|
| This It is of interest to the following WikiProjects: | ||||||||||
| |||||||||||
| The contents of the AXE method page were merged into VSEPR theory. For the contribution history and old versions of the redirected page, please see its history; for the discussion at that location, see its talk page. |
| The contents of the Kepert model page were merged into VSEPR theory on 5 September 2020. For the contribution history and old versions of the redirected page, please see its history; for the discussion at that location, see its talk page. |
| The contents of the Steric number page were merged into VSEPR theory on 6 September 2020. For the contribution history and old versions of the redirected page, please see its history; for the discussion at that location, see its talk page. |
I'm reading through Zumdahl's 8th edition of Chemistry for fun. Chapter 8 covers VSEPR theory and Lewis structures, but after linear and trigonal planar, I have difficulty deriving the ideal bond angles (not bond angles for specific molecules like methane, etc.). Are there any resources that explain it well? Could it be added here somewhere? Just a person with an electrical engineering background trying to pick up basic chemistry and having difficulty with the harder stuff... — Preceding unsigned comment added by 68.126.181.30 ( talk) 19:56, 17 August 2011 (UTC)
- The hardest of the important ones is the ideal tetrahedron such as methane. I have now added a link to a proof for this case in the Examples section. Dirac66 ( talk) 00:37, 8 May 2012 (UTC)
I removed the following text because I didn't know where it would fit better in the article. Please add it back if you figure out.
- If the connectivity, bond lengths, bond angles and torsional angles are found, a molecules exact geometry is known. For any non-linear molecule with N atoms, 3N - 6 internal coordinates need to be specified in order to know the exact geometry of the molecule.
-- Rune Welsh ταλκ 21:40, July 28, 2005 (UTC)
Is this correct? I always thought that VSEPR was part of Valence Bond Theory. I thought sigma and pi bonding were part of Molecular Orbital Theory.
"VSEPR theory is usually compared and contrasted with valence bond theory, which addresses molecular shape through orbitals that are energetically accessible for bonding. Valence bond theory concerns itself with the formation of sigma and pi bonds."
Captain Video 20:52, 9 August 2006 (UTC)Captain Video
VSEPR theory is not part of Valence Bond theory. See what the original authors od VSEPR say. It does however suggest similarities to some people. Sigma and pi bonding are part of both valence bond and molecular orbital approaches. I would keep the first sentence and delete the second. -- Bduke 23:39, 9 August 2006 (UTC)
"In fact, a steric number of seven is possible, but it occurs in uncommon compounds such as xenon hexafluoride. The base geometry for this is pentagonal bipyramidal. The trend for this configuration is the same as for the octahedral configuration: the first nonbonding electron domain would be in the axial position, making the actual molecular geometry pentagonal pyramidal."
This text is inaccurate - xenon hexaflouride is in fact a semi-octahedral geometry with a lone pair on it. It is indeed not pentagonal bipyramidal at all. —Preceding unsigned comment added by 143.167.238.53 ( talk) 17:13, 8 March 2008 (UTC)
XeF6 was indeed an incorrect example, but IF7 (with 0 lone pairs) is a valid example of pentagonal bipyramidal geometry. There are also several examples of anions with a central transition metal which have steric numbers 7, 8 and higher which can be included in the article. Dirac66 ( talk) 18:52, 8 March 2008 (UTC)
I have modified the paragraph to use IF7 as the example after mistakenly reverting the edit by the anon. -- Bduke ( talk) 00:59, 9 March 2008 (UTC)
I removed the following as unsourced, and lacking in good grammar. I'm no expert in VSEPR so I can't make a decision as to whether it is acceptable/correct or not. Anyone with more experience is invited to correct and reinsert material where appropriate. Hyenaste (tell) 22:07, 16 August 2006 (UTC)
- It must be emphasised that the VESPR is not really a theory, in spite of its often being referred to in this manner. It is a convenient method to get a rough idea about what the probable shape of a molecule might be. The actual shape that real molecules achieve is the result of quantum mechanics and the geometry of orbitals. One aspect of it that is particularly bad from a theoretical standpoint is the idea that "non bonding electrons repel more strongly than bonded ones." The real reason that stibine (SbH3) exhibits 90 degree bond angles is because the antimony (Sb) atom uses its p orbitals for forming the Sb-H bonds, and these are mutually perpendicular. Ammonia exhibits a 107 degree bond angle because the energy difference between 2s and 2p orbitals are small so hybridised orbitals can form. For s and p hybridised bonds the bond angle is given by acos(-s/p) where s is the "s" fraction of the orbital and p is the "p" component.
- The removed text actually is completely true. VSEPR is a sterics model that lacks nearly any serious theoretical background. It has been a colorful and easy-to-understand concept for freshmen students, but it had to be removed from the minds of advanced students in order to understand quantum mechanics-based bonding theory. The "rabbit ears" shape of lone pairs is a die-hard result of the widespread teaching of VSEPR theory.
- See [1] This source could also be cited as a source for the removed text. 134.76.64.163 ( talk) 08:01, 15 June 2012 (UTC)
I've given the article a quick assessment after adding the chemistry template. I only gave it a start class rating since it's quite short and lacks structure (sections). A little on the history of the theory would help with both these issues, for example. It's quite a good start class article all the same, maybe even a low B-class. Richard001 04:21, 16 January 2007 (UTC)
How is this possible?
There's quite a lot missing from the article; character of compounds and likely axial and equatorial postions of bonding pairs, multiple bonds, etc. Anyone up for a huge drive on this article, because it's pretty important in bonding theory
![]() ♥♥ ΜÏΠЄSΓRΘΠ€ ♥♥
slurp me!
12:06, 5 April 2007 (UTC)
♥♥ ΜÏΠЄSΓRΘΠ€ ♥♥
slurp me!
12:06, 5 April 2007 (UTC)
By the way, you can take a glance at the version of this article in Russian. There is explained the role of multiple bonds and some other features; unfortunately i could only barely help you because my English is definitely not suitable for writing article sections in English (i looked up some word in dictionary even to write this comment).--
159.148.226.100 (
talk)
17:49, 24 September 2009 (UTC)
- Thanks for the suggestion; I'll take a look and see if I can import some of that material. Спасибо!— Tetracube ( talk) 18:16, 24 September 2009 (UTC)
Sorry, i had clicked the wrong thing and mis-typed. I apologise :-)
![]() ♥♥ ΜÏΠЄSΓRΘΠ€ ♥♥
slurp me!
15:18, 8 April 2007 (UTC)
♥♥ ΜÏΠЄSΓRΘΠ€ ♥♥
slurp me!
15:18, 8 April 2007 (UTC)
I added the following text
"i) Pairs of electrons in the valence shell of a central atom repel each other. ii) These pairs of electrons tend to occupy positions in space that minimize repulsions and maximise the distance of separation between them. iii) The valence shell is taken as a sphere with electron pairs localising on the spherical surface at maximum distance from one another. iv) A multiple bond is treated as if it is a single electron pair and the two or three electron pairs of a multiple bond are treated as a single super pair. v) Where two or more resonance structures can depict a molecule the VSEPR model is applicable to any such structure."
thoug i am not an expert at this theory i am a student of it. if this is found to be wrong please forgive me and without hesitation delete this section hat i created.
I am not good at organising. so please any of you experts please organise it. thank you.-- Bandaruvamsi1991 15:41, 28 September 2007 (UTC)
As has been suggested by User:Bduke and others, I have merged the two articles. It will now redirect from the old article to here. Jokermole ( talk) 05:24, 21 January 2008 (UTC)
- A very good idea as AXE is just a notation for applying the VSEPR theory. The only problem was that you kept two identical copies of the table which appeared in both old articles, so I have deleted one copy of the table.
Dirac66 (
talk)
14:16, 21 January 2008 (UTC)
- Thanks! Happy Editing! Jokermole ( talk) 01:56, 22 January 2008 (UTC)
I know that it is hard to take seriously the anonymous numbered editor who earlier today first inserted HAHA and then two minutes later inserted "ALSO PRONOUNCED AS VESPER!!!!" all in capitals with 4 exclamation marks into the first line of the article. However I also know many chemists who do pronounce it informally as "vesper", and on picking up the first general chemistry book which comes to hand (Petrucci, Harwood and Herring "General Chemistry: Principles and Modern Applications", Prentice-Hall 8th edn 2002, p.410) I find that the theory is introduced as "the valence-shell electron-pair repulsion theory (written VSEPR and pronounced 'vesper')".
So perhaps it would be a good idea to let readers know that yes, this theory is the same "vesper" they have heard their profs mention, although without the capitals and the exclamation marks and perhaps not on the first line. I suggest modifying the second sentence, which also mentions an alternate name, to read "The theory is also called the Gillespie-Nyholm theory after the two main developers, and VSEPR is sometimes pronounced as "vesper" which is easier to say." Other opinions please. Dirac66 ( talk) 16:44, 19 March 2008 (UTC)
No objection after 1 week so I have done it. And I have also added an entry to the page Vespers (disambiguation), where some non-chemist who hears (rather than reads) the term might look. Dirac66 ( talk) 01:41, 26 March 2008 (UTC)
As of 9/29/2013 the lede claims that "technically" it is pronounced ...oh, wait; it doesn't say how it is pronounced, just that it is not "vesper". This is ridiculous and I am removing the offending text. There is NO "technically correct" pronunciation. I have been a chemist for more years than most here have been alive, and the only pronunciation I've ever heard is ves-per. It is just absurd and shows a real lack of thought that someone thinks VSE can not 'correctly' be pronounced 'ves', or that PR can't be pronounced 'per'. (One could argue that VSE could be 'vis' also). As the above shows, Petrucci, et al agree with me. Citing alternative authorities alternate pronunciation is fine if you are going to explain it. Citing it in a loose end is useless verbiage. You have a choice to either provide alternate pronunciations, or leave it alone, imho. Choosing between authorities is not the provenance of Wikipedia editors, especially for something this trivial. Note that I am not claiming that "vesper" is THE correct pronunciation, just that it IS A correct pronunciation. Ket-sup, ketch-up. 72.172.11.222 ( talk) 15:56, 29 September 2013 (UTC)
- "The lede claims that "technically" it is pronounced ...oh, wait; it doesn't say how it is pronounced, just that it is not "vesper"." This is incorrect, it DID say how it was pronounced - the phonetic pronunciation (which in this case is "vuh-seh-per", not "ves-per"). Unlike ket-sup/ket-chup, VSEPR is an acronym, meaning that it follows strictly phonetic pronunciation, which in this case is "vuh-seh-per". VSE can't correctly be pronounced "ves" - its correct pronunciation is "vuh-seh". PR's correct pronunciation is always "per", and no-one's arguing that it's anything different, so the correct pronunciation or "VSEPR" must be "vuh-seh-per".
Whoop whoop pull up
Bitching Betty |
Averted crashes
19:27, 29 September 2013 (UTC)
- I have never heard anyone say vuh-seh-per. The most frequent spoken usage is to spell it out (vee-ess-ee-pee-are), but ves-per is also common.
Dirac66 (
talk)
20:53, 29 September 2013 (UTC)
- I think "vesper" is most common in the US. Here in Australia I mostly hear it called "vee-ess-ee-pee-are" and I do not think I have ever heard it called "vuh-seh-per". -- Bduke (Discussion) 21:03, 29 September 2013 (UTC)
- I have never heard anyone say vuh-seh-per. The most frequent spoken usage is to spell it out (vee-ess-ee-pee-are), but ves-per is also common.
