Diablo homolog (DIABLO) is a mitochondrial protein that in humans is encoded by the DIABLO (direct IAP binding protein with low pI) gene on chromosome 12. [5] [6] [7] DIABLO is also referred to as second mitochondria-derived activator of caspases or SMAC. This protein binds inhibitor of apoptosis proteins (IAPs), thus freeing caspases to activate apoptosis. [7] [8] Due to its proapoptotic function, SMAC is implicated in a broad spectrum of tumors, and small molecule SMAC mimetics have been developed to enhance current cancer treatments. [7] [9]
Structure
Protein
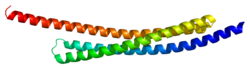
This gene encodes a 130 Å-long, arch-shaped homodimer protein. The full-length protein product spans 239 residues, 55 of which comprise the mitochondrial- targeting sequence (MTS) at its N-terminal. However, once the full-length protein is imported into the mitochondria, this sequence is excised to produce the 184-residue mature protein. [9] [10] [11] This cleavage also exposes four residues at the N-terminal, Ala-Val-Pro-Ile (AVPI), which is the core of the IAP binding domain and crucial for inhibiting XIAP. [9] [10] [11] Specifically, the tetrapeptide sequence binds the BIR3 domain of XIAP to form a stable complex between SMAC and XIAP. [9] [10] [11] The homodimer structure also facilitates SMAC-XIAP binding via the BIR2 domain, though it does not form until the protein is released into the cytoplasm as a result of outer mitochondrial membrane permeabilization. [11] Thus, monomeric SMAC mutants can still bind the BIR3 domain but not the BIR2 domain, which compromises the protein’s inhibitory function. [10] Meanwhile, mutations within the AVPI sequence lead to loss of function, though SMAC may still be able to perform IAP binding-independent functions, such as inducing the ubiquitinylation of XIAP. [10] [12]
Gene
Several alternatively spliced transcript variants that encode distinct isoforms have been described for this gene, but the validity of some transcripts, and their predicted ORFs, has not been determined conclusively. [7] [10] Two known isoforms both lack the MTS and the IAP binding domain, suggesting differential subcellular localization and function. [12]
Function
SMAC is a mitochondrial protein that promotes cytochrome c- and TNF receptor-dependent activation of apoptosis by inhibiting the effect of IAP – a group of proteins that negatively regulate apoptosis, or programmed cell death. [8] [13] SMAC is normally a mitochondrial protein localized to the mitochondrial intermembrane space, but it enters the cytosol when cells undergo apoptosis. [7] [10] [12] [14] Through the intrinsic pathway of apoptosis, BCL-2 proteins like BAK and BAX form a pore in the outer mitochondrial membrane, leading to mitochondrial membrane permeabilization and the release of both cytochrome c and SMAC. [9] [10] While cytochrome c directly activates APAF1 and caspase 9, SMAC binds IAPs, such as XIAP and cIAP proteins, to inhibit their caspase-binding activity and allow for caspase activation of apoptosis. [7] [9] [10] [12] [14] SMAC is ubiquitously expressed in many cell types, implicating it in various biological processes involving apoptosis. [15] Currently, nonapoptotic functions for SMAC remain unclear. [11]
Clinical significance
SMAC is involved in cancer, and its overexpression is linked to increased sensitivity in tumor cells to apoptosis. [7] [13] So far, SMAC overexpression has been observed to oppose cancer progression in head and neck squamous cell carcinoma, hepatocellular carcinoma, Hodgkin lymphoma, breast cancer, glioblastoma, thyroid cancer, renal cell carcinoma, testicular germ cell tumors, colorectal cancer, lung cancer, bladder cancer, endometrioid endometrial cancer, and other sarcomas. [13] [15] [16] However, the exact relationship between SMAC and leukemia and hematological diseases remains controversial. SMAC mimetics monotherapy displays improved cytotoxic effects on leukemic cell lines compared to combined therapy with other drugs, which is commonly more effective in other types of cancers. [17]
Following experimental elucidation of SMAC structure, small-molecule SMAC mimetics have been developed to mimic the tetrapeptide AVPI in the IAP binding domain of SMAC, which is responsible for binding the BIR3 domains in IAPs like XIAP, cIAP1, and cIAP2 to induce apoptosis, and sometimes, necroptosis. [9] [16] Several of the numerous SMAC mimetics designed within the last decade or so are now undergoing clinical trials, including SM-406 by Bai and colleagues and two mimetics by Genentech. These mimetics are also designed to target tumor cells directly through interacting with inflammatory proteins, such as IL-1β, which are commonly produced by solid tumor lesions. [9] Notably, preclinical studies indicate that the use of SMAC mimetics in conjunction with chemotherapy, death receptor ligands and agonists, as well as small molecule targeted drugs enhance the sensitivity of tumor cells to these treatments. [9] [13] [16] In addition to improving the success of tumor elimination, this increased sensitivity can permit smaller doses, thus minimizing side effects while maintaining efficacy. [16] Nonetheless, there still exists the potential for side effects, such as elevated levels of cytokines and chemokines in normal tissues, depending on the cellular environment. [9]
In addition to cancers, mutations in DIABLO is associated with young-adult onset of nonsyndromic deafness-64. [7]
Interactions
Diablo homolog has been shown to interact with:
- cIAP1, [9] [10] [18]
- cIAP2, [9] [10]
- BIRC5, [10] [19]
- LTBR, [20] and
- XIAP. [9] [10] [19] [21] [22] [23] [24]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000184047 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000029433 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ McNeish IA, Bell S, McKay T, Tenev T, Marani M, Lemoine NR (June 2003). "Expression of Smac/DIABLO in ovarian carcinoma cells induces apoptosis via a caspase-9-mediated pathway". Experimental Cell Research. 286 (2): 186–98. doi: 10.1016/S0014-4827(03)00073-9. PMID 12749848.
