|
| This It is of interest to the following WikiProjects: | ||||||||||
| |||||||||||
I think the article should begin with chemical and physical information about potassium cyanide, instead of opening by saying that it's the most common instrument of suicide. (Incidentally, if anyone can cite statistics supporting this claim, that would help.)
rajneesh 06:12, 8 Aug 2004 (UTC)
According to Suicide, sourced from [1], in the US firearms are by far the most common method of suicide & four times more common than the next. After that, it lists asphyxia, hanging, drug overdose, carbon monoxide poisoning, jumping from height, stabbing or exsanguination, and drowning. In the Republic of Congo, where firearms are less readily available, the most common method among 15-24 year olds is hanging, followed by firearms, followed by motor vehicle exhaust, followed by poisoning (all methods) [2]. And so on. Obviously methods will vary somewhat from one country to another, but this claim doesn't seem to be even remotely true, so I've removed it along with some accompanying material that seemed equally spurious. -- Calair 03:39, 20 Dec 2004 (UTC)
Was cyanide really 'commonly used as rat poison'? I know there's an anti-smoking ad out there mentioning the presence of cyanide in rat poison, but AFAIK most poisons are based on anticoagulants, heavy metals, or strychnine. -- Calair 00:03, 13 May 2005 (UTC)
- Anticoagulants are usually favoured these days, but cyanide salts have been used in the past, yes. Zyklon B was originally developed as a pesticide (for fumigation). Physchim62 10:16, 3 Jun 2005 (UTC)
- I remembered the use of cyanide for fumigating insects, just hadn't heard of it being used for rats until this article. -- Calair 15:01, 3 Jun 2005 (UTC)
- Potassium cyanide - needs chemist to check factually dubious section. RayGirvan 21:37, 2 Jun 2005 (UTC)
Citation for the alleged antidotal effect of glucose? Quite apart from cake icing mostly being sucrose, it looks chemically nonsense to me. I suspect it's a misinterpretation of texts (eg [3] and [4]) reporting that glucose reduces the toxicity of dicobalt edetate when the latter is used as a cyanide antidote. RayGirvan 20:20, 2 Jun 2005 (UTC)
- After a lot of Googling I found one article [5] that appears to claim an antidotal effect for glucose alone. By my limited Spanish and with the aid of a machine translator, it appears they mixed glucose with a cyanide solution and found it less toxic to mice than the same concentration of cyanide without glucose. Running part of that article through a machine translator:
- In relation to the glucose, anecdotally it is mentioned that, the abundant sweet wine ingestion was the reason for which Rasputín (Grigori Yefímovich, 1872-1916), Russian monk hated by the nobility, survived an attempt of envenenamiento with cyanide. Without a detailed scientific explanation, the use of hypertonic glucosado serum to the 30-50% intravenous route like main antidótico treatment, or cotreatment of the fundamental antidótica therapy is suggested. The glucose would react with the in the middle acid cyanide [I suspect that's actually "would react with cyanide in an acid medium"], giving a heptosa, with disappearance of the ciánica molecule in the reaction... In the present test, a solution of D (+) anhydrous glucose to 40%, antagonizó totally the poisonous effect of 40 a lethal dose of mg/kg [should read "a lethal dose of 40 mg/kg", I think] of potassium cyanide, after one hour of incubation... according to the observed results, the union cyanide-glucose is apparently stable, at least under physiological conditions in mice and to the contact with the peritoneal liquid. On the other hand, according to the expressed thing by Calabuig (2004), this one union is facilitated in the middle acid, reason why it would be possible to hope that, under a toxic picture, where this present the lactic acidosis product of the increased anaerobic metabolism, the reaction took place in a smaller time... This, nevertheless, also contradicts the expressed thing by other authors (Pavia and col., 1976), that indicates the requirement of slightly basic means for the spontaneous occurrence of the reaction nonsingle, but, who in the middle acid, the cianohidrina would be disturbed releasing cyanide. Thus, the possible molecule formation must even be considered spontaneous that is nontoxic and stable and that derives in the partial or total diminution from the toxic power of the cyanide, or, as Calabuig mentions (2004), takes place the disappearance of the ciánica molecule in the reaction.