Dirac66 (
talk)
20:53, 29 September 2013 (UTC)
Assuming there were such a compound as XeF8, what would be its molecular geometry? An arrangement like the corners of a cube seems likely. Are there, for instance, any metal complexes with a coordination number of 8 (or possibly higher)? 212.137.63.86 ( talk) 15:22, 30 July 2008 (UTC)
- There are metal complexes with larger coordination numbers, but VSEPR fails for most metal complexes anyway. -- Itub ( talk) 05:28, 31 July 2008 (UTC)
There is no neutral XeF8, but the ion XeF82- exists as Cs and Rb salts. The geometry is square antiprismatic, that is one square of 4F atoms above the Xe and a second square of 4F atoms below but rotated 45o relative to the first. Reference C.E. Housecroft and A.G. Sharpe, "Inorganic Chemistry", 2nd edn (2005), p.498. The ion in principle has nine valence electron pairs, but probably the lone pair on Xe is stereochemically inactive. Dirac66 ( talk) 17:42, 31 July 2008 (UTC)
- I also found the following reference that discusses the geometry of complexes with high coordination numbers: Egon Wiberg, Nils Wiberg, Arnold Frederick Holleman, Inorganic Chemistry, Academic Press, 2001, ISBN 0123526515, 9780123526519. On page 1165, 3 possible geometries for 8-coordinate complexes are discussed: cubic, square antiprismic, and dodecahedral (note that this does not refer to the regular dodecahedron, but to a non-regular triangular-faced polyhedron, which can be thought of as the superposition of two tetrahedra, one stretched, and one flattened). The most common configuration for 8-coordinates is square antiprismic. The dodecahedral arrangement is less preferred, and the cubic arrangement is the least stable, and is adopted only when the stability of the resulting crystal lattice offsets the repulsion between the eclipsed ligands. Examples of the square antiprismic configuration include Mo(CN)84− and ZrF84−. In general, 8-coordinate complexes are favored with large metal cations with charges 3+ to 5+ from the 2nd or 3rd transition metal series.
- Complexes with 9 ligands adopt a capped square antiprism configuration (square antiprism with an additional vertex above one of the square faces, the result of course being distorted so that the capped square face is slightly larger than the uncapped square face, to compensate for increased repulsion between ligands), or the tricapped
triangular prism configuration (triangular prism with 3 extra vertices above each of its square faces). An example of a capped square antiprism complex is the two-centered, bridged complex [LaCl(H
2O)
7
24+. The tricapped triangular prism is more common, although still quite rare compared to 8-coordinate complexes. Examples include hydrates of trivalent lanthanides, some rhenium and technetium hydrides, and KPrF
4, whose crystal structure consists of PrF
9 (tricapped triangular prism) and KF
7 (monocapped triangular prism) units (Werner, F., Weil, M. & Kubel, F. (2003). Acta Cryst. E59, 147-149).
- Complexes with 10-coordination adopt a double-capped square antiprism configuration (square antiprism with both square faces capped); with 11-coordination, an octadecahedron (an 18-sided polyhedron); and with 12-coordination, an icosahedron. None of these occur with monodentate ligands, and complexes with multi-dentate ligands having these geometries can often be re-analysed as lower-coordination complexes where some multidentate ligands are treated as a unit.— Tetracube ( talk) 18:04, 7 May 2009 (UTC)
- What is this octadecahedron? Can you describe its geometry? Double sharp ( talk) 12:11, 3 May 2012 (UTC)
- Check the references I gave, that's where I got the description from.— Tetracube ( talk) 14:03, 7 May 2012 (UTC)
- Is this dodecahedron the snub disphenoid, by the way? The description seems to fit: the snub disphenoid has only triangular faces and has 8 vertices. Double sharp ( talk) 15:12, 28 April 2014 (UTC)
I had thought the term "valent electron" had been outdated and replaced by the much more accessable term "outer electron". I also beleived the theory was known as Electron Pair Repulsion Theory or Sidgwick-Powell Theory. -- 90.192.91.83 ( talk) 18:42, 20 November 2008 (UTC)
- Do you have a reference for your reminiscences? I still see new books and articles talking about VSEPR and valence shells. See for example these
433 Google Scholar hits for "valence shell" in 2008 alone (there are even more for "valence electron"). --
Itub (
talk)
20:14, 20 November 2008 (UTC)
- I am not sure about "outer electron" v "valence electron" but it, or something very much like it, was certainly called Electron Pair Repulsion Theory or Sidgwick-Powell Theory or the Sidgwick-Powell rules. VSEPR was an improvement of the Sidgwick-Powell theory made by Gillespie and Nyholm. Googling Sidgwick-Powell Theory gives lots of results, but many are teaching materials and I could not find a good definitive one. Incidentally Powell at the time the Sidgwick-Powell rules were put forward was an undergraduate at Oxford. He went on to be a crystallographer at Oxford. I attended his lectures 50 years ago. -- Bduke (Discussion) 21:21, 20 November 2008 (UTC)
- From W.L.Jolly, Modern Inorganic Chemistry (McGraw-Hill 1984), p.77
- "The essentials of this valuable concept were outlined by Sidgwick and Powell in 1940; the method has been refined and popularized by Gillespie and Nyholm." The references cited are :N.V.Sidgwick and H.M.Powell, Proc.Roy.Soc.A 176, 153 (1940); R.J.Gillespie and R.S.Nyholm, Quart.Rev. 11, 339 (1957); R.J.Gillespie, J.Chem.Educ. 47, 18(1970)
Dirac66 (
talk)
23:32, 20 November 2008 (UTC)
- Excellent. The Chemical Society Review, Vol 21, 59, (1992) by Gillespie says the name was proposed in 1963 by him in J.Chem.Educ. 40, 295, (1963), so there was an earlier name. Sorry, too flat out to check this out.
- Or there was previously no name. I have now looked at the Sidgwick-Powell article, which was actually a Bakerian lecture. The lecture is called "Stereochemical types and valency groups", and is not really presented as a theory with a name. Rather the authors just note that the "stereochemical type" seems to be correlated with the number of valence electrons both shared and unshared, and present the examples known at the time.
- Essentially they present the ideal examples, considering shared and unshared electron pairs as equivalent. For example they note that "triangular" OH2 can be "derived from a tetrahedron", but do not mention that the bond angle is less than the tetrahedral angle, which was later explained by Gillespie and Nyholm. Similarly they suggest that the large dipole moment of TeCl4 indicates a trigonal bipyramid with one position missing, "probably equatorial", but do not explain why the lone pair should be equatorial, which was again left for Gillespie and Nyholm.
- Given all this, perhaps the article should mention Sidgwick and Powell. Perhaps to say that they pointed out that molecular geometry depends on number of shared and unshared valence electrons [they didn't mention pairs], and that Gillespie and Nyholm used this concept to build a more detailed theory capable of choosing between various alternative geometries. Dirac66 ( talk) 03:23, 21 November 2008 (UTC)
Done. Dirac66 ( talk) 04:04, 28 November 2008 (UTC)
I think that this table should be ordered by increasing the increasing steric number, as opposed to its current arrangement which isn't as intuitive if you are trying to learn it. -- Rolen05 ( talk) 20:28, 2 December 2008 (UTC)
I agree for consistency with the first table, as well as with most textbooks and instructors. Dirac66 ( talk) 02:42, 3 December 2008 (UTC)
The anion in tetramethylammonium pentafluoroxenate is an example of this geometry. I would add it but it needs a standard image- and the table sounds as if it may be reorganised.-- Axiosaurus ( talk) 14:49, 5 December 2008 (UTC)
How are the "standard" images generated? Are they generated by
POVray? (If so, do we have the .pov files?) I'd like to add images for steric numbers 7, 8, and 9. Steric number 8 is attested in species like ZrF
84−, and steric number 9 is attested in ReH
92− and M(H
2O)
93+ where M is a lanthanide.—
Tetracube (
talk)
20:18, 7 May 2009 (UTC)
Good question. However the source code for the tables shows that each image is entered as a separate line and entered as a png file. So I think you could just draw your own images for added geometries using POVray and insert them as png files at the bottom of the tables. The existing images may or may not have been drawn using another program but what difference does that make? Dirac66 ( talk) 20:53, 7 May 2009 (UTC)
- I just thought it would be better to use the same POV models for the balls and sticks and colors so that the result has a uniform style. Or perhaps I should just redo all the images myself and post the POV files somewhere so that in the future people can just reuse the same style (or make it even better).— Tetracube ( talk) 21:52, 7 May 2009 (UTC)
- I don't know how different your style would be from the image of the existing style. Why not try pre-visualizing (without saving the edit) the addition of images for one geometry to see if you are satisfied with the similarity to the existing images? If so this will save you the work of redoing all the images again. If not then redoing them is a good suggestion.
- In any case I do think that more images for 7,8 and 9 are needed. Dirac66 ( talk) 01:01, 8 May 2009 (UTC)
- Also you can add AX5E2 as pentagonal planar, as suggested by Axiosaurus in the preceding section of this discussion. The two known examples from Housecroft and Sharpe (Inorganic Chemistry, 2d edn, Pearson 2005) are [IF52- (p.481) and [XeF5- (p.498). Dirac66 ( talk) 12:58, 8 May 2009 (UTC) Modified Dirac66 ( talk) 13:04, 8 May 2009 (UTC)
i changed the pictures on the trigonal planar and bent molecules grouping three. they were mismatched. i do not however know how to change the pictures so they now match bonding atoms and electron pairs —Preceding unsigned comment added by 136.165.12.53 ( talk) 03:20, 12 January 2010 (UTC)
- (I have moved this new section to the end.) In fact the table was correct so I have reverted your edit. However I have also added two sentences to explain that words such as "bent" refer only to the atomic positions (AX2) and not to the electron arrangement (AX2E). So in naming the pictures, the lone pairs are ignored. The second table is actually clearer as it shows both molecular geometry and electron arrangement. Dirac66 ( talk) 22:54, 12 January 2010 (UTC)
I've added a new See also, I dont know how to create an article but if someone know to do it, please, I think it is interesting:
Foundation We begin by assuming a Lewis structure model for chemical bonding based on valence shell electron pair sharing and the octet rule. We thus assume the nuclear structure of the atom, and we further assume the existence of a valence shell of electrons in each atom which dominates the chemical behavior of that atom. A covalent chemical bond is formed when the two bonded atoms share a pair of valence shell electrons between them. In general, atoms of Groups IV through VII bond so as to complete an octet of valence shell electrons. A number of atoms, including C, N, O, P, and S, can form double or triple bonds as needed to complete an octet. We know that double bonds are generally stronger and have shorter lengths than single bonds, and triple bonds are stronger and shorter than double bonds.
Goals We should expect that the properties of molecules, and correspondingly the substances which they comprise, should depend on the details of the structure and bonding in these molecules. The relationship between bonding, structure, and properties is comparatively simple in diatomic molecules, which contain two atoms only, e.g. HCl or O2. A polyatomic molecule contains more than two atoms. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. In molecules with more than three atoms, there are many more possible geometries. What geometries are actually observed? What determines which geometry will be observed in a particular molecule? We seek a model which allows us to understand the observed geometries of molecules and thus to predict these geometries.
Once we have developed an understanding of the relationship between molecular structure and chemical bonding, we can attempt an understanding of the relationship of he structure and bonding in a polyatomic molecule to the physical and chemical properties we observe for those molecules.
