 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
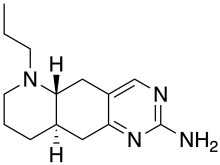
| Formula | C14H22N4 |
| Molar mass | 246.358 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Quinelorane is a drug which acts as a dopamine agonist for the D2 [1] [2] [3] and D3 receptor.
See also
References
- ^ Caine SB, Negus SS, Mello NK, Patel S, Bristow L, Kulagowski J, Vallone D, Saiardi A, Borrelli E (April 2002). "Role of dopamine D2-like receptors in cocaine self-administration: studies with D2 receptor mutant mice and novel D2 receptor antagonists". Journal of Neuroscience. 22 (7): 2977–88. doi: 10.1523/JNEUROSCI.22-07-02977.2002. PMC 6758322. PMID 11923462.
- ^ Gasior M, Paronis CA, Bergman J (January 2004). "Modification by dopaminergic drugs of choice behavior under concurrent schedules of intravenous saline and food delivery in monkeys". The Journal of Pharmacology and Experimental Therapeutics. 308 (1): 249–59. doi: 10.1124/jpet.103.052795. PMID 14563783. S2CID 5879400.
- ^ Brooks JM, Sarter M, Bruno JP (September 2007). "D2-like receptors in nucleus accumbens negatively modulate acetylcholine release in prefrontal cortex". Neuropharmacology. 53 (3): 455–63. doi: 10.1016/j.neuropharm.2007.06.006. PMC 2000917. PMID 17681559.