- So there does seem to be some basis for believing that glucose alone might be an antidote to cyanide poisoning, but I don't think Wikipedia should be suggesting that this is anything other than an experimental treatment. (I'm vaguely bothered by the number of sites that are faithfully reproducing Wikipedia's claims on this, too.) I'll take a stab at rewording that section - see whether you think the tag needs to stay on after that? -- Calair 00:04, 3 Jun 2005 (UTC)
- Well done for finding the Spanish article, this is the first investigation I have seen of the oft-repeated Rasputin story. However, dispite the title of the article, they do not show that glucose is a cyanide antidote, merely that administering glucode and cyanide at the same time (in fact, after a 10 min incubation of the glucose/cyanide mixture) reduces the toxicity of cyanide. This is probably due to a condensation reaction between the cyanide and the aldehyde grouping of the glucose molecule, leading to a cyanoheptose which is less toxic and more easily dealt with by the body.
- Three cyanide antidotes are known, although there is some controversy over the best one to use.
- In any case, this section is probably better placed in Cyanide (although the relevant section there should also be cleaned up a bit).
- Physchim62 10:13, 3 Jun 2005 (UTC)
- (Also note that amyl nitrate is used in the management of cyanide poisoning.) [Unsigned edit by
Big Bob the Finder 23:01, 12 Aug 2005 (UTC)]
- It is, but see HSE recommendations for first aid treatment of cyanide poisoning (UK) for a discussion of its probable lack of effectiveness. Physchim62 18:11, 12 August 2005 (UTC)
- (Also note that amyl nitrate is used in the management of cyanide poisoning.) [Unsigned edit by
Big Bob the Finder 23:01, 12 Aug 2005 (UTC)]
- You're right, the experiment doesn't actually show glucose to be an antidote (except in a sort of pre-emptive fashion). However, from what I could make out the discussion was speculating that the same process might take place within the bloodstream, which would make IV glucose a useful antidote. Of course, speculation is not demonstration, but it does at least show that under some circumstances glucose can neutralise cyanide.
- And yes, an awful lot of this article is really about CN- in general rather than specifically KCN, and should go in Cyanide.-- Calair 16:33, 3 Jun 2005 (UTC)
- And yes, . I agree that a lot of this article should really be under Cyanide
- Regardless, we're not here to draw conclusions ourselves. We don't use primary sources. This isn't a place for original thesis. Thus, unless it is published first by another source, I don't think the effects of glucose should be published here. Comatose51 03:42, 14 Jun 2005 (UTC)
- The German paper Handlungsfähigkeit bei tödlichen oralen Intoxikationen mit Cyan-Verbindungen from a journal for forensic medecine (Rechtsmedizin 9: 56 - 61, 1999) cites a case of a girl who accidentally drank poisoned, sugared tea and whose poisoning symptoms did not became evident until after half an hour later. They state that this could be due to the sugar binding some of the CN- ions. Although they do not give any details, they cite glucose as one of the factors responsible for a delayed poisoning by cyanide. There are no reasonable sources about Rasputin, but it may also be that his stomach was not acid enough or that he used lemon for his tea (thereby releasing HCN before drinking it) for getting poisoned. Phrood 21:42, 24 Jun 2005 (UTC)
IPCS/CEC refers to amyl nitrite as a potential antidote [9], whereas the HSE discusses amyl nitrate [10]. Are amyl nitrate and nitrite both in use as CN- antidotes, or has one of these sources goofed? -- Calair 23:52, 14 August 2005 (UTC)
- I think the HSE has goofed, as have I, further up this talk page! Amyl nitrite ( CAS number [110-46-3]) is the "traditional" first aid for cyanide poisoning. Amyl nitrate would probably be better than nothing, as it should act as a vasodilator and also increase methemoglobin levels, but it would have a shorter shelf live (even the nitrite should be stored at <15 °C) and a nasty habit of exploding. (Check-source Bretherick, L. (Ed.) (1986). Hazards in the Chemical Laboratory (4th Edn.). London:Royal Society of Chemistry. ISBN 0-85186-489-9.). Physchim62 20:50, 15 August 2005 (UTC)
The toxicology section at cyanide now seems to be in a state to receive the information which is here and relates to cyanides in general. Does anyone object to me merging across this material? I would leave a short paragraph mentioning that KCN is poisonous as are all alkali cyanides and directing the reader to cyanide for more information. Physchim62 18:02, 16 August 2005 (UTC)
- Yes, that's where it ought to be. I kept meaning to do the same thing myself, but never got around to it :-) -- Calair 23:09, 16 August 2005 (UTC)
__________________________________________________________
I have a few questions maybe someone can help
If cyanide and carbon monoxide inhibit the same protein complex on the electron transport chain then would the clinical presentations be the same? If so why do we treat carbon monoxide poisoning different then cyanide poisoning?
