Breneman, G. L.; Willett, R. D. (1967). "The crystal structure of phosphorus heptabromide, PBr7". Acta Crystallographica. 23 (3): 467–471. doi: 10.1107/S0365110X67002981.
- ^ T. E. (Thomas Edward) Thorpe. A dictionary of applied chemistry (Volume 4)