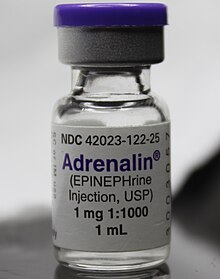
Epinephrine, also known as adrenaline, is a medication and hormone. [7] [8] As a medication, it is used to treat several conditions, including anaphylaxis, cardiac arrest, asthma, and superficial bleeding. [5] Inhaled epinephrine may be used to improve the symptoms of croup. [9] It may also be used for asthma when other treatments are not effective. [5] It is given intravenously, by injection into a muscle, by inhalation, or by injection just under the skin. [5]
Common side effects include shakiness, anxiety, and sweating. [5] A fast heart rate and high blood pressure may occur. [5] Occasionally, it may result in an abnormal heart rhythm. [5] While the safety of its use during pregnancy and breastfeeding is unclear, the benefits to the mother must be taken into account. [5]
Epinephrine is normally produced by both the adrenal glands and a small number of neurons in the brain, where it acts as a neurotransmitter. [7] [10] It plays an essential role in the fight-or-flight response by increasing blood flow to muscles, heart output, pupil dilation, and blood sugar. [11] [12] Epinephrine does this through its effects on alpha and beta receptors. [12] It is found in many animals and some single-celled organisms, [13] [14] but the medication is produced synthetically and is not harvested from animals. [15]
Jōkichi Takamine first isolated epinephrine in 1901, and it came into medical use in 1905. [16] [17] It is on the World Health Organization's List of Essential Medicines. [18] It is available as a generic medication. [5] In 2021, it was the 221st most commonly prescribed medication in the United States, with more than 1 million prescriptions. [19] [20]
Medical uses
Epinephrine is used to treat a number of conditions, including cardiac arrest, anaphylaxis, and superficial bleeding. [21] It has been used historically for bronchospasm and low blood sugar, but newer treatments for these that are selective for β2 adrenoceptors, such as salbutamol, are currently preferred.[ citation needed]
Heart problems
While epinephrine is often used to treat cardiac arrest, it has not been shown to improve long-term survival or mental function after recovery. [22] [23] [24] It does, however, improve return of spontaneous circulation. [24] When used intravenously to treat cardiac arrest in adults, epinephrine is typically given at a dose of 1 mg every three to five minutes. [25]
In cases of IV infusion for bradycardia the dose is 1 mg epinephrine is mixed with 500ml of NS or D5W. The infusion should run at 2-10 micrograms/min (titrated to effect). IV infusion for post-cardiac arrest hypotension: The dosing is 2 to 10 micrograms/min.
Endotracheal Tube: 2-2.5 mg epinephrine is diluted in 10cc NS and given directly into the ET tube. [26]
Anaphylaxis
Epinephrine is the drug of choice for treating allergic reaction anaphylaxis. The commonly used epinephrine autoinjector delivers a 0.3 mg epinephrine injection (0.3 mL, 1:1000). It is indicated in the emergency treatment of allergic reactions, including anaphylaxis to stings, contrast agents, medicines, or people with a history of anaphylactic reactions to known triggers. A single dose is recommended for people who weigh 30 kg or more, repeated if necessary. A lower-strength product is available for children. [27] [28] [29] [30]
Intramuscular injection can be complicated in that the depth of subcutaneous fat varies and may result in subcutaneous injection, or may be injected intravenously in error, or the wrong strength used. [31] Intramuscular injection gives a faster and higher pharmacokinetic profile compared to subcutaneous injection. [32]
Asthma
Epinephrine is also used as a bronchodilator for asthma if specific β2 agonists are unavailable or ineffective. [33]
When given by the subcutaneous or intramuscular routes for asthma, an appropriate dose is 0.3 to 0.5 mg. [34] [35]
Because of the high intrinsic efficacy (receptor binding ability) of epinephrine, high drug concentrations cause adverse side effects when treating asthma. The value of using nebulized epinephrine in acute asthma is unclear. [36]
Croup
Racemic epinephrine has historically been used for the treatment of croup. [37] [38] Regular epinephrine, however, works equally well[ fact or opinion?][ medical citation needed]. Racemic adrenaline is a 1:1 mixture of the two enantiomers of adrenaline. [39] The L-form is the active component. [39] Racemic adrenaline works by stimulating the alpha-adrenergic receptors in the airway, with resultant mucosal vasoconstriction and decreased subglottic edema, and by stimulating the β adrenergic receptors, with resultant relaxation of the bronchial smooth muscle. [38]
Bronchiolitis