- ^ Yu J, Wang P, Ming L, Wood MA, Zhang L (June 2007). "SMAC/Diablo mediates the proapoptotic function of PUMA by regulating PUMA-induced mitochondrial events". Oncogene. 26 (29): 4189–98. doi: 10.1038/sj.onc.1210196. PMID 17237824. S2CID 25230886.
- ^ a b c d e f g h "Entrez Gene: DIABLO diablo homolog (Drosophila)".
- ^ a b Vucic D, Deshayes K, Ackerly H, Pisabarro MT, Kadkhodayan S, Fairbrother WJ, Dixit VM (April 2002). "SMAC negatively regulates the anti-apoptotic activity of melanoma inhibitor of apoptosis (ML-IAP)". The Journal of Biological Chemistry. 277 (14): 12275–9. doi: 10.1074/jbc.M112045200. PMID 11801603.
- ^ a b c d e f g h i j k l m Bai L, Smith DC, Wang S (October 2014). "Small-molecule SMAC mimetics as new cancer therapeutics". Pharmacology & Therapeutics. 144 (1): 82–95. doi: 10.1016/j.pharmthera.2014.05.007. PMC 4247261. PMID 24841289.
- ^ a b c d e f g h i j k l m Shi Y (May 2001). "A structural view of mitochondria-mediated apoptosis". Nature Structural Biology. 8 (5): 394–401. doi: 10.1038/87548. PMID 11323712. S2CID 19501646.
- ^ a b c d e Galluzzi L, Joza N, Tasdemir E, Maiuri MC, Hengartner M, Abrams JM, Tavernarakis N, Penninger J, Madeo F, Kroemer G (July 2008). "No death without life: vital functions of apoptotic effectors". Cell Death and Differentiation. 15 (7): 1113–23. doi: 10.1038/cdd.2008.28. PMC 2917777. PMID 18309324.
- ^ a b c d Martinez-Ruiz GU, Victoria-Acosta G, Vazquez-Santillan KI, Jimenez-Hernandez L, Muñoz-Galindo L, Ceballos-Cancino G, Maldonado V, Melendez-Zajgla J (2014). "Ectopic expression of new alternative splice variant of Smac/DIABLO increases mammospheres formation". International Journal of Clinical and Experimental Pathology. 7 (9): 5515–26. PMC 4203164. PMID 25337193.
- ^ a b c d Zeng H, Zhang S, Yang KY, Wang T, Hu JL, Huang LL, Wu G (December 2010). "Knockdown of second mitochondria-derived activator of caspase expression by RNAi enhances growth and cisplatin resistance of human lung cancer cells". Cancer Biotherapy & Radiopharmaceuticals. 25 (6): 705–12. doi: 10.1089/cbr.2010.0786. PMID 21204765.
- ^ a b Anguiano-Hernandez YM, Chartier A, Huerta S (July 2007). "Smac/DIABLO and colon cancer". Anti-Cancer Agents in Medicinal Chemistry. 7 (4): 467–73. doi: 10.2174/187152007781058631. PMID 17630921.