- The quick answer is that they don't. Cyanide ions inhibit the mitochodrial electron transport chain, carbon monoxide blocks hemaglobin. Herein lies the difference in clinical symptoms. Physchim62 19:20, 29 October 2005 (UTC)
- I heard that iron II and iron III are the points of attack of these two molecules. So one has more affinity to iron II the other to Iron III so you get a difference. Stone 07:57, 30 January 2006 (UTC)
Anybody know how you'd find Potassium Cyanide (KCN) around the house? I need it for a lab experiment. March 6, 2006
- If you're qualified to work with KCN and have a legitimate reason for doing so, you should already know the proper channels for obtaining it. If you're not, I don't think giving you that information would be a terribly good idea. -- Calair 09:08, 7 March 2006 (UTC)
Well, guess what? I got a hold of it :)
- Cool, does it work? Hello? Hello? Just zis Guy you know? 15:13, 25 April 2006 (UTC)
- Does what work? It's a compound. What do you mean?
Anyway, encyclopedia needs data about how to obtain various substances otherwise it would be either incomplete or censored. Potassium (or any other alkali metal) cyanide could be synthesized by direct reaction between hydroxide (KOH in this case) and hydrocyanic acid. Is there a more effective way ? - Yyy 10:24, 20 July 2007 (UTC)
First I want to say that I'm not a depressed person or anything like that. It was a promiss, and about honour.
God sake!
For about a half year ago I took potassium cyanide. It had a good effect. I begun deep and fast breathing and lost contions after about a minutte.
I woke up at a hospitale. It was a miracle.
Yesterday and the day before yestarday I took the same amount again (ca 1 gram), but it had almost no effect at all. I was just deep and fast breathing for about half a minutte.
Is it possible that they have given me some kind of vaccine?
I work on a lab so the quality were about 99% for all tree cases, but the first atemt was from a other container, can that bee the reason?
I stord the KCN for the two last atempt at me room in a good isolated sample glass for tree mouth, but can it have been oxidated anyway?
Or am I just not supposed to die? I think so...
- Any news articles/publications/etc to confirm this story?
- Also, please do not delete material from other people's comments on Talk pages (as you just did to mine) without a good reason. -- Calair 23:18, 1 April 2006 (UTC)
Well, I think it depends, how much did you take? If you took like, a tablespoon the first time, and just a miligram the second time, then that makes sense... Or, maybe your body just rejected it.
- Oh, never mind, 1 gram. Well, I assume you put it in a drink. Did you let it sit for a few minutes, or just drink it right up?
I reverted these edits because it looks suspiciously like copyvio, and because the material is inaccurate. For instance, it claims: "Nobody has ever lived to tell the tale — how does potassium cyanide taste?" This is false, as is the claim of instant death; there have been quite a few cases of people who ingested cyanide and survived for days afterwards, some eventually making a full recovery after treatment.
See e.g. this article, with case reports including this one: "A 54-year-old man ingested 3 g potassium cyanide and arrived in hospital unconscious and with respiratory arrest. The patient was resuscitated and 12 g of sodium thiosulfate was given intravenously... After 30 min the patient breathed spontaneously, after 90 min he moved his extremities and eyes, and after 6 h he was fully alert. No sequelae were observed." -- Calair 00:10, 9 July 2006 (UTC)
Not much to say here because KCN, due to its annoying toxicity, is not usually employed in a laboratory. It is a pain to handle and NaCN can usually be used instead (cheaper and slightly less toxic) So its a bit of an irrelevant article Stui 21:12, 4 August 2006 (UTC)
Is this really relevant or useful to anyone who didn't look up 'potassium cyanide' without the intention to learn about minutiae from random video games/films?