There is a lack of consensus as to whether inhaled nebulized epinephrine is beneficial in the treatment of bronchiolitis, with most guidelines recommending against its use. [40]
Local anesthetics
When epinephrine is mixed with local anesthetics, such as bupivacaine or lidocaine, and used for local anesthesia or intrathecal injection, it prolongs the numbing effect and motor block effect of the anesthetic by up to an hour. [41] Epinephrine is frequently combined with local anesthetic and can cause panic attacks. [42]
Epinephrine is mixed with cocaine to form Moffett's solution, used in nasal surgery. [43]
Upper airway obstruction
Upper airway obstruction with edema and stridor can be treated with racemic epinephrine. [44]
Adverse effects
Adverse reactions to adrenaline include palpitations, tachycardia, arrhythmia, anxiety, panic attack, headache, anorexia, tremor, hypertension, and acute pulmonary edema. The use of epinephrine based eye-drops, commonly used to treat glaucoma, may also lead to a buildup of adrenochrome pigments in the conjunctiva, iris, lens, and retina.
Rarely, exposure to medically administered epinephrine may cause Takotsubo cardiomyopathy. [45]
Use is contraindicated in people on nonselective β-blockers because severe hypertension and even cerebral hemorrhage may result. [46]
Mechanism of action
| Organ | Effects |
|---|---|
| Heart | Increases heart rate; contractility; conduction across AV node |
| Lungs | Increases respiratory rate; bronchodilation |
| Liver | Stimulates glycogenolysis |
| Brain | |
| Systemic | Vasoconstriction and vasodilation |
| Triggers lipolysis | |
| Muscle contraction |
As a hormone, epinephrine acts on nearly all body tissues. Its actions vary by tissue type and tissue expression of adrenergic receptors. For example, high epinephrine levels cause smooth muscle relaxation in the airways but cause contraction of the smooth muscle that lines most arterioles.[ citation needed]
Epinephrine acts by binding to a variety of adrenergic receptors. Epinephrine is a nonselective agonist of all adrenergic receptors, including the major subtypes α1, α2, β1, β2, and β3. [46] Epinephrine's binding to these receptors triggers several metabolic changes. Binding to α-adrenergic receptors inhibits insulin secretion by the pancreas, stimulates glycogenolysis in the liver and muscle, [47] and stimulates glycolysis and inhibits insulin-mediated glycogenesis in muscle. [48] [49] β adrenergic receptor binding triggers glucagon secretion in the pancreas, increased adrenocorticotropic hormone (ACTH) secretion by the pituitary gland, and increased lipolysis by adipose tissue. Together, these effects increase blood glucose and fatty acids, providing substrates for energy production within cells throughout the body. [49] In the heart, the coronary arteries have a predominance of β2 receptors, which cause vasodilation of the coronary arteries in the presence of epinephrine. [50]
Its actions increase peripheral resistance via α1 receptor-dependent vasoconstriction and increase cardiac output via its binding to β1 receptors. The goal of reducing peripheral circulation is to increase coronary and cerebral perfusion pressures and therefore increase oxygen exchange at the cellular level. [51] While epinephrine does increase aortic, cerebral, and carotid circulation pressure, it lowers carotid blood flow and end-tidal CO2 or ETCO2 levels. It appears that epinephrine may improve macrocirculation at the expense of the capillary beds where perfusion takes place. [52]
History
Extracts of the adrenal gland were first obtained by Polish physiologist Napoleon Cybulski in 1895. These extracts, which he called nadnerczyna, contained adrenaline and other catecholamines. [53] American ophthalmologist William H. Bates discovered adrenaline's usage for eye surgeries prior to 20 April 1896. [54] Japanese chemist Jōkichi Takamine and his assistant Keizo Uenaka independently discovered adrenaline in 1900. [55] [56] In 1901, Takamine successfully isolated and purified the hormone from the adrenal glands of sheep and oxen. [57] Adrenaline was first synthesized in the laboratory by Friedrich Stolz and Henry Drysdale Dakin, independently, in 1904. [56]
Society and culture
Legal status
In June 2024, the Committee for Medicinal Products for Human Use of the European Medicines Agency adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Eurneffy, intended for emergency treatment of allergic reactions (anaphylaxis) due to insect stings or bites, foods, medicinal products and other allergens as well as idiopathic or exercise induced anaphylaxis. [58] [59] [60] The applicant for this medicinal product is ARS Pharmaceuticals IRL Limited. [58]
Brand names
Common brand names include Asthmanefrin, Micronefrin, Nephron, VapoNefrin, and Primatene Mist.