Observation 1: Geometries of molecules The geometry of a molecule includes a description of the arrangements of the atoms in the molecule. At a simple level, the molecular structure tells us which atoms are bonded to which. At a more detailed level, the geometry includes the lengths of all of these bonds, that is, the distances between the atoms which are bonded together, and the angles between pairs of bonds. For example, we find that in water, H2O, the two hydrogens are bonded to the oxygen and each O-H bond length is 95.72pm (where 1pm=10-12m). Furthermore, H2O is a bent molecule, with the H-O-H angle equal to 104.5°. (The measurement of these geometric properties is difficult, involving the measurement of the frequencies at which the molecule rotates in the gas phase. In molecules in crystalline form, the geometry of the molecule is revealed by irradiating the crystal with x-rays and analyzing the patterns formed as the x-rays diffract off of the crystal.)
Not all triatomic molecules are bent, however. As a common example, CO2 is a linear molecule. Larger polyatomics can have a variety of shapes, as illustrated in Figure 1. Ammonia, NH3, is a pyramid-shaped molecule, with the hydrogens in an equilateral triangle, the nitrogen above the plane of this triangle, and a H-N-H angle equal to 107°. The geometry of CH4 is that of a tetrahedron, with all H-C-H angles equal to 109.5°. (See also Figure 2(a).) Ethane, C2H6, has a geometry related to that of methane. The two carbons are bonded together, and each is bonded to three hydrogens. Each H-C-H angle is 109.5° and each H-C-C angle is 109.5°. By contrast, in ethene, C2H4, each H-C-H bond angle is 116.6° and each H-C-C bond angle is 121.7°. All six atoms of ethene lie in the same plane. Thus, ethene and ethane have very different geometries, despite the similarities in their molecular formulae.
Figure 1 Molecular Structures
We begin our analysis of these geometries by noting that, in the molecules listed above which do not contain double or triple bonds (H2O, NH3, CH4and C2H6), the bond angles are very similar, each equal to or very close to the tetrahedral angle 109.5°. To account for the observed angle, we begin with our valence shell electron pair sharing model, and we note that, in the Lewis structures of these molecules, the central atom in each bond angle of these molecules contains four pairs of valence shell electrons. For methane and ethane, these four electron pairs are all shared with adjacent bonded atoms, whereas in
ammonia or water, one or two (respectively) of the electron pairs are not shared with any other atom. These unshared electron pairs are called lone pairs . Notice that, in the two molecules with no lone pairs, all bond angles are exactly equal to the tetrahedral angle, whereas the bond angles are only close in the molecules with lone pairs
One way to understand this result is based on the mutual repulsion of the negative charges on the valence shell electrons. Although the two electrons in each bonding pair must remain relatively close together in order to form the bond, different pairs of electrons should arrange themselves in such a way that the distances between the pairs are as large as possible. Focusing for the moment on methane, the four pairs of electrons must be equivalent to one another, since the four C-H bonds are equivalent, so we can assume that the electron pairs are all the same distance from the central carbon atom. How can we position four electron pairs at a fixed distance from the central atom but as far apart from one another as possible? A little reflection reveals that this question is equivalent to asking how to place four points on the surface of a sphere spread out from each other as far apart as possible. A bit of experimentation reveals that these four points must sit at the corners of a tetrahedron, an equilateral triangular pyramid, as may be seen in Figure 2(b). If the carbon atom is at the center of this tetrahedron and the four electron pairs at placed at the corners, then the hydrogen atoms also form a tetrahedron about the carbon. This is, as illustrated in Figure 2(a), the correct geometry of a methane molecule. The angle formed by any two corners of a tetrahedron and the central atom is 109.5°, exactly in agreement with the observed angle in methane. This model also works well in predicting the bond angles in ethane.
Figure 2 Tetrahedral Structure of Methane (a) The dotted lines illustrate that the hydrogens form a tetrahedron about the carbon atom.
(b) The same tetrahedron is formed by placing four points on a sphere as far apart from one another as possible.
We conclude that molecular geometry is determined by minimizing the mutual repulsion of the valence shell electron pairs. As such, this model of molecular geometry is often referred to as the valence shell electron pair repulsion (VSEPR) theory . For reasons that will become clear, extension of this model implies that a better name is the Electron Domain (ED) Theory .
This model also accounts, at least approximately, for the bond angles of H2O and NH3. These molecules are clearly not tetrahedral, like CH4, since neither contains the requisite five atoms to form the tetrahedron. However, each molecule does contain a central atom surrounded by four pairs of valence shell electrons. We expect from our Electron Domain model that those four pairs should be arrayed in a tetrahedron, without regard to whether they are bonding or lone-pair electrons. Then attaching the hydrogens (two for oxygen, three for nitrogen) produces a prediction of bond angles of 109.5°, very close indeed to the observed angles of 104.5° in H2O and 107° in NH3.
Note, however, that we do not describe the geometries of H2O and NH3 as "tetrahedral," since the atoms of the molecules do not form tetrahedrons, even if the valence shell electron pairs do. (It is worth noting that these angles are not exactly equal to 109.5°, as in methane. These deviations will be discussed later.)
We have developed the Electron Domain model to this point only for geometries of molecules with four pairs of valence shell electrons. However, there are a great variety of molecules in which atoms from Period 3 and beyond can have more than an octet of valence electrons. We consider two such molecules illustrated in Figure 3.
Figure 3 More Molecular Structures
First, PCl5 is a stable gaseous compound in which the five chlorine atoms are each bonded to the phosphorous atom. Experiments reveal that the geometry of PCl5 is that of a trigonal bipyramid : three of the chlorine atoms form an equilateral triangle with the P atom in the center, and the other two chlorine atoms are on top of and below the P atom. Thus there must be 10 valence shell electrons around the phosphorous atom. Hence, phosphorous exhibits what is called an expanded valence in PCl5. Applying our Electron Domain model, we expect the five valence shell electron pairs to spread out optimally to minimize their repulsions. The required geometry can again be found by trying to place five points on the surface of a sphere with maximum distances amongst these points. A little experimentation reveals that this can be achieved by placing the five points to form a trigonal bipyramid. Hence, Electron Domain theory accounts for the geometry of PCl5.
Second, SF6 is a fairly unreactive gaseous compound in which all six fluorine atoms are bonded to the central sulfur atom. Again, it is clear that the octet rule is violated by the sulfur atom, which must therefore have an expanded valence. The observed geometry of SF6, as shown in Figure 3, is highly symmetric: all bond lengths are identical and all bond angles are 90°. The F atoms form an octahedron about the central S atom: four of the F atoms form a square with the S atom at the center, and the other two F atoms are above and below the S atom. To apply our Electron Domain model to understand this geometry, we must place six points, representing the six electron pairs about the central S atom, on the surface of a sphere with maximum distances between the points. The requisite geometry is found, in fact, to be that of an octahedron, in agreement with the observed geometry.
As an example of a molecule with an atom with less than an octet of valence shell electrons, we consider boron trichloride, BCl3. The geometry of BCl3 is also given in Figure 3: it is trigonal planar , with all four atoms lying in the same plane, and all Cl-B-Cl bond angles equal to 120°. The three Cl atoms form an equilateral triangle. The Boron atom has only three pairs of valence shell electrons in BCl3. In applying Electron Domain theory to understand this geometry, we must place three points on the surface of a sphere with maximum distance between points. We find that the three points form an equilateral triangle in a plane with the center of the sphere, so Electron Domain is again in accord with the observed geometry.
We conclude from these predictions and observations that the Electron Domain model is a reasonably accurate way to understand molecular geometries, even in molecules which violate the octet rule.
Observation 2: Molecules with Double or Triple Bonds In each of the molecules considered up to this point, the electron pairs are either in single bonds or in lone pairs. In current form, the Electron Domain model does not account for the observed geometry of C2H4, in which each H-C-H bond angle is 116.6° and each H-C-C bond angle is 121.7° and all six atoms lie in the same plane. Each carbon atom in this molecule is surrounded by four pairs of electrons, all of which are involved in bonding, i.e. there are no lone pairs. However, the arrangement of these electron pairs, and thus the bonded atoms, about each carbon is not even approximately tetrahedral. Rather, the H-C-H and H-C-C bond angles are much closer to 120°, the angle which would be expected if three electron pairs were separated in the optimal arrangement, as just discussed for BCl3.
This observed geometry can be understood by re-examining the Lewis structure. Recall that, although there are four electron pairs about each carbon atom, two of these pairs form a double bond between the carbon atoms. It is tempting to assume that these four electron pairs are forced apart to form a tetrahedron as in previous molecules. However, if this were this case, the two pairs involved in the double bond would be separated by an angle of 109.5° which would make it impossible for both pairs to be localized between the carbon atoms. To preserve the double bond, we must assume that the two electron pairs in the double bond remain in the same vicinity. Given this assumption, separating the three independent groups of electron pairs about a carbon atom produces an expectation that all three pairs should lie in the same plane as the carbon atom, separated by 120° angles. This agrees very closely with the observed bond angles. We conclude that the our model can be extended to understanding the geometries of molecules with double (or triple) bonds by treating the multiple bond as two electron pairs confined to a single domain. It is for this reason that we refer to the model as Electron Domain theory.
Applied in this form, Electron Domain theory can help us understand the linear geometry of CO2. Again, there are four electron pairs in the valence shell of the carbon atom, but these are grouped into only two domains of two electron pairs each, corresponding to the two C=O double bonds. Minimizing the repulsion between these two domains forces the oxygen atoms to directly opposite sides of the carbon, producing a linear molecule. Similar reasoning using Electron Domain theory as applied to triple bonds correctly predicts that acetylene, HCCH, is a linear molecule. If the electron pairs in the triple bond are treated as a single domain, then each carbon atom has only two domains each. Forcing these domains to opposite sides from one another accurately predicts 180° H-C-C bond angles.
Observation 3: Distortions from Expected Geometries It is interesting to note that some molecular geometries (CH4, CO2, HCCH) are exactly predicted by the Electron Domain model, whereas in other molecules, the model predictions are only approximately correct. For examples, the observed angles in ammonia and water each differ slightly from the tetrahedral angle. Here again, there are four pairs of valence shell electrons about the central atoms. As such, it is reasonable to conclude that the bond angles are determined by the mutual repulsion of these electron pairs, and are thus expected to be 109.5°, which is close but not exact.
One clue as to a possible reason for the discrepancy is that the bond angles in ammonia and water are both less than 109.5°. Another is that both ammonia and water molecules have lone pair electrons, whereas there are no lone pairs in a methane molecule, for which the Electron Domain prediction is exact. Moreover, the bond angle in water, with two lone pairs, is less than the bond angles in ammonia, with a single lone pair. We can straightforwardly conclude from these observations that the lone pairs of electrons must produce a greater repulsive effect than do the bonded pairs. Thus, in ammonia, the three bonded pairs of electrons are forced together slightly compared to those in methane, due to the greater repulsive effect of the lone pair. Likewise, in water, the two bonded pairs of electrons are even further forced together by the two lone pairs of electrons.
