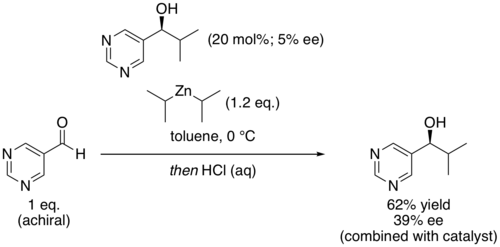
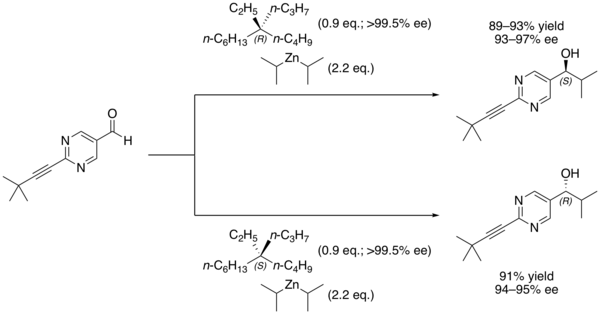
In organic chemistry, the Soai reaction is the alkylation of pyrimidine-5-carbaldehyde with diisopropylzinc. The reaction is autocatalytic and leads to rapidly increasing amounts of the same enantiomer of the product. The product pyrimidyl alcohol is chiral and induces that same chirality in further catalytic cycles. Starting with a low enantiomeric excess ("ee") produces a product with very high enantiomeric excess. [1] The reaction has been studied for clues about the origin of homochirality among certain classes of biomolecules. [2]
The Japanese chemist Kensō Soai (1950–) discovered the reaction in 1995. [3] [4] For his work in "elucidating the origins of chirality and homochirality", Soai received the Chemical Society of Japan award in 2010. [5]
Other chiral additives can be used as the initial source of asymmetric induction, with the major product of that first reaction being rapidly amplified. For example, Soai's group has demonstrated that even chiral quaternary hydrocarbons, which have no clear Lewis basic site for binding the nucleophile, are nonetheless capable of inducing asymmetric catalysis in the reaction. [6]
The chiral induction is believed to occur as a result of interactions between the C–H bonds of the alkane and the pi electrons of the aldehyde. [6]
In another example, Soai and coworkers showed that even [15N](2R, 3S)-bis(dimethylamino)butane, whose chirality results solely due to the difference between 14N and 15N (7% isotopic mass difference), gave 45% ee when used as a stoichiometric ligand. [7]
References
- ^ Flügel RM (January 11, 2011). Chirality and Life: A Short Introduction to the Early Phases of Chemical Evolution. Springer. pp. 16–17. ISBN 978-3-642-16977-9.
- ^ Blackmond DG (April 2004). "Asymmetric autocatalysis and its implications for the origin of homochirality". Proceedings of the National Academy of Sciences of the United States of America. 101 (16): 5732–6. Bibcode: 2004PNAS..101.5732B. doi: 10.1073/pnas.0308363101. PMC 395976. PMID 15067112.
- ^ Soai K, Shibata T, Morioka H, Choji K (1995). "Asymmetric autocatalysis and amplification of enantiomeric excess of a chiral molecule". Nature. 378 (6559): 767–768. Bibcode: 1995Natur.378..767S. doi: 10.1038/378767a0. S2CID 4258847.
- ^ Shibata T, Morioka H, Hayase T, Choji K, Soai K (1996). "Highly Enantioselective Catalytic Asymmetric Automultiplication of Chiral Pyrimidyl Alcohol". Journal of the American Chemical Society. 118 (2): 471–472. doi: 10.1021/ja953066g.
- ^ "CSJ Award 2010: Prof. Kenso Soai". The Chemical Society of Japan. Retrieved 2015-04-14.
- ^ a b Kawasaki T, Tanaka H, Tsutsumi T, Kasahara T, Sato I, Soai K (May 2006). "Chiral discrimination of cryptochiral saturated quaternary and tertiary hydrocarbons by asymmetric autocatalysis". Journal of the American Chemical Society. 128 (18): 6032–3. doi: 10.1021/ja061429e. PMID 16669661.
- ^ Matsumoto A, Ozaki H, Harada S, Tada K, Ayugase T, Ozawa H, et al. (December 2016). "Asymmetric Induction by a Nitrogen 14 N/15 N Isotopomer in Conjunction with Asymmetric Autocatalysis". Angewandte Chemie. 55 (49): 15246–15249. doi: 10.1002/anie.201608955. PMC 5132014. PMID 27754589.
Further reading
- Islas JR, Lavabre D, Grevy JM, Lamoneda RH, Cabrera HR, Micheau JC, Buhse T (September 2005). "Mirror-symmetry breaking in the Soai reaction: a kinetic understanding". Proceedings of the National Academy of Sciences of the United States of America. 102 (39): 13743–8. Bibcode: 2005PNAS..10213743I. doi: 10.1073/pnas.0503171102. PMC 1236534. PMID 16174731.
- Micskei K, Rábai G, Gál E, Caglioti L, Pályi G (July 2008). "Oscillatory symmetry breaking in the Soai reaction". The Journal of Physical Chemistry B. 112 (30): 9196–200. doi: 10.1021/jp803334b. PMID 18593153.
- Caglioti L, Hajdu C, Holczknecht O, Zékány L, Zucchi C, Micskei K, Pályi G (June 2006). "The concept of racemates and the Soai reaction" (PDF). Viva Orig. 34: 62–80. Archived from the original (PDF) on 12 August 2017.
- Podlech J, Gehring T (September 2005). "New aspects of Soai's asymmetric autocatalysis". Angewandte Chemie. 44 (36): 5776–7. doi: 10.1002/anie.200501742. PMID 16078286.