Well, Some one told me in my chemistry class that if potassium cyanied touches your blood or got mixed with it due to some kind of wound, it start crystalizing your blood and you will die ????? was it a romur or is it true ??? wanna know that really if we got potassium cynaide got mixed with our blood due to some wound, do it harm that much that a person can die ????
Mohammad Adil
19:39, 27 August 2007 (UTC)
In the infobox on the right, what does "solubility in other solvents" mean? What kind of "other solvents"?
And also, I find that 71.6 g/100 ml is actually the solubility in water. See:
-- Quest for Truth ( talk) 19:41, 6 March 2008 (UTC)
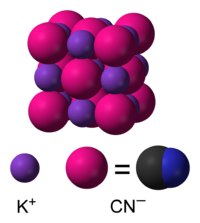
I noticed this discussion (and its other half) and decided to reinvestigate the structure of KCN, a compound I had tried to draw the structure of a while ago. The problem is that the cyanide ions are constantly rotating, so it is difficult to represent the structure. Short of creating some sort of fuzzy ion, the best option is to represent the cyanide ion as a sphere, as shown below.
The drawback to this approach is that it needs explanation, and may confuse readers who are not familiar with the structure of the cyanide ion.
Perhaps such an image would be best placed in a section on the structure of KCN, where the disorder could be discussed. If the image went in the infobox, it would be difficult to ensure readers had seen the accompanying explanation.
Ben ( talk) 17:58, 29 December 2008 (UTC)
- Nice work, as usual. Where did you get the coordinates and are they for below the disorder-order transition? My conversation with Aushulz was mainly to help him not depict KCN as a triatomic molecule and then we digressed into discussion of the fact that ionic solids are polymeric. Regarding your image, I agree that the blurred image would blow the minds of readers and require a lond explanation. Wells' Structural Inorganic book discusses the NaCN structure as being "closely related to NaCl" with six-coordination at the anions and the cations. And he points out that KCN is isostructural. Each K+ has two pi-bonds to CN and two bent K---C and two bent K---N links.-- Smokefoot ( talk) 18:27, 30 December 2008 (UTC)
I think this discussion could form the basis of a nice subsection on structure! The paper I got the coordinates from is given on the image description page. I really should get a copy of Wells, but it's out of print. Auschulz is just the sort of reader would appreciate a discussion of structure in this article.
Season's greetings!
Ben ( talk) 23:42, 30 December 2008 (UTC)
- OK, I've made a more descriptive image, which I hope will be useful:
- X-ray crystallographic data from H. T. Stokes, D. L. Decker, H. M. Nelson, J. D. Jorgensen (1993). "Structure of potassium cyanide at low temperature and high pressure determined by neutron diffraction". Phys. Rev. B. 47 (17): 11082–11092.
doi:
10.1103/PhysRevB.47.11082.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link).
- "The CN− ions undergo rapid reorientations centered at the Wyckoff 4a position (0,0,0). The time-averaged symmetry of the CN− ions about that point is cubic (point group m-3m). The rotation is not free, but is hindered. The potential energy of the CN− ion is lower for the <111> directions of orientation, and the CN− ion spends more time near those minima."
The photo of the KCN crystals is shown against a Euro 1 cent piece and what appears to be the tip of a writing device. People outside the EU may not be able to discern the size of the sample shown. Suggest using a photo with the crystal sample against a measurement device such as a ruler.
173.62.183.174 ( talk) 21:23, 8 April 2010 (UTC)
I am a highschool chemistry teacher, and I found this in the supply closet. It has never been opened. The other chemistry teacher and I had no idea on what its used would be in a high school classroom. Is it something that needs to be removed? I did not notice it on the state band list. A cracked bottle of Bromine was found in the closet the year before I was hired (there has been a new chemistry teacher every year for a while) —Preceding unsigned comment added by 71.143.178.136 ( talk) 17:33, 16 October 2010 (UTC)
Not really the best place to ask. This page is for discussing improvements to the article. But since you did... If you aren't confidant of using it (and you don't sound confidant), you probably should get a contractor to haul it away. See
[11],
[12],
[13]. --
Rifleman 82 (
talk)
18:47, 16 October 2010 (UTC)
- UPDATE: The MSDS has been restored, as the Important Notes section states under "Effects of short-term exposure" that "The substance is severely irritating to the eyes, skin and respiratory tract. The substance may cause effects on the cellular respiration. This may result in convulsions and unconsciousness. Exposure could cause death." 96.231.177.2 ( talk) 12:28, 24 Oct 2013 (UTC) ***
I was amazed to see that the hazardous effects of inhalation, ingestion and absorption do not include death. (In the linked MSDS.)