- ^ a b Dobrzycka B, Terlikowski SJ, Bernaczyk PS, Garbowicz M, Niklinski J, Chyczewski L, Kulikowski M (December 2010). "Prognostic significance of smac/DIABLO in endometrioid endometrial cancer". Folia Histochemica et Cytobiologica / Polish Academy of Sciences, Polish Histochemical and Cytochemical Society. 48 (4): 678–81. doi: 10.2478/v10042-010-0091-2. PMID 21478115.
- ^ a b c d Sun Q, Zheng X, Zhang L, Yu J (April 2011). "Smac modulates chemosensitivity in head and neck cancer cells through the mitochondrial apoptotic pathway". Clinical Cancer Research. 17 (8): 2361–72. doi: 10.1158/1078-0432.CCR-10-2262. PMC 3079009. PMID 21242120.
- ^ Scavullo C, Servida F, Lecis D, Onida F, Drago C, Ferrante L, Seneci P, Barcellini W, Lionetti M, Todoerti K, Neri A, Delia D, Deliliers GL (July 2013). "Single-agent Smac-mimetic compounds induce apoptosis in B chronic lymphocytic leukaemia (B-CLL)". Leukemia Research. 37 (7): 809–15. doi: 10.1016/j.leukres.2013.03.016. PMID 23618690.
- ^ Hegde R, Srinivasula SM, Datta P, Madesh M, Wassell R, Zhang Z, Cheong N, Nejmeh J, Fernandes-Alnemri T, Hoshino S, Alnemri ES (October 2003). "The polypeptide chain-releasing factor GSPT1/eRF3 is proteolytically processed into an IAP-binding protein". The Journal of Biological Chemistry. 278 (40): 38699–706. doi: 10.1074/jbc.M303179200. PMID 12865429.
- ^ a b Song Z, Yao X, Wu M (June 2003). "Direct interaction between survivin and Smac/DIABLO is essential for the anti-apoptotic activity of survivin during taxol-induced apoptosis". The Journal of Biological Chemistry. 278 (25): 23130–40. doi: 10.1074/jbc.M300957200. PMID 12660240.
- ^ Kuai J, Nickbarg E, Wooters J, Qiu Y, Wang J, Lin LL (April 2003). "Endogenous association of TRAF2, TRAF3, cIAP1, and Smac with lymphotoxin beta receptor reveals a novel mechanism of apoptosis". The Journal of Biological Chemistry. 278 (16): 14363–9. doi: 10.1074/jbc.M208672200. PMID 12571250.
- ^ Verhagen AM, Ekert PG, Pakusch M, Silke J, Connolly LM, Reid GE, Moritz RL, Simpson RJ, Vaux DL (July 2000). "Identification of DIABLO, a mammalian protein that promotes apoptosis by binding to and antagonizing IAP proteins". Cell. 102 (1): 43–53. doi: 10.1016/s0092-8674(00)00009-x. PMID 10929712. S2CID 3192775.
- ^ Hunter AM, Kottachchi D, Lewis J, Duckett CS, Korneluk RG, Liston P (February 2003). "A novel ubiquitin fusion system bypasses the mitochondria and generates biologically active Smac/DIABLO". The Journal of Biological Chemistry. 278 (9): 7494–9. doi: 10.1074/jbc.C200695200. PMID 12511567.
- ^ Davoodi J, Lin L, Kelly J, Liston P, MacKenzie AE (September 2004). "Neuronal apoptosis-inhibitory protein does not interact with Smac and requires ATP to bind caspase-9". The Journal of Biological Chemistry. 279 (39): 40622–8. doi: 10.1074/jbc.M405963200. PMID 15280366.
- ^ Verhagen AM, Silke J, Ekert PG, Pakusch M, Kaufmann H, Connolly LM, Day CL, Tikoo A, Burke R, Wrobel C, Moritz RL, Simpson RJ, Vaux DL (Jan 2002). "HtrA2 promotes cell death through its serine protease activity and its ability to antagonize inhibitor of apoptosis proteins". The Journal of Biological Chemistry. 277 (1): 445–54. doi: 10.1074/jbc.M109891200. PMID 11604410.
Further reading
- Shi Y (2001). "A structural view of mitochondria-mediated apoptosis". Nat. Struct. Biol. 8 (5): 394–401. doi: 10.1038/87548. PMID 11323712. S2CID 19501646.
- Anguiano-Hernandez YM, Chartier A, Huerta S (2007). "Smac/DIABLO and colon cancer". Anti-Cancer Agents in Medicinal Chemistry. 7 (4): 467–73. doi: 10.2174/187152007781058631. PMID 17630921.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi: 10.1016/0378-1119(94)90802-8. PMID 8125298.