This model accounts for the comparative bond angles observed experimentally in these molecules. The valence shell electron pairs repel one another, establishing the geometry in which the energy of their interaction is minimized. Lone pair electrons apparently generate a greater repulsion, thus slightly reducing the angles between the bonded pairs of electrons. Although this model accounts for the observed geometries, why should lone pair electrons generate a greater repulsive effect? We must guess at a qualitative answer to this question, since we have no description at this point for where the valence shell electron pairs actually are or what it means to share an electron pair. We can assume, however, that a pair of electrons shared by two atoms must be located somewhere between the two nuclei, otherwise our concept of "sharing" is quite meaningless. Therefore, the powerful tendency of the two electrons in the pair to repel one another must be significantly offset by the localization of these electrons between the two nuclei which share them. By contrast, a lone pair of electrons need not be so localized, since there is no second nucleus to draw them into the same vicinity. Thus more free to move about the central atom, these lone pair electrons must have a more significant repulsive effect on the other pairs of electrons.
These ideas can be extended by more closely examining the geometry of ethene, C2H4 . Recall that each H-C-H bond angle is 116.6° and each H-C-C bond angle is 121.7°, whereas the Electron Domain theory prediction is for bond angles exactly equal to 120°. We can understand why the H-C-H bond angle is slightly less than 120° by assuming that the two pairs of electrons in the C=C double bond produce a greater repulsive effect than do either of the single pairs of electrons in the C-H single bonds. The result of this greater repulsion is a slight "pinching" of the H-C-H bond angle to less than 120°.
The concept that lone pair electrons produce a greater repulsive effect than do bonded pairs can be used to understand other interesting molecular geometries. Sulfur tetrafluoride, SF4, is a particularly interesting example, shown in Figure 4.
Figure 4 Molecular Structure of SF4
Note that two of the fluorines form close to a straight line with the central sulfur atom, but the other two are approximately perpendicular to the first two and at an angle of 101.5° to each other. Viewed sideways, this structure looks something like a seesaw.
To account for this structure, we first prepare a Lewis structure. We find that each fluorine atom is singly bonded to the sulfur atom, and that there is a lone pair of electrons on the sulfur. Thus, with five electron pairs around the central atom, we expect the electrons to arrange themselves in a trigonal bipyramid, similar to the arrangement in PCl5 in Figure 3. In this case, however, the fluorine atoms and the lone pair could be arranged in two different ways with two different resultant molecular structures. The lone pair can either go on the axis of the trigonal bipyramid (i.e. “above” the sulfur) or on the equator of the bipyramid (i.e. “beside” the sulfur).
The actual molecular structure in Figure 4 shows clearly that the lone pair goes on the equatorial position. This can be understood if we assume that the lone pair produces a greater repulsive effect than do the bonded pairs. With this assumption, we can deduce that the lone pair should be placed in the trigonal bipyramidal arrangement as far as possible from the bonded pairs. The equatorial position does a better job of this, since only two bonding pairs of electrons are at approximately 90° angles from the lone pair in this position. By contrast, a lone pair in the axial position is approximately 90° away from three bonding pairs. Therefore, our Electron Domain model assumptions are consistent with the observed geometry of SF4. Note that these assumptions also correctly predict the observed distortions away from the 180° and 120° angles which would be predicted by a trigonal bipyramidal arrangement of the five electron pairs.
[end of proposed text]
This looks very much as if it is copied directly from somewhere and would therefore be a copyright violation. In what sense is this topic notable enough for a separate article rather than being incorporated in one or more other article? -- Bduke (Discussion) 20:35, 1 February 2010 (UTC)
The picture for AX3E0 in the AXE Method section looks needlessly complicated to me. Couldn't it be exactly the same as the AX2E1 picture, but surrounded completely by Xes? 155.33.145.250 ( talk) 23:22, 17 September 2010 (UTC)
- Yes, that would be correct also, if someone wants to redraw the diagram.
The new section that was added by User:B.esselman has some serious issues. It is not the job of VSEPR to predict the correct hybridization. It has nothing to do with hybridization. Its job is to predict the correct shape. Whether the lone pairs are different or equivalent is a matter of choice. Density functional theory (DFT), ab initio, and semi-empirical calculations, as molecular orbital calculations, of course give one MO that is an out-of plane p-orbital and one that is essentially an in-plane lone pair. They give orbitals that reflect the symmetry of the molecule. They do not give two bonds either. However if one transforms the MOs to localised orbitals (a unitary transformation that leave the wave function unchanged, you get two equivalent bond orbitals and two equivalent lone pairs (each being the MO lone pair ± the p orbital). I have discussed this further at User talk:B.esselman. However, this is really beside the point as hybridisation has no role in VSEPR. The edit was to add the following below a section headed "Water" under the major "Exceptions" header:
"VSEPR fails to predict the correct hybridization of the oxygen in water molecules or other disubstituted oxygen compounds. VSEPR predicts a tetrahedral electron geometry for water with two coordinates occupied by lone pairs resulting in an sp3 hybridization. If VSEPR was correct it requires the oxygen atom to have two equal energy, sp3, lone pairs. Density functional theory (DFT), ab initio, and semi-empirical calculations all agree that the oxygen has two non-equivalent lone pairs. One of the lone pairs is in a purely p-type orbital with its electron density perpendicular to the bonding plane of the O-H bonds. The other lone pair is close to an sp2 hybrid, though this is an over-simplification of the orbital mixing. This lone pair is located in the plane of the O-H bonds. It is a major misconception in many introductory textbooks that oxygen is an sp3 hybrid in water and other analogous structures. [2] [3]"
I have removed it to get further input from other editors. -- Bduke (Discussion) 23:07, 24 April 2011 (UTC)
- I have looked at the sources given. Schreiner argues that MO calculations do a better job than simple steric pictures of explaining steric effects such as the rotational barrier in ethane and the anomeric effect. He does not mention VSEPR, hybridization or water. Clauss and Nelsen argue for the use of computational molecular modelling in the undergraduate organic curriculum rather than "looking at colorful pictures in the textbook", though admitting that the former are more difficult for students to understand. They point out that the simple MO picture (sp2-p) appears to contradict the simple VSEPR picture with two localized lone pairs, which confuses students. They also describe the molecular modelling picture as "consistent with experimental results", possibly referring to observation of four nonequivalent valence-shell MOs for water by ultraviolet photoelectron spectroscopy.
- As BDuke correctly points out, these arguments neglect the fact that the two descriptions of the molecular ground state are actually equivalent, since the many-electron wave function is invariant to a linear transformation of the occupied MOs. We therefore should not use language here which implies that one picture is correct and the other wrong, for example "VSEPR fails to predict" and "major misconception".
- However I think this article could provide some guidance to upper-year organic undergraduates who are wondering why the modelling picture apparently disagree with VSEPR, as noted by Clauss and Nelsen, and who do not understand the quantum-mechanical basis of their modelling. (An increasingly common situation unfortunately.) Perhaps a last section which explains that the two models provide different but equivalent descriptions of the ground state, and then comparing the advantages of each. VSEPR = simple description of ground state geometry, MO = more quantitative accuracy, and can describe excited and ionized states. Dirac66 ( talk) 01:58, 25 April 2011 (UTC)
OK, I have written a suggested new section for this article, to explain the relation between localized and delocalized (or canonical) MOs without saying that one is wrong. Comments?
The VSEPR theory places each pair of valence electrons in a bond or lone pair corresponding to a local region of the molecule. Molecular orbital theory yields a set of orbitals which have the symmetry of the molecule and which are often delocalized over several atoms. However these orbitals can be transformed into an equivalent set of localized molecular orbitals
In the water molecule for example, molecular orbital calculations yield two lone pairs, one an s-p hybrid in the plane of the molecule and one a pure p orbital perpendicular to this plane. These orbitals can be combined into two sp3 lone pairs which are equivalent to each other. Similarly there are two calculated bonding orbitals each extending over all three atoms, which can be combined into two localized orbitals, one for each bond.
The delocalized and localized orbitals provide completely equivalent descriptions of the ground state, since the total wavefunction for all electrons is a Slater determinant which is unchanged by the transformation of the orbitals.[Ref: Levine I.N., “Quantum Chemistry” (4th ed., Prentice-Hall 1991) sec.15.8] Dirac66 ( talk) 02:43, 4 May 2011 (UTC)
- Good wording in general. Maybe change "are often delocalized over several atoms" to "are delocalized over several atoms". I am a bit unhappy about "which correspond to the electron pairs of VSEPR theory". Gillespie is very careful to say that the electron pairs are not LMOs, VB pairs, Lewis structure pairs, or anything but what the VSEPR theory says they are. Maybe "which can be compared to the electron pairs of VSEPR theory". --
Bduke
(Discussion)
02:57, 4 May 2011 (UTC)
- I have now added this section to the article. Thanks for the corrections, which I have tried to incorporate. I didn't actually find the phrase "which correspond to the electron pairs of VSEPR theory", but I removed "correspond to" and added "compared to", both here and at the LMO page.
- Also Gillespie did eventually collaborate with his departmental colleague
Richard Bader to try to better relate VSEPR to QM. About 1995(?) I heard a presentation by Bader who I think identified lone pairs (and bond pairs too?) with maxima in the Laplacian of the charge density. Too technical for Wikipedia I think. (Even if I have it correct).
Dirac66 (
talk)
01:55, 5 May 2011 (UTC)
- That is correct and very interesting it is indeed and it should be mentioned in this article. However, it is a connection to the QM density not to the wavefunction, which any connection to MOs or VB orbitals would be. I have Bader's book and some of his articles and I will see if I can add something about it. It may be technical, but it should be mentioned. --
Bduke
(Discussion)
02:05, 5 May 2011 (UTC)
- OK, I don't have his book so I'll wait for you to do the reading. Eventually we could have a single section "Relation of VSEPR to quantum mechanics" with two subsections - one on LMOs and one on the QM density.
Dirac66 (
talk)
02:37, 5 May 2011 (UTC)
- OK, I will see what I can do. Do we actually have a source that specifically links LMOs with VSEPR? -- Bduke (Discussion) 05:20, 5 May 2011 (UTC)
- OK, I don't have his book so I'll wait for you to do the reading. Eventually we could have a single section "Relation of VSEPR to quantum mechanics" with two subsections - one on LMOs and one on the QM density.
Dirac66 (
talk)
02:37, 5 May 2011 (UTC)
- That is correct and very interesting it is indeed and it should be mentioned in this article. However, it is a connection to the QM density not to the wavefunction, which any connection to MOs or VB orbitals would be. I have Bader's book and some of his articles and I will see if I can add something about it. It may be technical, but it should be mentioned. --
Bduke
(Discussion)
02:05, 5 May 2011 (UTC)
Probably not a useful one since we have agreed not to identify VSEPR electron pairs with QM orbitals. But it is evident that VSEPR is a local theory in the vague sense that the pretty pictures place the electrons in specific regions. My concern is to answer the argument which you recently removed from the article, to the effect that the best model of reality is MO theory which is delocalized, therefore VSEPR as a local theory is wrong. The simple counter-argument is that MO theory can also be transformed to an equivalent localized form and so does not invalidate other local theories. Perhaps you can express this more clearly in the text than I have done. Dirac66 ( talk) 14:30, 5 May 2011 (UTC)
I doubt that there are only eight electron pairs around the xenon atom, based on its Lewis structure. How would it be square antiprismatic?-- Jasper Deng (talk) 01:53, 27 April 2012 (UTC)
- Good point. The linked article on
nitrosonium octafluoroxenate(VI) cites a 1971 article by Peterson et al. which I looked up. X-ray crystallography does give a (very slightly distorted) square antiprism, and the authors comment that "there is no clearly defined coordination position for the ninth pair" [which] "appears to offer support for electron repulsion models". That is, they realized in 1971 that this molecule is an exception to VSEPR. So in this article we probably should a more typical example in the tables, such as the Mo or the Zr complex mentioned in the examples.