I consider this so irresponsible that I am removing the link. I understand that this is debatable, since the SDS is by a pseudo-government body (International Programme on Chemical Safety) But it does not meet the basic requirement of properly notifying reader of potential hazard - death qualifies, don't you think? This is the link I removed:
http://www.inchem.org/documents/icsc/icsc/eics0671.htm. I spent several minutes looking for another authoritative MSDS, but didn't find one, sorry. A bit further inquiry indicates even via dermal absorption, a couple of milliliters of saturated NaCN could be fatal (assuming toxic response is same as in rabbits on a per kg basis)
72.172.11.228 (
talk)
23:10, 30 May 2013 (UTC)
I have seen this sentence in this article "The moist solid emits small amounts of hydrogen cyanide due to hydrolysis, which tastes like bitter almonds". But this statement does not have any citation. So either it should be removed or the author should cite it properly with reference. Abhilashkrishn ( talk) 14:03, 12 August 2013 (UTC)
- It just seems plain vandalism, if you ask me. Engalazillo ( talk) 08:37, 22 November 2013 (UTC)
Visually, does anyone know of a reliable picture for potassium cyanide crystals (ie: What they actually look like) that can be placed onto the Wikipedia page? Are Potassium Cyanide crystals distinctive in some way? If you look at :
http://bioprepwatch.com/stories/510507628-chemist-sentenced-for-potassium-cyanide-possession
You see a picture that states it is a picture of potassium cyanide:
https://jnswire.s3.amazonaws.com/jns-media/57/f5/49204/potassiumcyanide.png
Can anyone visually verify that this picture is of Potassium Cyanide. If so, what features does Potassium Cyanide have that enable it to be tested VISUALLY?
It is said that Potassium Cyanide produces Hydrogen Cyanide, with the Hydrogen Cyanide (HCN) smelling of "bitter almonds" WHEN it is added to acids or acid salts (according to www.inchem.org/documents/cicads/cicads/cicad61.htm). So this can be used as a test of Potassium Cyanide.
Otherwise, how would one visually inspect a Potassium Cyanide crystal to make sure it is Potassium Cyanide? ASavantDude ( talk) 18:51, 28 July 2015 (UTC)
Hello fellow Wikipedians,
I have just modified one external link on Potassium cyanide. Please take a moment to review my edit. If you have any questions, or need the bot to ignore the links, or the page altogether, please visit this simple FaQ for additional information. I made the following changes:
- Added archive https://web.archive.org/web/20060517035532/http://www.npi.gov.au/database/substance-info/profiles/29.html to http://www.npi.gov.au/database/substance-info/profiles/29.html
When you have finished reviewing my changes, you may follow the instructions on the template below to fix any issues with the URLs.
This message was posted before February 2018.
After February 2018, "External links modified" talk page sections are no longer generated or monitored by InternetArchiveBot. No special action is required regarding these talk page notices, other than
regular verification using the archive tool instructions below. Editors
have permission to delete these "External links modified" talk page sections if they want to de-clutter talk pages, but see the
RfC before doing mass systematic removals. This message is updated dynamically through the template {{
source check}} (last update: 5 June 2024).
- If you have discovered URLs which were erroneously considered dead by the bot, you can report them with this tool.
- If you found an error with any archives or the URLs themselves, you can fix them with this tool.
Cheers.— InternetArchiveBot ( Report bug) 02:04, 22 May 2017 (UTC)
In this article:
A number of prominent persons were killed or committed suicide using potassium cyanide, including members of the Young Bosnia and famous personalities in the Third Reich, such as Erwin Rommel, Hitler's longtime companion Eva Braun, Joseph Goebbels, Heinrich Himmler, and Hermann Göring.