Delivery forms
Epinephrine is available in an autoinjector delivery system.
There is an epinephrine metered-dose inhaler sold over the counter in the United States to relieve bronchial asthma. [61] [62] It was introduced in 1963 by Armstrong Pharmaceuticals. [63]
A common concentration for epinephrine is 2.25% w/v epinephrine in solution, which contains 22.5 mg/mL, while a 1% solution is typically used for aerosolization.
- Adults: 0.5–0.75 ml of a 2.25% solution in 2.0 ml normal saline. [64]
- Pediatrics: 0.25–0.75 ml of a 2.25% solution in 2.0 ml normal saline. [64] [65]
References
- ^ Andersen AM (1975). "Structural Studies of Metabolic Products of Dopamine. III. Crystal and Molecular Structure of (−)-Adrenaline". Acta Chem. Scand. 29b (2): 239–244. doi: 10.3891/acta.chem.scand.29b-0239. PMID 1136652.
- ^ El-Bahr SM, Kahlbacher H, Patzl M, Palme RG (May 2006). "Binding and clearance of radioactive adrenaline and noradrenaline in sheep blood". Veterinary Research Communications. 30 (4). Springer Science and Business Media LLC: 423–32. doi: 10.1007/s11259-006-3244-1. PMID 16502110. S2CID 9054777.
- ^ Franksson G, Anggård E (13 March 2009). "The plasma protein binding of amphetamine, catecholamines and related compounds". Acta Pharmacologica et Toxicologica. 28 (3). Wiley: 209–14. doi: 10.1111/j.1600-0773.1970.tb00546.x. PMID 5468075.
- ^ Peaston RT, Weinkove C (January 2004). "Measurement of catecholamines and their metabolites". Annals of Clinical Biochemistry. 41 (Pt 1). SAGE Publications: 17–38. doi: 10.1258/000456304322664663. PMID 14713382. S2CID 2330329.
- ^ a b c d e f g h i "Epinephrine". The American Society of Health-System Pharmacists. Archived from the original on 6 September 2015. Retrieved 15 August 2015.
- ^ Hummel MD (2012). "Emergency Medications". In Pollak AN (ed.). Nancy Caroline's Emergency Care in the Streets (7th ed.). Burlington: Jones & Bartlett Learning. p. 557. ISBN 9781449645861. Archived from the original on 8 September 2017.
- ^ a b Lieberman M, Marks A, Peet A (2013). Marks' Basic Medical Biochemistry: A Clinical Approach (4 ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 175. ISBN 9781608315727. Archived from the original on 8 September 2017.
- ^ "(-)-adrenaline". Guide to Pharmacology. IUPS/BPS. Archived from the original on 1 September 2015. Retrieved 21 August 2015.
- ^ Everard ML (February 2009). "Acute bronchiolitis and croup". Pediatric Clinics of North America. 56 (1): 119–33, x–xi. doi: 10.1016/j.pcl.2008.10.007. PMID 19135584.
-
^ Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 6: Widely Projecting Systems: Monoamines, Acetylcholine, and Orexin". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, USA: McGraw-Hill Medical. p. 157.
ISBN
9780071481274.
Epinephrine occurs in only a small number of central neurons, all located in the medulla. Epinephrine is involved in visceral functions, such as the control of respiration. It is also produced by the adrenal medulla.
- ^ Bell DR (2009). Medical physiology : principles for clinical medicine (3rd ed.). Philadelphia: Lippincott Williams & Wilkins. p. 312. ISBN 9780781768528. Archived from the original on 8 September 2017.
- ^ a b Khurana (2008). Essentials of Medical Physiology. Elsevier India. p. 460. ISBN 9788131215661. Archived from the original on 8 September 2017.