Dirac66 (
talk)
03:18, 27 April 2012 (UTC)
- So, it's implied that the lone pair on Xe has no effect on the molecular geometry?-- Jasper Deng (talk) 03:23, 27 April 2012 (UTC)
- Oh, I see. It makes me wonder where that pair would go, and yep this does mean we have better examples to cite.--
Jasper Deng
(talk)
03:25, 27 April 2012 (UTC)
- OK, in the tables in the AXE method section I have replaced XeF82- by ZrF84-, which really has 8 pairs. As for the question of where the lone pair would go, I think this is an example of the inert pair effect which can include steric inactivity, especially for heavier atoms. This is probably a consequence of relativistic effects which lower the Xe 5s energy relative to Xe 5p, so where it goes is closer to the Xe nucleus where it does not affect the geometry of the bonding pairs. The octahedral geometry of XeF6 (7 pairs!) has a similar explanation, although in that case the octahedral and C3v geometries are very close in energy. Dirac66 ( talk) 03:02, 28 April 2012 (UTC)
Today we seem to have a disagreement as to where put XeF82- in the table of geometries and examples. Does it go on the line for steric number 8 or 9? As discussed above, the problem is that although simple electron counting (Lewis structure) gives 9 pairs (8 bonds + 1 lone pair), the experimental geometry is the square antiprism expected for 8 pairs. That is, the VSEPR theory gives the wrong answer for this molecule, as first noted in 1971 by Peterson et al. who determined the experimental structure.
So I think it is misleading to put this molecule on EITHER line. Placing it at n=8 is misleading because the VSEPR model is based on simple electron counting which gives n=9. And placing it at n=9 is misleading because it suggests that VSEPR predicts a square antiprism for n=9 and one lone pair, which is not true. (I think - if it does we need a reference).
I think the answer is to remove this molecule as an example in the tables, which should only use examples for which VSEPR predicts the correct geometry. Exceptions are dealt with in the next section where they are clearly labelled as such. We can add XeF82- as an exception after XeF6, which is octahedral for somewhat similar reasons. Dirac66 ( talk) 21:40, 6 May 2012 (UTC)
 Done
Double sharp (
talk)
03:38, 11 May 2012 (UTC)
Done
Double sharp (
talk)
03:38, 11 May 2012 (UTC)
- Thanks to Double sharp and I agree with your edits for XeF82-. However they have now been reverted by Whoop whoop pull up with the edit summaries "Rv unexplained removal of content" and "Rm nonsense". To Whoop whoop pull up: the removal was in fact explained in this section of the talk page, so please respond to the above comments and explain why you think that Double sharp's edit is nonsense. While I have myself on other occasions used similar edit summaries, I would not do so when the edits have been justified on the talk page.
Dirac66 (
talk)
19:28, 12 May 2012 (UTC)
- Here is the reason why. Consider a molecule with a capped square antiprismatic geometry. Now ignore the ligand capping the square antiprism. What do you get? A slightly distorted square antiprism. What is the geometry of XeF82-? A slightly distorted square antiprism—which is exactly the geometry predicted by VSEPR theory for an AX8E1 molecule!
Whoop whoop pull up
Bitching Betty |
Averted crashes
22:32, 12 May 2012 (UTC)
- Well, that was explained quite well in the edit summaries; ignoring the lone pair on the octafluoroxenate ion is a special case that confuses our readers.--
Jasper Deng
(talk)
22:37, 12 May 2012 (UTC)
- So who do you agree with?
Whoop whoop pull up
Bitching Betty |
Averted crashes
22:43, 12 May 2012 (UTC)
- Dirac66 - after all, I was the one who noted the confusion about this particular ion.--
Jasper Deng
(talk)
22:44, 12 May 2012 (UTC)
- So you're one of the ones being confused? Read my post above and see why I am right and he is wrong.
Whoop whoop pull up
Bitching Betty |
Averted crashes
22:49, 12 May 2012 (UTC)
- Most people cannot visualize the shape. Even if you are technically correct, the shapes aren't exactly the same, and therefore, this ion is a bad example of this geometry.--
Jasper Deng
(talk)
22:52, 12 May 2012 (UTC)
- Then what would be a good example of an AX8E1 molecule?
Whoop whoop pull up
Bitching Betty |
Averted crashes
23:58, 12 May 2012 (UTC)
- Perhaps ReH84-, if existing? Just any complex that has a steric # of 9.-- Jasper Deng (talk) 00:21, 13 May 2012 (UTC)
- Then what would be a good example of an AX8E1 molecule?
Whoop whoop pull up
Bitching Betty |
Averted crashes
23:58, 12 May 2012 (UTC)
- Most people cannot visualize the shape. Even if you are technically correct, the shapes aren't exactly the same, and therefore, this ion is a bad example of this geometry.--
Jasper Deng
(talk)
22:52, 12 May 2012 (UTC)
- So you're one of the ones being confused? Read my post above and see why I am right and he is wrong.
Whoop whoop pull up
Bitching Betty |
Averted crashes
22:49, 12 May 2012 (UTC)
- Dirac66 - after all, I was the one who noted the confusion about this particular ion.--
Jasper Deng
(talk)
22:44, 12 May 2012 (UTC)
- So who do you agree with?
Whoop whoop pull up
Bitching Betty |
Averted crashes
22:43, 12 May 2012 (UTC)
- Well, that was explained quite well in the edit summaries; ignoring the lone pair on the octafluoroxenate ion is a special case that confuses our readers.--
Jasper Deng
(talk)
22:37, 12 May 2012 (UTC)
- Here is the reason why. Consider a molecule with a capped square antiprismatic geometry. Now ignore the ligand capping the square antiprism. What do you get? A slightly distorted square antiprism. What is the geometry of XeF82-? A slightly distorted square antiprism—which is exactly the geometry predicted by VSEPR theory for an AX8E1 molecule!
Whoop whoop pull up
Bitching Betty |
Averted crashes
22:32, 12 May 2012 (UTC)
- Thanks to Double sharp and I agree with your edits for XeF82-. However they have now been reverted by Whoop whoop pull up with the edit summaries "Rv unexplained removal of content" and "Rm nonsense". To Whoop whoop pull up: the removal was in fact explained in this section of the talk page, so please respond to the above comments and explain why you think that Double sharp's edit is nonsense. While I have myself on other occasions used similar edit summaries, I would not do so when the edits have been justified on the talk page.
Dirac66 (
talk)
19:28, 12 May 2012 (UTC)
I think this is a good time to remember that Wikipedia is supposed to be based on sources. For the higher coordination numbers we should be especially careful to find references, since their geometries are not as familiar as those of ammonia, water, etc.
I have started by checking Housecroft and Sharpe which we have already cited as a source for AX6E1 exceptions such as XeF6. In the same paragraph (p.48) they say that IF8- and TeF82- are square antiprismatic (two more examples of AX8E0), and then add that XeF82- "also adopts this structure, indicating that the lone pair of electrons is stereochemically inactive." So it is an exception similar to XeF6 and should be in the Exceptions section, not in the table. So Double Sharp's edit should be restored, and now we have a source for the statement.
Whoop whoop pull up's argument for AX8E1 is based on a comparison with a capped square antiprismatic geometry AX9E0, using ReH92- as an example. However the article on Potassium nonahydridorhenate describes the geometry (in the text) as tricapped trigonal prismatic, with a reference to the book by Holleman and Wiberg. I added today Housecroft and Sharpe as a second source which agrees. The figure in that article was capped square antiprismatic (since 2008!), so today I changed the figure to agree with the text and the two sources. So in the tables of this article, AX9E0 should be given as tricapped trigonal prismatic. As for AX8E1, if there is no known (sourced) example other than XeF82- then it should just be deleted from the table.
Finally for n = 10, 11 and 12 I have not yet found any sources. If we have no sources, then WP:OR means that we cannot just insert the geometry that seems logical to one group of editors. The correct policy is to leave them out of the article unless someone finds evidence that their VSEPR geometry has been published. Dirac66 ( talk) 01:58, 13 May 2012 (UTC)
- XeF82- does not have a regular square antiprismatic geometry. It has a distorted square antiprismatic geometry. Which is what VSEPR theory predicts for an AX8E1 molecule. Whoop whoop pull up Bitching Betty | Averted crashes 13:41, 27 May 2012 (UTC)
- *turns pale with fury* Tricapped trigonal prismatic and capped square antiprismatic are equivalent! When are you going to realise that?!?!?!?!?!?!?!?!?! Whoop whoop pull up Bitching Betty | Averted crashes 11:10, 13 May 2012 (UTC)
- I added a source for n = 10, 11 and 12. (It doesn't agree with Whoop whoop pull up.)
Double sharp (
talk)
15:32, 26 May 2012 (UTC)
- ...and Whoop whoop pull up has reverted me, but (oddly) hasn't changed or removed my source, which supports the molecular geometries I gave. I've reverted him back. Double sharp ( talk) 06:30, 13 June 2012 (UTC)
Apparently, they're not equivalent. This review article states that tricapped trigonal prismatic and capped square antiprismatic are distinct geometries. Nonetheless, these geometries are similar. Real, distorted structures can often be described as a distortion of either ideal geometry equally well. But that is not the same thing as saying the two idealised geometries are equivalent. This other review states capped square antiprismatic geometry has C4v symmetry, whereas tricapped trigonal prismatic has D3h symmetry. -- Ben ( talk) 12:20, 13 May 2012 (UTC)
- Another nice molecule with steric number nine is
Ln(H2O)3+
9 ( Tetracube suggested this one some way up the talk page). For steric numbers 10 and above, see Holleman & Wiberg, p. 1165. Another interesting set of molecules is the closo-boranates [BnHn2− for 6 ≤ n ≤ 12, which form the deltahedra from the octahedron to the icosahedron. For n = 11, where an 18-faced deltahedron is necessary (despite the fact that the ideal octadecadeltahedron with equilateral triangular faces would not be convex as it would have coplanar faces), the solution adopted by the atoms is a kind of hybrid square-pentagonal antiprism, when one base is a tetragon and the other is a pentagon (Holleman & Wiberg, p. 960). (Obviously, the triangles cannot be equilateral without being coplanar.) - @Whoop whoop pull up: The capped square antiprismatic geometry is the
gyroelongated square pyramid, while the tricapped triangular prismatic geometry is the
triaugmented triangular prism. These are different polyhedra: one has 1 square face and 12 triangular faces, while the other has 14 triangular faces.
Double sharp (
talk)
15:13, 22 May 2012 (UTC)
- They may not be identical, but they are surely very similar.
Whoop whoop pull up
Bitching Betty |
Averted crashes
02:49, 24 May 2012 (UTC)
- They're identical or they're not.--
Jasper Deng
(talk)
02:51, 24 May 2012 (UTC)
- They are very similar.
Whoop whoop pull up
Bitching Betty |
Averted crashes
13:26, 27 May 2012 (UTC)
- Well, although they have different symmetry, I think (from the pictures below) that if you subdivide the square face of the gyroelongated square pyramid (left, capped square antiprismatic) by a diagonal, and deform the triangles till they are all equilateral again, you could get the triaugmented triangular prism (right, tricapped trigonal prismatic).