In Cyanide poisoning article:
Cyanide, in the form of pure liquid prussic acid (a historical name for hydrogen cyanide), was the favored suicide agent of the Third Reich. It was used to commit suicide by Erwin Rommel (1944), after being accused of conspiring against Hitler; Adolf Hitler's wife, Eva Braun (1945); and by Nazi leaders Heinrich Himmler (1945), possibly Martin Bormann (1945), and Hermann Göring (1946).
-- Inconexo ( talk) 12:05, 4 August 2017 (UTC)
The article contained the following lines in the section about KCN toxicity:
[Potassium cyanide's] toxicity when ingested depends on the acidity of the stomach, because it must react with an acid to become hydrogen cyanide, the deadly form of cyanide. Grigori Rasputin may have survived a potassium cyanide poisoning because his stomach acidity was unusually low.
The source for these claims was cited as John Emsley's The Elements of Murder: A History of Poison, a pop science book. Emsley's book does contain this brief mention of potassium cyanide, which is buried in the middle of a much lengthier discussion of mercury cyanide:
That Barnett did not immediately die of cyanide poisoning can be attributed either to the fact that the solution [of mercury cyanide] he drank was not acidic, or that his stomach contents weren't. This latter state of affairs is very rare, but not unknown, and indeed it had saved the infamous Russian monk, Rasputin, who survived an attempt to poison him with potassium cyanide in 1916. The deadly form of cyanide is hydrogen cyanide (HCN) which requires acid for its formation.
Emsley does not provide any sources for these remarkable claims, which are highly implausible.
- In this section of the book, Emsley has been discussing mercury cyanide. He claims that an acid can break the relatively strong Hg-CN bonds, freeing up both the mercury and cyanide and making the compound toxic. This is at least plausible. By contrast, potassium cyanide readily dissociates in aqueous solution. There is no need for an acid to break the K-CN bonds, as there is with Hg(CN)2, and I fear that Emsley has here conflated the two cyanide compounds.
- But perhaps Emsley's claim is not that undissociated KCN isn't toxic, but rather that the CN- ion isn't toxic unless converted to HCN, as he states in his last sentence. Such a claim is far from obvious, and accepting it requires references beyond a single factoid line in a pop sci book.
- Suppose we take this claim that cyanide is only toxic as HCN at face value. Is it true that HCN will be formed from KCN in the stomachs of most people, but not in people who have low stomach acidity? The pKa of HCN is 9.2, which means that below a pH of 9.2 prussic acid is mostly found as the compound HCN, while above pH 9.2 it mostly exists as the dissociated ions H+ and CN-. The normal pH range of the contents of human stomachs is 1.5 to 3.5, so virtually all the prussic acid in the stomach of a poisoned person will be undissociated. In order to have a significantly lower amount of undissociated HCN, Rasputin's stomach would have needed to have a pH close to 9(!), which strains credulity.
- ...and even if the toxicity of cyanide is strongly pH-dependent, presumably the relevant pH is the pH of the target sites of cyanide's action, i.e. the mitochondria of healthy cells and not the contents of the stomach.
- Finally, there is simply no corroborating evidence to indicate that Rasputin ever had a low stomach acidity.
Brkkp ( talk) 19:18, 13 October 2018 (UTC)
The reference to John B McLemore and 'S-Town' are fictional and do not belong here. — Preceding unsigned comment added by 194.9.244.28 ( talk) 10:50, 8 October 2021 (UTC)
- McLemore was real, and so was his suicide. Here's John McLemore's obituary: https://web.archive.org/web/20170926191033/http://obits.al.com/obituaries/birmingham/obituary.aspx?n=john-b-mclemore&pid=175165387
- I can't speak to the accuracy of the podcast, but no one from the town seems to have contradicted the method of his death. Martin Blank ( talk) 03:45, 29 December 2021 (UTC)
As a lay person I had much difficulty understanding the technical terms in article. It would be helpful if technical jargon was parenthetically defined in lay language. Hyperlinks are fine, but many times the landing article is just as technical as the original one. Scottinkga ( talk) 13:11, 19 August 2023 (UTC)