- ^ Buckley E (2013). Venomous Animals and Their Venoms: Venomous Vertebrates. Elsevier. p. 478. ISBN 9781483262888. Archived from the original on 8 September 2017.
- ^ Animal Physiology: Adaptation and Environment (5 ed.). Cambridge University Press. 1997. p. 510. ISBN 9781107268500. Archived from the original on 8 September 2017.
- ^ "Epinephrine".
- ^ Wermuth CG (2008). The practice of medicinal chemistry (3 ed.). Amsterdam: Elsevier/Academic Press. p. 13. ISBN 9780080568775. Archived from the original on 8 September 2017.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 541. ISBN 9783527607495.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl: 10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Epinephrine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "Epinephrine". The American Society of Health-System Pharmacists. Archived from the original on 7 March 2011. Retrieved 3 April 2011.
- ^ Kempton H, Vlok R, Thang C, Melhuish T, White L (March 2019). "Standard dose epinephrine versus placebo in out of hospital cardiac arrest: A systematic review and meta-analysis". The American Journal of Emergency Medicine. 37 (3): 511–517. doi: 10.1016/j.ajem.2018.12.055. PMID 30658877. S2CID 58580872. Archived from the original on 28 August 2021. Retrieved 27 January 2019.
- ^ Reardon PM, Magee K (2013). "Epinephrine in out-of-hospital cardiac arrest: A critical review". World Journal of Emergency Medicine. 4 (2): 85–91. doi: 10.5847/wjem.j.issn.1920-8642.2013.02.001. PMC 4129833. PMID 25215099.
- ^ a b Lin S, Callaway CW, Shah PS, Wagner JD, Beyene J, Ziegler CP, et al. (June 2014). "Adrenaline for out-of-hospital cardiac arrest resuscitation: a systematic review and meta-analysis of randomized controlled trials". Resuscitation. 85 (6): 732–40. doi: 10.1016/j.resuscitation.2014.03.008. PMID 24642404.
- ^ Link MS, Berkow LC, Kudenchuk PJ, Halperin HR, Hess EP, Moitra VK, et al. (November 2015). "Part 7: Adult Advanced Cardiovascular Life Support: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care". Circulation. 132 (18 Suppl 2): S444-64. doi: 10.1161/CIR.0000000000000261. PMID 26472995.
- ^ "ACLS and epinephrine".
- ^ Mylan Specialty L.P. "EPIPEN®- epinephrine injection, EPIPEN Jr®- epinephrine injection" (PDF). FDA Product Label. Archived (PDF) from the original on 1 February 2014. Retrieved 22 January 2014.
- ^ ECC Committee, Subcommittees and Task Forces of the American Heart Association (2005). "2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Part 10.6: Anaphylaxis". Circulation. 112 (24 suppl): IV–143–IV–145. doi: 10.1161/circulationaha.105.166568.
- ^ Neumar RW, Otto CW, Link MS, Kronick SL, Shuster M, Callaway CW, et al. (November 2010). "Part 8: adult advanced cardiovascular life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care". Circulation. 122 (18 Suppl 3): S729–67. doi: 10.1161/CIRCULATIONAHA.110.970988. PMID 20956224.
- ^ Lieberman P, Nicklas RA, Oppenheimer J, Kemp SF, Lang DM, Bernstein DI, et al. (September 2010). "The diagnosis and management of anaphylaxis practice parameter: 2010 update". The Journal of Allergy and Clinical Immunology. 126 (3): 477–80.e1–42. doi: 10.1016/j.jaci.2010.06.022. PMID 20692689.
- ^ Pennsylvania Patient Advisory. "Let's Stop this "Epi"demic!—Preventing Errors with Epinephrine". Archived from the original on 2 February 2014. Retrieved 22 January 2014.
- ^ McLean-Tooke AP, Bethune CA, Fay AC, Spickett GP (December 2003). "Adrenaline in the treatment of anaphylaxis: what is the evidence?". BMJ. 327 (7427): 1332–5. doi: 10.1136/bmj.327.7427.1332. PMC 286326. PMID 14656845.