-

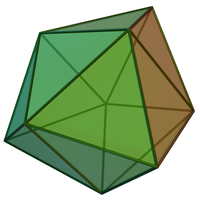
- Double sharp ( talk) 12:30, 5 June 2012 (UTC)
- They are very similar.
Whoop whoop pull up
Bitching Betty |
Averted crashes
13:26, 27 May 2012 (UTC)
- They're identical or they're not.--
Jasper Deng
(talk)
02:51, 24 May 2012 (UTC)
- They may not be identical, but they are surely very similar.
Whoop whoop pull up
Bitching Betty |
Averted crashes
02:49, 24 May 2012 (UTC)
They're not identical, they have different symmetry. Several journal articles I've read do say they're closely related. I assume this means it doesn't take much energy to change shape from one to the other. I am working on images for the article. If you want models and references for closo-borane anions, see this Jmol page I made a while ago. -- Ben ( talk) 07:43, 24 May 2012 (UTC)
- The octadecahedron indeed has 6 triangles meeting at some vertices, so the triangles are not equilateral. As I mentioned earlier, it may be described as a biaugmented square-pentagonal antiprism. Double sharp ( talk) 14:28, 24 May 2012 (UTC)
Two true capped square antiprismatic compounds are [ Pr(terpy)Cl3(H2O)5]·3H2O and [Th(Tropolon)4(H2O)]. Two geometries for steric number 10 are bicapped dodecahedral and bicapped square antiprismatic, and a geometry for steric number 14 (occurs in [An(BH4)4], An = Th, Pa, or U) is bicapped hexagonal antiprismatic (Holleman & Wiberg, p. 1721). Double sharp ( talk) 04:35, 23 November 2012 (UTC)
- I've added these (with the source) to the article. I replaced dodecahedral with the synonymous (but clearer) dodecadeltahedral, however, to make it clear that we are talking about the snub disphenoid and not the regular pentagonal dodecahedron or quasiregular rhombic dodecahedron. Double sharp ( talk) 15:18, 28 April 2014 (UTC)
Today again some of the blue (working) links in the tables have been replaced by red (broken) links. For example, square antiprism is blue and leads to a real article), but has been replaced by square antiprismatic molecular geometry which is red and leads to an error message. Well, an invitation to create your own article. This is not really helpful to the 99.9% of readers (there were 23K page views in the last 30 days) who have no intention of creating their own article on this subject, but may just want to know what a square antiprism looks like. The square antiprism article at least does show a picture and some discussion, even if it is not an article with molecular geometry in its title.
So I am going to revert the latest removal and restore the link to square antiprism, as well as the link to gyroelongated square pyramid inserted by Double Sharp earlier today. Also I am going to replace the red link to pentagonal planar molecular geometry by a simple link to Pentagon, again to illustrate the term as well as possible using an existing article. Please do not restore the red linked terms ... unless you are actually going to write the missing articles to which they point. Dirac66 ( talk) 02:53, 4 May 2012 (UTC)
 Done I've restored the blue links.
Double sharp (
talk)
03:00, 4 May 2012 (UTC)
Done I've restored the blue links.
Double sharp (
talk)
03:00, 4 May 2012 (UTC)
- The articles have been written and I have restored the links to them.
Whoop whoop pull up
Bitching Betty |
Averted crashes
12:53, 12 May 2012 (UTC)
- OK, now these links lead to useful information. Dirac66 ( talk) 19:10, 12 May 2012 (UTC)
- The articles have been written and I have restored the links to them.
Whoop whoop pull up
Bitching Betty |
Averted crashes
12:53, 12 May 2012 (UTC)
The most recent controversies are about the inclusion of diatomics in the article and the likely geometries for steric numbers 10, 11 and 12.
I agree with Whoop Whoop Pull Up that diatomic molecules do not belong in this article. The reason is that VSEPR theory is not required to find the geometry of a diatomic molecule, which must be linear because any two points define a line. This was obvious long before VSEPR and even before electrons were known to exist. So diatomics are not an example of the use of VSEPR theory, even if some general chemistry books include them in a table of molecular geometries.
For n = 10, 11 and 12 however, I cannot agree with Whoop Whoop Pull Up's repeated reversion of the edits by Double Sharp. Wikipedia is based on sources - see for example WP:Sources which says that any material challenged or likely to be challenged must be attributed to a reliable published source. The geometries given by each of these editors for n = 10-12 have been challenged by the other, but only Double Sharp has given a source. Whoop Whoop Pull Up has given no source or even real reasoning other than to say in his edit summaries that I am right and you are wrong (though not in those words).
Since we have a source for one set of geometries and not the other, we must use the sourced geometries. And even if Whoop Whoop Pull Up does find a future source for the other set of geometries, then we would have conflicting sources so we would mention both opinions.
Also in his latest edit, Whoop Whoop Pull Up has just deleted Double Sharp's source without explanation. Since the source is a standard textbook, it is not acceptable to just remove it because it does not support your point of view.
In view of the above, I will now restore Double Sharp's last version for n = 10-12 with the source, but not the diatomic molecules. Dirac66 ( talk) 01:02, 14 June 2012 (UTC)
Today's edits have raised the question of odd-electron species: NO2, ClO2 and UF5. From memory the rule is that the odd electron (or "half electron pair") counts as a full electron pair for determining the basic shape, but takes up less space for determining the bond angle - ex. bent 134° in NO2 vs. bent 120° (approx.) in NO2- and 117-118° in ClO2 vs. close to tetrahedral (109°) in ClO2-. And UF5 is square pyramidal like BrF5. However most books omit the rare odd-electron molecules from VSEPR discussions and I have not actually found a source in a 15-minute search.
If someone can find a source to confirm the rule, then I would suggest discussing the odd-electron examples in a separate section after Exceptions. Then we can leave the AXE table for the even-electron examples which are discussed in most books. The theory is after all called valence shell electron PAIR repulsion. Dirac66 ( talk) 00:43, 18 June 2012 (UTC)





A half electron pair acts mostly like a full electron pair, but can also allow the molecule to act more like one lacking the electron pair. For instance: NO2 is intermediate between NO2+ and NO2-; ClO2 is intermediate between ClO2+ and ClO2-. Whoop whoop pull up Bitching Betty | Averted crashes 01:26, 18 June 2012 (UTC)
- Yes, the intermediate character of the odd-electron species can be mentioned also. I think the subject of odd-electron species is complex enough for a separate section. Another reason is that it is not covered in most standard texts, and we still should look for a source, although many chemists just seem to know the rules for odd-electron species.
- Also CH3 which is now in the AXE table is a more subtle case. The unsubstituted CH3 is actually planar (see source listed at Methyl radical), but the energy cost of distortion to pyramidal is low and substituted methyls are mostly pyramidal as the above discussion would suggest. So CH3 itself is another exception to be mentioned in the separate section. Dirac66 ( talk) 16:29, 1 July 2012 (UTC)
All this extra complexity is a distraction from the main purpose of VSEPR, which is making things simple for students. Once you start getting into detail about exceptions, you're moving into full-scale molecular geometry calculation and all the computational chemistry involved. Any discussion of odd-electron species and exceptions should be short and to the point. -- Ben ( talk) 17:48, 1 July 2012 (UTC)
- Any discussion of odd-electron species should be at the same level as the rest of the article, which excludes all computational chemistry and all experimental structure methods too. But I think the current table is misleading in just including AX2E0.5 etc. with no extra information; it suggests that these odd-electron species are part of the standard theory although they are not found in most textbooks. So I propose to remove the odd-electron lines from the table and replace them by a short section near the end of the article with the following information.
- VSEPR can be extended to odd-electron molecules by treating odd electron as a pair.
- Molecule with odd electron is intermediate between molecules with 0 and 2, perhaps with a citation needed tag
- NO2 as example and compare to NO2+ and NO2-
- Brief mention of other examples as treated "similarly" - ClO2, UF5(? obscure example), CH3
- CH3 should be pyramidal but actually planar though distortion to pyramidal very easy
- I think this would be sufficient. Dirac66 ( talk) 17:03, 2 July 2012 (UTC)
I support that. Ignore UF5, it's not main group. Probably square planar due to second order Jahn-Teller or some other complicated effects. -- Ben ( talk) 19:05, 2 July 2012 (UTC)
This is quoted as an exception to VSEPR-- it is unreferenced--the fact that the structure details probably relate to the solid state could account for the deviation from linearity- mercury fulminate may have had its structure determined in the vapour phase which would be a real tour de force, and if so a bent configuration found there would be relevant. I do not have the access to journals to get to the bottom of this. Axiosaurus ( talk) 16:52, 19 June 2013 (UTC)
- You are right – it's not bent: see [1]. Removed, and added a {{cn}} tag for the other unreferenced example there. Double sharp ( talk) 15:35, 22 June 2013 (UTC)
- Trigonal prismatic – W(CH3)6, [Zr(CH3)62−
- Capped trigonal prismatic – [NbF72−
- Capped octahedral – [W(CO4Br3−
- Cubic – La(bpyO
2)+
4 - Dodecahedral ( snub disphenoid) – [Mo(CN)83−
- Not named? (looks like a distorted icosahedron) – a cerium nitrato complex
All from http://www.d.umn.edu/~pkiprof/ChemWebV2/
Are these even part of VSEPR theory? Or are most of these just exceptional cases like W(CH3)6 when VSEPR fails to predict the correct geometry? Double sharp ( talk) 14:30, 11 May 2014 (UTC)
- These are all transition metal compounds, which are mentioned as the first group in the section on Exceptions. In fact your first example, W(CH3)6, is the first molecule mentioned in the Exceptions section, and its geometry is described in its own article.
Dirac66 (
talk)
18:03, 11 May 2014 (UTC)
- Ah, OK. Perhaps more of these ought to be mentioned as examples of unusual geometries somewhere, though. Double sharp ( talk) 08:25, 12 May 2014 (UTC)
The missing talk entries from 12 May to this date? Asked because I recall having posted in talk, extensively, while I was editing last night, and at least that Talk Section appears to have been deleted. Please reply, knowledgable/responsible parties. Meanwhile, I am calling it to attention of admins. Cheer. Le Prof Leprof 7272 ( talk) 01:07, 31 May 2014 (UTC)
- Well, the page history there shows no posts to the talk page between November 2013 and the end of April 2014, and no removals or deletions - there isn't anything missing from the page. -
The Bushranger
One ping only
01:34, 31 May 2014 (UTC)
- Must have gotten lost then Bush, thanks for checking. Was not sure if there was an administrative way to "disappear" entries without leaving a trace that editors could follow. Will post about the edits, anew. Cheers. Le Prof Leprof 7272 ( talk) 02:03, 1 June 2014 (UTC)
Left an extensive Talk edit prior to editing a couple nights back, and it was somehow lost, so here in bullet are the changes made, and explanations as necessary. The key thrust is unverifiable text, and I tried to use a minimally destructively approach to begin to address and flag this. Note, though I did add a fair number of tags, several of those appearing, including the disilyne citation needed tag, were already in place. From bottom to top of article, here is a description of what was done:
- VSEPR and localized orbitals section:
- Moved specialized comment regarding nature of Slater determinant to the ref, so still accessible to ones seeking more info. Merged related sentences to make needed citations easier to provide. Reimprove section tag added (only citation is to Slater sentence, no other referencing in Section).
