| |
| Names | |
|---|---|
| Other names
sodium metagermanate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.031.535 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Na2GeO3 | |
| Molar mass | 166.62 g/mol |
| Appearance | white solid |
| Odor | odorless |
| Density | 3.31 g/cm3 |
| Melting point | 1,060 °C (1,940 °F; 1,330 K) |
| 14.4 g/100 mL (0 °C) 23.8 g/100 mL (25 °C) | |
Refractive index (nD)
|
1.59 |
| Structure | |
| orthorhombic | |
| Related compounds | |
Related compounds
|
Sodium silicate |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium germanate is an inorganic compound with the chemical formula Na2GeO3. It exists as a colorless solid. Sodium germanate is primarily used for the synthesis of other germanium compounds.
Preparation and reactions
Sodium germanate can be prepared by the fusion of germanium oxide with sodium hydroxide at high temperatures:
- 2 NaOH + GeO2 → Na2GeO3 + H2O
An intermediate in this reaction is the protonated derivative NaHGeO3, which is a water-soluble salt.
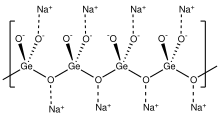
Structure
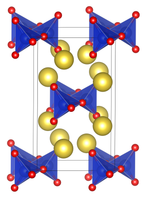
it is structurally analogous to sodium metasilicate, Na2SiO3, consisting of polymeric GeO32− anions made up of vertex sharing {GeO4} tetrahedra. [1] [2]
See also
References
- ^ Cruickshank, D. W. J.; Kálmán, A.; Stephens, J. S. (1978). "A Reinvestigation of Sodium Metagermanate". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry. 34 (4): 1333–1334. Bibcode: 1978AcCrB..34.1333C. doi: 10.1107/S0567740878005488.
- ^ C. C. Addison, Inorganic Chemistry of the Main-Group Elements, vol 1, 1973, The chemical Society, ISBN 9780851867526