- ^ Koninckx M, Buysse C, de Hoog M (June 2013). "Management of status asthmaticus in children". Paediatric Respiratory Reviews. 14 (2): 78–85. doi: 10.1016/j.prrv.2013.03.003. PMID 23578933.
- ^ Soar, Perkins, et al (2010) European Resuscitation Council Guidelines for Resuscitation 2010 Section 8. Cardiac arrest in special circumstances: Electrolyte abnormalities, poisoning, drowning, accidental hypothermia, hyperthermia, asthma, anaphylaxis, cardiac surgery, trauma, pregnancy, electrocution. Resuscitation. Oct. pp.1400–1433
- ^ Fisher, Brown, Cooke (Eds) (2006) Joint Royal Colleges Ambulance Liaison Committee. UK Ambulance Clinical Practice Guidelines.
- ^ Abroug F, Dachraoui F, Ouanes-Besbes L (March 2016). "Our paper 20 years later: the unfulfilled promises of nebulised adrenaline in acute severe asthma". Intensive Care Medicine. 42 (3): 429–31. doi: 10.1007/s00134-016-4210-1. PMID 26825950. S2CID 37328426.
- ^ Bjornson CL, Johnson DW (January 2008). "Croup". Lancet. 371 (9609): 329–39. doi: 10.1016/S0140-6736(08)60170-1. PMC 7138055. PMID 18295000.
- ^ a b Thomas LP, Friedland LR (January 1998). "The cost-effective use of nebulized racemic epinephrine in the treatment of croup". The American Journal of Emergency Medicine. 16 (1): 87–9. doi: 10.1016/S0735-6757(98)90073-0. PMID 9451322.
- ^ a b Malhotra A, Krilov LR (January 2001). "Viral croup". Pediatrics in Review. 22 (1): 5–12. doi: 10.1542/pir.22-1-5. PMID 11139641. S2CID 41978318.
- ^ Kirolos A, Manti S, Blacow R, Tse G, Wilson T, Lister M, et al. (August 2019). "A Systematic Review of Clinical Practice Guidelines for the Diagnosis and Management of Bronchiolitis". J. Infect. Dis. 222 (Supplement_7): S672–S679. doi: 10.1093/infdis/jiz240. hdl: 20.500.11820/7d4708e3-7cdc-49f7-a9b3-a29040f4ff4e. PMID 31541233.
- ^ Tschopp C, Tramèr MR, Schneider A, Zaarour M, Elia N (July 2018). "Benefit and Harm of Adding Epinephrine to a Local Anesthetic for Neuraxial and Locoregional Anesthesia: A Meta-analysis of Randomized Controlled Trials With Trial Sequential Analyses" (PDF). Anesth. Analg. 127 (1): 228–239. doi: 10.1213/ANE.0000000000003417. PMID 29782398. S2CID 29154283.
- ^ Rahn R, Ball B (2001). Local Anesthesia in Dentistry: Articaine and Epinephrine for Dental Anesthesia (1 st ed.). Seefeld, Germany: 3M ESPE. p. 44. ISBN 978-3-00-008562-8.
- ^ Benjamin E, Wong DK, Choa D (December 2004). "'Moffett's' solution: a review of the evidence and scientific basis for the topical preparation of the nose". Clinical Otolaryngology and Allied Sciences. 29 (6): 582–7. doi: 10.1111/j.1365-2273.2004.00894.x. PMID 15533141.
- ^ Markovchick V (2007). Critical Care Secrets (fourth ed.).
- ^ Nazir S, Lohani S, Tachamo N, Ghimire S, Poudel DR, Donato A (February 2017). "Takotsubo cardiomyopathy associated with epinephrine use: A systematic review and meta-analysis". Int. J. Cardiol. 229: 67–70. doi: 10.1016/j.ijcard.2016.11.266. PMID 27889211.
- ^ a b Shen, Howard (2008). Illustrated Pharmacology Memory Cards: PharMnemonics. Minireview. p. 4. ISBN 978-1-59541-101-3.
- ^ Arnall DA, Marker JC, Conlee RK, Winder WW (June 1986). "Effect of infusing epinephrine on liver and muscle glycogenolysis during exercise in rats". The American Journal of Physiology. 250 (6 Pt 1): E641–9. doi: 10.1152/ajpendo.1986.250.6.E641. PMID 3521311.