- Odd electron molecules section:
- Unnecessary line breaks removed, some sentence merging, and inline citation needed tags added where sentences were factual, quantitative, and not common knowledge. Reimprove section tag added.
- Exceptions section:
- Well referenced section, almost no work. Tightened up the Some AX2E2 molecules subsection/paragraph, including making overly long Si-O-Si example explanation as short as I could manage. Having this example occupy 80% of prose in this section is perhaps too much. Reduce this, and add other examples?
- Examples section:
- Bottom half of paragraph well referenced, top half not at all. Removed broken wikilink markup language. Did not tag section, but added inline citation needed tags where sentences are factual, quantitative, and not common knowledge.
- AXE method section:
- All factual, descriptive, and decidedly not common knowledge, yet not a ref to table content origin or any text sources; a completely unverifiable section! Tag added at opening.
- Description section:
- Moved VESPR pronunciation info and citation to this section from lede, and shortened text given the non-authoritative nature of that one source. Reduced paragraphing, shortened ax/eq trig bipyr descript. As above, this section is factual, descriptive, and decidedly not common knowledge, yet not a ref appears (apart from one minor I moved in). Leaving out inline citation needed tags; whole bloody section is unverifiable.
- Lede:
- Left an extensive comment about the structure of the lede, and lede citation, but did little to change the current lede (except to move out a reference to half of the pronunciation sentence, to a frosh Gen Chem text). See WP:LEAD regarding general structure expected, and so possible improvements, and to WP:LEADCITE to see aim of placing citations in main body, and having lede as easily read, and without content that is not verified in the main body. For reasons covered in preceding bullets, overarching article tags were placed.
Note, this all was more spread out, over the hours of the edits, originally, but since I am "backfilling" (reproducing the original comments), had to communicate this all at once. Apologies, cheers. Le Prof Leprof 7272 ( talk) 21:22, 1 June 2014 (UTC)
- I have now started to add some of the needed references. More coming.
Dirac66 (
talk)
00:05, 16 June 2014 (UTC)
- I have now added many of the long list of missing references, mostly to undergraduate textbooks. Here is the much shorter list of the claims for which I have not yet found references.
- History section: first suggestion credited to Tsuchida (1939)
- AXE method, Table 1, n=9, capped square antiprismatic geometry. [Note that the references for n=2-6 are given in the column headings.]
- AXE method, Table 2, CCl2 molecule (singlet state?), phosphate ion, and the three ions for n=8.
- Odd-electron molecules - several references missing, can find no support for the basic claim that one electron is treated as half a lone pair.
Dirac66 (
talk)
17:15, 25 June 2014 (UTC)
- Surprise. The 1957 paper of Gillespie and Nyholm does discuss the odd-electron species NO2 as intermediate between NO2+ and NO2-. I have now inserted this source, with some rewriting of the paragraph to conform to the source article. Dirac66 ( talk) 03:31, 12 February 2015 (UTC)
- I have now started to add some of the needed references. More coming.
Dirac66 (
talk)
00:05, 16 June 2014 (UTC)
Electron pair repulsion- not electrostatic according to Gillespie and why are transition metal complexes used as examples?
Two concerns:-
- According to Gillespie the repulsion between electron pairs in VSEPR theory is NOT electrostatic but due to the Pauli exclusion principle.(See as an example this paper Chemisty Education: Research and Practise in Europe: vol.2, No.2, pp 73-90 )
- Transition metal complexes are not normally considered in VSEPR theory - isn't it therefore confusing to use TiCl4 as an example. There are also other T. metal complexes quoted.
Axiosaurus (
talk)
15:56, 16 June 2014 (UTC)
- Reference one (Jolly) says electron repulsion. V8rik ( talk) 18:51, 16 June 2014 (UTC)
- Good questions which will require some reference checking. From memory I think most early papers and many recent books (such as Jolly) just say electron repulsion. However some more recent theoretical work does attempt to distinguish between Coulomb repulsion (electrostatic) and Pauli repulsion (effect of exclusion principle), and Pauli is often as important if not more so. Check the papers of Gillespie with his departmental colleague Richard Bader who was a quantum chemist.
- As for transition metals, some molecules do obey VSEPR predictions (see examples in the text) and many do not. I think we can give examples of each to show that VSEPR is not very reliable for TM complexes. Also, we should check the sources for the molecules which do have VSEPR geometries to see how many of these sources actually mention VSEPR.
Dirac66 (
talk)
00:16, 17 June 2014 (UTC)
- I have now deleted electrostatic repulsion and noted that Gillespie emphasized the importance of the Pauli principle, using as source another article of his in Coordination Chemistry Reviews. Dirac66 ( talk) 03:06, 18 June 2014 (UTC)
- thanks- real life is too pressing at the moment, questions are easier than answers! Axiosaurus ( talk) 08:46, 18 June 2014 (UTC)
I would suggest to rename the "Exceptions". Half of the examples are interesting cases, items under discussion, but not the exceptions. P99am ( talk) 09:06, 16 October 2014 (UTC)
- What name would you suggest for the "Exceptions" section? And which examples in that section are not exceptions? Dirac66 ( talk) 02:25, 17 October 2014 (UTC)
(This isn't a suggestion for the article, but rather for octadecahedron and edge-contracted icosahedron; I'm putting it here because the issue is a bit larger, but is not a problem for all the other polyhedron names; and also because I think this page would be quite a bit more watched.)
The names used by chemists and mathematicians for these polyhedra seem to be different:
- The chemist's trigonal is more often triangular among mathematicians (although I've seen the trigonal forms too);
- The chemist's seesaw is a mathematician's disphenoid;
- The chemist's capped is a mathematician's augmented;
- The chemist's dodecadeltahedron is a mathematician's snub disphenoid;
- The chemist's capped square antiprism and bicapped square antiprism are a mathematician's gyroelongated square pyramid and gyroelongated square bipyramid;
- The chemist's octadecahedron doesn't appear to have a standard name among mathematicians, but I haven't seen octadecahedron used in mathematics; a reasonable name using mathematical terminology would be an edge-contracted icosahedron.
This makes for great fun for the article titles! The obvious solution, I guess, is for the chemistry articles to primarily use the chemists' names (but cross-referencing the mathematicians' ones at the first mention), and for the mathematics articles to do the opposite. For the rarer molecular shapes (like bicapped square antiprismatic), the mathematicians' usage is probably primary.
The only trouble comes for the octadecahedron, which is barely mentioned at all in mathematics (because it cannot be constructed using equilateral triangles, it doesn't get an invitation to the Royal Deltahedron Club), and so its main appearance is in chemistry (where equilateral triangles aren't necessary)! However, the article uses an extrapolated mathematical name (i.e. a name that would be expected, but in practice I haven't seen used much if at all in mathematics), and reserves octadecahedron for its mathematical meaning (any polyhedron with 18 faces!), never mind that the name octadecahedron is very rarely encountered in mathematics because there it's so ambiguous, possibly referring to over 1014 topologically distinct shapes!
Per WP:PRIMARYTOPIC I think the subject of octadecahedron probably ought to be the [B11H112− structure, as it is in chemistry, with a brief note about the other 18-faced polyhedra at the bottom. Double sharp ( talk) 07:37, 28 March 2015 (UTC)
- P.S. Oh, this just gets better. About dodecadeltahedron; in chemistry this means the
snub disphenoid but in mathematics it more often means the
hexagonal bipyramid according to the latter article. Which is the primary use? In the meantime I've made dodecadeltahedron a disambiguation page listing both meanings.
Double sharp (
talk)
07:41, 28 March 2015 (UTC)
- Bravo for your efforts to sort out all this mess. Presumably the chemists who determined the more obscure molecular geometries just went ahead and named them without checking the mathematical literature. I agree with your approach to the problem - use the usual names in each field but cross-reference the other names as well. Dirac66 ( talk) 18:31, 29 March 2015 (UTC)
- Heads-up on the broken citation.--
Officer781 (
talk)
11:30, 30 March 2015 (UTC)
- I have turned dodecadeltahedron into a redirect to snub disphenoid, as I cannot find any example of the purported mathematical usage mentioned above outside Wikipedia (I have removed that from the hexagonal bipyramid article as well). Double sharp ( talk) 14:47, 19 September 2018 (UTC)
- Heads-up on the broken citation.--
Officer781 (
talk)
11:30, 30 March 2015 (UTC)
I understand that there was a debate above on the water molecule. However, I think this section, instead of talking about MO theory and its equivalence to localized orbitals, should instead focus on the difference between VSEPR and MO or VB orbitals. This should include both the purpose (as a way to predict the shape rather than rationalize the exact bond angles and other properties by computational methods) and also the difference between VSEPR electron domains and such orbitals (eg. the double bond, regardless of having two electron pairs, is regarded as a single electron domain. Likewise, each electron pair in hypervalent molecules is counted as an electron domain even though some of those pairs are nonbonding ligand pairs). The debate about the water molecule should not be done here but at the hybridization article and the distancing of VSEPR from orbitals itself should serve to negate the argument. I shall try to re-write this section for now.-- Officer781 ( talk) 11:37, 30 March 2015 (UTC)
- Speaking of which, can we settle on the issue of whether, in AXE, the X represents a pair of electrons or an electron domain. In most other areas of this article it seems to imply the latter by stating that X represents a ligand while E represents a lone pair, but under the "Description" section it seems to imply the former, then struggles to explain double and triple bonds.-- Officer781 ( talk) 12:29, 30 March 2015 (UTC)
- It is true that the equivalence of localized and delocalized orbitals is not the main point here, and in fact it is discussed elsewhere at Localized molecular orbitals. However I think we should still mention that VSEPR is related to localized MO's, with a link to that article for more details.
- Also, I think some rewriting is still necessary. For example, I do not understand the claim that VSEPR predicts bond angles but quantum mechanics justifies them. I would say that VSEPR predicts bond angles approximately, and quantum mechanics can make more exact predictions of bond angles and other properties.
- As for the AXE notation, X represents a ligand whether it forms a single or a multiple bond. This is clear for example in the book by Petrucci et al., which says for HCHO (p.416) that We count three (authors' italics) electron groups around C, 2 for the C-H bonds and 1 for the C=O double bond. We should make this clear in the article, and point out that it is justified by the bond angles. Dirac66 ( talk) 01:36, 31 March 2015 (UTC)
- Gillespie has always been clear that VSEPR has nothing to do with MOs, LMOs or VB. I do not think we should mention any of them in a way that suggests there is a link. -- Bduke (Discussion) 02:07, 31 March 2015 (UTC)
OK, we now have the following short concluding paragraph:
- The VSEPR theory places each pair of valence electrons in a bond or a lone pair found in a local region of the molecule based on the Pauli exclusion principle. While this is frequently taught in chemistry textbooks in conjunction with orbital models such as orbital hybridisation and molecular orbital theory, the approach is completely different. Electron pairs can constitute degrees of repulsion independent of their valence configuration, which is why bonding pairs are treated identically regardless of the bond order and certain molecules can have more than 4 electron pairs regardless of the octet rule.