- Bonaldo MF, Lennon G, Soares MB (1997). "Normalization and subtraction: two approaches to facilitate gene discovery". Genome Res. 6 (9): 791–806. doi: 10.1101/gr.6.9.791. PMID 8889548.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi: 10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Du C, Fang M, Li Y, Li L, Wang X (2000). "Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition". Cell. 102 (1): 33–42. doi: 10.1016/S0092-8674(00)00008-8. PMID 10929711. S2CID 11268259.
- Verhagen AM, Ekert PG, Pakusch M, Silke J, Connolly LM, Reid GE, Moritz RL, Simpson RJ, Vaux DL (2000). "Identification of DIABLO, a mammalian protein that promotes apoptosis by binding to and antagonizing IAP proteins". Cell. 102 (1): 43–53. doi: 10.1016/S0092-8674(00)00009-X. PMID 10929712. S2CID 3192775.
- Srinivasula SM, Datta P, Fan XJ, Fernandes-Alnemri T, Huang Z, Alnemri ES (2000). "Molecular determinants of the caspase-promoting activity of Smac/DIABLO and its role in the death receptor pathway". J. Biol. Chem. 275 (46): 36152–7. doi: 10.1074/jbc.C000533200. PMID 10950947.
- Chai J, Du C, Wu JW, Kyin S, Wang X, Shi Y (2000). "Structural and biochemical basis of apoptotic activation by Smac/DIABLO". Nature. 406 (6798): 855–62. Bibcode: 2000Natur.406..855C. doi: 10.1038/35022514. PMID 10972280. S2CID 4385614.
- Liu Z, Sun C, Olejniczak ET, Meadows RP, Betz SF, Oost T, Herrmann J, Wu JC, Fesik SW (2001). "Structural basis for binding of Smac/DIABLO to the XIAP BIR3 domain". Nature. 408 (6815): 1004–8. doi: 10.1038/35050006. PMID 11140637. S2CID 4397833.
- Wu G, Chai J, Suber TL, Wu JW, Du C, Wang X, Shi Y (2001). "Structural basis of IAP recognition by Smac/DIABLO". Nature. 408 (6815): 1008–12. doi: 10.1038/35050012. PMID 11140638. S2CID 4422796.
- Srinivasula SM, Hegde R, Saleh A, Datta P, Shiozaki E, Chai J, Lee RA, Robbins PD, Fernandes-Alnemri T, Shi Y, Alnemri ES (2001). "A conserved XIAP-interaction motif in caspase-9 and Smac/DIABLO regulates caspase activity and apoptosis". Nature. 410 (6824): 112–6. Bibcode: 2001Natur.410..112S. doi: 10.1038/35065125. PMID 11242052. S2CID 4380050.
- Huang Y, Park YC, Rich RL, Segal D, Myszka DG, Wu H (2001). "Structural basis of caspase inhibition by XIAP: differential roles of the linker versus the BIR domain". Cell. 104 (5): 781–90. doi: 10.1016/S0092-8674(01)00273-2. PMID 11257231. S2CID 14019346.
- Roberts DL, Merrison W, MacFarlane M, Cohen GM (2001). "The Inhibitor of Apoptosis Protein-Binding Domain of Smac Is Not Essential for Its Proapoptotic Activity". J. Cell Biol. 153 (1): 221–8. doi: 10.1083/jcb.153.1.221. PMC 2185525. PMID 11285287.
- Verhagen AM, Silke J, Ekert PG, Pakusch M, Kaufmann H, Connolly LM, Day CL, Tikoo A, Burke R, Wrobel C, Moritz RL, Simpson RJ, Vaux DL (2002). "HtrA2 promotes cell death through its serine protease activity and its ability to antagonize inhibitor of apoptosis proteins". J. Biol. Chem. 277 (1): 445–54. doi: 10.1074/jbc.M109891200. PMID 11604410.
- Adrain C, Creagh EM, Martin SJ (2002). "Apoptosis-associated release of Smac/DIABLO from mitochondria requires active caspases and is blocked by Bcl-2". EMBO J. 20 (23): 6627–36. doi: 10.1093/emboj/20.23.6627. PMC 125329. PMID 11726499.
- Sun XM, Bratton SB, Butterworth M, MacFarlane M, Cohen GM (2002). "Bcl-2 and Bcl-xL inhibit CD95-mediated apoptosis by preventing mitochondrial release of Smac/DIABLO and subsequent inactivation of X-linked inhibitor-of-apoptosis protein". J. Biol. Chem. 277 (13): 11345–51. doi: 10.1074/jbc.M109893200. PMID 11801595.