- ^ Raz I, Katz A, Spencer MK (March 1991). "Epinephrine inhibits insulin-mediated glycogenesis but enhances glycolysis in human skeletal muscle". The American Journal of Physiology. 260 (3 Pt 1): E430–5. doi: 10.1152/ajpendo.1991.260.3.E430. PMID 1900669.
- ^ a b Sabyasachi Sircar (2007). Medical Physiology. Thieme Publishing Group. p. 536. ISBN 978-3-13-144061-7.
- ^ Sun D, Huang A, Mital S, Kichuk MR, Marboe CC, Addonizio LJ, et al. (July 2002). "Norepinephrine elicits beta2-receptor-mediated dilation of isolated human coronary arterioles". Circulation. 106 (5): 550–5. doi: 10.1161/01.CIR.0000023896.70583.9F. PMID 12147535.
- ^ "Guideline 11.5: Medications in Adult Cardiac Arrest" (PDF). Australian Resuscitation Council. December 2010. Archived from the original on 12 March 2015. Retrieved 7 March 2015.
- ^ Burnett AM, Segal N, Salzman JG, McKnite MS, Frascone RJ (August 2012). "Potential negative effects of epinephrine on carotid blood flow and ETCO2 during active compression-decompression CPR utilizing an impedance threshold device". Resuscitation. 83 (8): 1021–4. doi: 10.1016/j.resuscitation.2012.03.018. PMID 22445865.
- ^ Skalski JH, Kuch J (April 2006). "Polish thread in the history of circulatory physiology". Journal of Physiology and Pharmacology. 57 (Suppl 1): 5–41. PMID 16766800. Archived from the original on 10 March 2011.
- ^ Bates WH (16 May 1896). "The Use of Extract of Suprarenal Capsule in the Eye". New York Medical Journal: 647–650. Archived from the original on 3 April 2015. Retrieved 7 March 2015.
- ^ Yamashima T (May 2003). "Jokichi Takamine (1854-1922), the samurai chemist, and his work on adrenalin". Journal of Medical Biography. 11 (2): 95–102. doi: 10.1177/096777200301100211. PMID 12717538. S2CID 32540165.
- ^ a b Bennett MR (June 1999). "One hundred years of adrenaline: the discovery of autoreceptors". Clinical Autonomic Research. 9 (3): 145–59. doi: 10.1007/BF02281628. PMID 10454061. S2CID 20999106.
-
^ Takamine J (1901).
The isolation of the active principle of the suprarenal gland. Great Britain: Cambridge University Press. pp. xxix–xxx.
{{ cite book}}:|work=ignored ( help) - ^ a b "European Medicines Agency". Eurneffy EPAR. 27 June 2024. Retrieved 29 June 2024. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "First nasal adrenaline spray for emergency treatment against allergic reactions". European Medicines Agency (Press release). 28 June 2024. Retrieved 29 June 2024.
- ^ "Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 24-27 June 2024". European Medicines Agency. 28 June 2024. Archived from the original on 12 July 2024. Retrieved 12 July 2024.
- ^ "Background". Armstrong Pharmaceuticals. Archived from the original on 13 September 2013. Retrieved 12 December 2016.
- ^ "Statement from FDA Commissioner Scott Gottlieb, M.D., and Janet Woodcock, M.D., director of the Center for Drug Evaluation and Research, on approval of OTC Primatene Mist to treat mild asthma". FDA archive. 24 March 2020.
-
^
"Frequent Asked Questions". Armstrong Pharmaceuticals. Archived from
the original on 25 September 2011. Retrieved 22 September 2011.
Primatene® Mist was launched in 1963. The Primatene® Mist brand has built a long-time heritage for over-the-counter relief of bronchial asthma.
- ^ a b Wiebe K, Rowe BH (July 2007). "Nebulized racemic epinephrine used in the treatment of severe asthmatic exacerbation: a case report and literature review". Canadian Journal of Emergency Medicine. 9 (4): 304–8. doi: 10.1017/s1481803500015220. PMID 17626698.
- ^ Davies MW, Davis PG (2002). "Nebulized racemic epinephrine for extubation of newborn infants". The Cochrane Database of Systematic Reviews (1): CD000506. doi: 10.1002/14651858.CD000506. PMC 7038644. PMID 11869578.