I think the first two sentences are clear and quite satisfactory. However the final sentence now seems to be the most confusing in the entire article. What is a degree of repulsion? And what exactly is being claimed in the which is why clause? Can this be rewritten more clearly? If not, perhaps we should just delete the sentence and end the article with the approach is completely different.— Preceding unsigned comment added by Dirac66 ( talk • contribs)
- Removed. Although I find this to make the paragraph sound like a cliffhanger. Would probably want to explain how the approaches are different.-- Officer781 ( talk) 03:19, 1 April 2015 (UTC)
After digging through VSEPR papers I realized Gillespie does mention a section in one of his papers that exceptions transition metal molecules can be explained in that the ligands are strongly-interacting with the atomic core and produce electron pairs that occupy two regions on opposite sides of the atom. Sounds too similar to sd hybrids but I feel that amounts to an explanation so I've rewritten it and removed it as an exception (which is meant to show the limiting areas of VSEPR without a sufficiently convincing explanation).-- Officer781 ( talk) 02:19, 17 April 2015 (UTC)
This article so far only distinguishes bonding pairs from lone pairs. VSEPR has an analogue of Bent's rule which should be introduced: electron pairs of more electronegative substituents constitute greater repulsion. Lone pairs are a special case of this rule. Also, bond order affects degree of repulsion. Putting this as a to-do for myself and in case anyone else has any input or wants to give a go.-- Officer781 ( talk) 14:02, 17 April 2015 (UTC)
The user Haminoon who I suspect has no formal chemistry training keeps reverting my edit concerning using a formula to to predict lone pairs as appose to using a Lewis diagram.
View the edit here: https://en.wikipedia.org/?title=VSEPR_theory&oldid=prev&diff=685751252
- I have taught university chemistry and I agree with Haminoon that this method does not belong in the Wikipedia article. The task of Wikipedia is to present subjects as described in widely used textbooks, rather than simplified versions on obscure web sites, which is presumably what Haminoon means by not notable. And in this case there is a good reason why textbooks do not use this simplified method: it only works for atoms with a single central atom surrounded by either all hydrogens or no hydrogens. For a molecule such as CHCl3, one would have to specify that the number of hydrogens is multiplied by two and the number of other non-central atoms by eight. Worse, for molecules with more than one central atom (e.g. ethylene, acetic acid, benzene, S2F10, etc. etc.) there is no simple general formula and the best advice to students is to draw a Lewis structure. So the textbook authors have decided that it is simpler to skip the shortcut which only works sometimes and just present the one method which works most often.
- I will re-delete the paragraph in question. Dirac66 ( talk) 00:47, 15 October 2015 (UTC)
So it sounds like your pretty educated on the topic, but all the cases you presented are accounted for with knowledge.
H and Non-H molecule:
L = v -(?x8) + (?x2)
Multiple central atoms:
Treat each center as half and then times by 2(or by as many centers as there are)
eg S2F10
L = (41-(5x*8)) x 2
So in my view this formula is actually more accurate and more simple to learn then Lewis. Look over the formula and insure your not thinking of the old one.
Unit388 (
talk)
02:24, 15 October 2015 (UTC)
That may or may not be the case, but it is not important. Until this new idea has been well discussed in reliable sources, it can not be included in Wikipedia. Wikiversity is not a reliable source. -- Bduke (Discussion) 08:21, 15 October 2015 (UTC)
- Agreed. Also for Unit388's information, note that changing the method on this talk page is original research which is not allowed in Wikipedia, partly because the ideas have not been tested by the usual processes in the scientific literature such as refereeing and reaction by others. In this case your new fix is quickly shown to be incorrect: for S2F10 L = (41-(5x8)) x 2 = 2 lone pair electrons or one lone pair. However the Lewis structure shows that there is no lone pair but instead an S-S single bond which is not included in your calculation. Similarly for ethylene, L = (6 - (2x2)) x 2 = 4 or two lone pairs. Again there are no lone pairs but rather a C=C double bond. This illustrates why we want reliable sources. Dirac66 ( talk) 11:37, 15 October 2015 (UTC)
I was a first year chemistry student at Otago University when I created this formula. Now I see yes you're right, It doesn't work for all cases such as double bonds but, it does work almost every problem a first year chemistry student would be given. Anyway quit science to do law.
Unit388 (
talk)
- @ Officer781:
- @ Norbornene:
To Officer781: On 23 April 2015 you wrote the following first sentence in the section Transition metal molecules: Many transition metal compounds have unusual geometries, which can be ascribed to absence of valence shell lone pairs and ligand bonding interaction with the d-subshell. As editor Norbornene pointed out yesterday, this can be interpreted in 2 ways: it can be read which can be ascribed to absence of valence shell lone pairs and TO ligand bonding interactions ..., meaning that the ligand bonding interactions are PRESENT. Or it can be read as which can be ascribed to absence of valence shell lone pairs and OF ligand bonding interactions ..., meaning that the ligand bonding interactions are ABSENT. Norbornene could not figure out which is correct, and neither can I even after looking briefly through the source paper by Kaupp. So could I ask you to decide which you meant, and then to clarify the sentence, perhaps by adding TO or OF as appropriate. Thanks in advance. Dirac66 ( talk) 01:20, 14 August 2016 (UTC)
- Clarified. Hopefully this clears it up. Do ping me if there are any further issues.-- Officer781 ( talk) 02:38, 15 August 2016 (UTC)
@ Bear-rings: Could you explain why you unilaterally moved the article? There is no policy against acronyms in titles. VSEPR is the most common name for it. Since the move is likely to be controversial, as the name has been VSEPR theory for years, you should do a requested move. Could you move the page back, and if you think the name should be changed, do a WP:RM. Galobtter ( pingó mió) 13:19, 23 November 2017 (UTC)
- I've moved it back pending further discussion. The acronym is much more commonly used than the spelled-out name, indeed!
Double sharp (
talk)
13:43, 23 November 2017 (UTC)
- Many non-English speaking readers do not know the meaning of this acronym.
Bear-rings (
talk)
14:19, 23 November 2017 (UTC)
- Perhaps they might not, but given that "VSEPR" alone is by far much more common than spelling it out in the literature, and the originally spelt-out version is given in the first line of the text, I don't see why that should be a problem.
Double sharp (
talk)
14:30, 23 November 2017 (UTC)
- It's not a problem, just a help to non-English readers — Preceding
unsigned comment added by
Bear-rings (
talk •
contribs)
- I'd say it'd help non-chemistry readers (don't see how native speakers will understand what VSEPR stands for more than non-native/non-english ones)- but it's not like "valence shell" and "electron pair repulsion" mean anything to non-chemistry people.
Galobtter (
pingó mió)
15:03, 23 November 2017 (UTC)
- Therefore, there are two complementary reasons for replacing this acronym with a text consisting of common language words
- I'd say it'd help non-chemistry readers (don't see how native speakers will understand what VSEPR stands for more than non-native/non-english ones)- but it's not like "valence shell" and "electron pair repulsion" mean anything to non-chemistry people.
Galobtter (
pingó mió)
15:03, 23 November 2017 (UTC)
- It's not a problem, just a help to non-English readers — Preceding
unsigned comment added by
Bear-rings (
talk •
contribs)
- Perhaps they might not, but given that "VSEPR" alone is by far much more common than spelling it out in the literature, and the originally spelt-out version is given in the first line of the text, I don't see why that should be a problem.
Double sharp (
talk)
14:30, 23 November 2017 (UTC)
- Many non-English speaking readers do not know the meaning of this acronym.
Bear-rings (
talk)
14:19, 23 November 2017 (UTC)
I checked two general chemistry textbooks (Petrucci et al 8th ed, Whitten et al 4th ed). They both combine the full name AND the acronym, so that the title of the section which introduces this subject is Valence-Shell Electron-Pair Repulsion (VSEPR) Theory (one book with the hyphens, one without). I suggest this combined version would be the most helpful to non-chemist readers unfamiliar with the subject. More advanced readers can of course just type in VSEPR and use the redirect. Dirac66 ( talk) 00:01, 24 November 2017 (UTC)
- I checked my general chemistry book - zumdahl chemistry - and that full form is only used the first time it is introduced - it's not used for section titles or any time afterwards. Same with many acronyms - the full form is introduced but the acronym is used much more. Non-chemist readers - well I don't know how a non-chemist would benefit from "valence-shell" and "electron-pair repulsion" - doesn't really help does it? It's also far too long.
Galobtter (
pingó mió)
03:59, 24 November 2017 (UTC)
- Congratulations, finally an excellent point of view, the title would be too long. :-))). Bear-rings ( talk) 14:43, 24 November 2017 (UTC)
Hello fellow Wikipedians,
I have just modified one external link on VSEPR theory. Please take a moment to review my edit. If you have any questions, or need the bot to ignore the links, or the page altogether, please visit this simple FaQ for additional information. I made the following changes:
- Added
{{ dead link}}tag to http://people.ok.ubc.ca/wsmcneil/339/Sci2004.pdf - Added archive http://arquivo.pt/wayback/20160523113736/http://www.iumsc.indiana.edu/ to http://www.iumsc.indiana.edu/
When you have finished reviewing my changes, you may follow the instructions on the template below to fix any issues with the URLs.
This message was posted before February 2018.
After February 2018, "External links modified" talk page sections are no longer generated or monitored by InternetArchiveBot. No special action is required regarding these talk page notices, other than
regular verification using the archive tool instructions below. Editors
have permission to delete these "External links modified" talk page sections if they want to de-clutter talk pages, but see the
RfC before doing mass systematic removals. This message is updated dynamically through the template {{
source check}} (last update: 5 June 2024).
- If you have discovered URLs which were erroneously considered dead by the bot, you can report them with this tool.
- If you found an error with any archives or the URLs themselves, you can fix them with this tool.
Cheers.— InternetArchiveBot ( Report bug) 12:50, 24 December 2017 (UTC)
ভিএসইপিআর তত্ত্ব(VSEPR Theory- Valence Shell Electron Pair Theory") হল একটি এমন তত্ত্ব যার মাধ্যমে কোনো রাসায়নিক গঠনের বা যৌগের বিভিন্ন রকমের প্রধান চারিত্রিক বৈশিষ্ট্য যেমন-ইলেকট্রন ঘনত্ব,অণুর গঠন,বন্ধন ক্ষমতা ইত্যাদি নির্ণয় করা সম্ভব। Aishik Datta ( talk) 20:42, 25 August 2020 (UTC)
This article can change by all. Aishik Datta ( talk) 20:43, 25 August 2020 (UTC)
- The following discussion is closed. Please do not modify it. Subsequent comments should be made in a new section. A summary of the conclusions reached follows.
- The result of this discussion was merge. Officer781 ( talk) 00:04, 6 September 2020 (UTC)
I propose to merge steric number here as the rationale is pretty much the same as Talk:AXE method. Basically, the term is only used in the context of VSEPR and as such does not warrant a separate page. Pinging @ Dirac66: and @ Bduke: for comment.-- Officer781 ( talk) 11:09, 5 September 2020 (UTC)
- Yes, I agree. I think Steric number should be one section of this article on VSEPR theory, perhaps following Overview. And I also notice that the article on Steric number is now completely unreferenced. I will now add a reference for the definition of steric number, which can of course be transferred to the new merged article. Dirac66 ( talk) 15:17, 5 September 2020 (UTC)
- @ Dirac66: Thanks. I have now merged the article here as a section following Overview.-- Officer781 ( talk) 00:00, 6 September 2020 (UTC)
- C-Class vital articles
- Wikipedia level-5 vital articles
- Wikipedia vital articles in Physical sciences
- C-Class level-5 vital articles
- Wikipedia level-5 vital articles in Physical sciences
- C-Class vital articles in Physical sciences
- C-Class Chemistry articles
- High-importance Chemistry articles
- WikiProject Chemistry articles
