 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.001.124 |
| Chemical and physical data | |
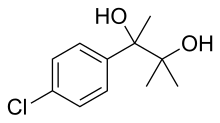
| Formula | C11H15ClO2 |
| Molar mass | 214.69 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Phenaglycodol (brand names Acalmid, Acalo, Alterton, Atadiol, Felixyn, Neotran, Pausital, Remin, Sedapsin, Sinforil, Stesil, Ultran) [2] is a drug described as a tranquilizer or sedative which has anxiolytic and anticonvulsant properties. [3] [4] It is related pharmacologically to meprobamate, though it is not a carbamate. [5] [6]
Synthesis

p-Chloroacetophenone and NaCN are reacted together to give the corresponding cyanohydrin (cf Strecker synthesis), CID:12439573. The cyano group is then hydrated in acid to the corresponding amide, p-chloroatrolactamide, CID:15255544 (4). The amide group is then further hydrolyzed with a 2nd equivalent of water in concentrated lye to p-chloroatrolactic acid, [4445-13-0] (5). Esterification to Ethyl p-chloroatrolactate [100126-96-3](6). Finally, nucleophilic addition a couple of equivalents of MeMgI are added to the ester give Phenaglycodol (7) crystals.

A mixed Pinacol coupling rxn between 4-chloroacetophenone [99-91-2] and acetone with magnesium activated with a small amount of trimethylsilyl chloride gave a 40% yield of phenglycodol.
Notes
- See "Novel trifluoromethyl derivatives of substituted diols" U.S. patent 3,134,819 also.
- A Pinacol rearrangement occurs in acidic water: [10]
See also
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ Usdin E, Efron DH, U.S. National Institute of Mental Health (1972). Psychotropic drugs and related compounds. National Institute of Mental Health; [for sale by the Supt. of Docs., U.S. Govt. Print. Off., Washington. ISBN 9780080255101.
- ^ Vida J (19 July 2013). Anticonvulsants. Elsevier. pp. 578–. ISBN 978-0-323-14395-0.
- ^ Haddad LM, Winchester JF (1983). Clinical Management of Poisoning and Drug Overdose. Saunders. ISBN 978-0-7216-4447-9.
- ^ Drill VA (1958). Pharmacology in Medicine: A Collaborative Textbook. McGraw-Hill.
- ^ Beckman H (1961). Pharmacology; the nature, action and use of drugs. Saunders.
- ^ Jack Mills "2-chlorophenyl-3-methyl-2, 3-butanediols" U.S. patent 2,812,363 (1957 to Eli Lilly Co.)
- ^ GB 788896, Mills, Jack, "Improvements in or relating to novel substituted butanediol", published 1958-01-08, assigned to Eli Lilly & Co.
- ^ Maekawa, Hirofumi; Yamamoto, Yoshimasa; Shimada, Hisashi; Yonemura, Kazuaki; Nishiguchi, Ikuzo (2004). "Mg-promoted mixed pinacol coupling". Tetrahedron Letters. 45 (20): 3869–3872. doi:10.1016/j.tetlet.2004.03.109.
- ^ Murphy, Hubert W. (1964). "Pinacol Rearrangement of Phenaglycodol I". Journal of Pharmaceutical Sciences. 53 (3): 298–301. doi:10.1002/jps.2600530311.