| |
| Names | |
|---|---|
|
IUPAC name
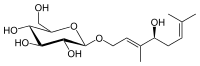
(2R,3R,4S,5S,6R)-2-{[(2E,4S)-4-Hydroxy-3,7-dimethylocta-2,6-dien-1-yl]oxy}-6-(hydroxymethyl)oxane-3,4,5-triol
| |
| Identifiers | |
3D model (
JSmol)
|
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| Properties | |
| C16H28O7 | |
| Molar mass | 332.393 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Rosiridin is a chemical compound that has been isolated from Rhodiola sachalinensis. [1] Rosiridin can inhibit monoamine oxidases A and B, possibly meaning that the compound could help in the treatment of depression and senile dementia. [2] [3]
References
- ^ Yoshikawa, Masayuki; Nakamura, Seikou; Li, Xuezheng; Matsuda, Hisashi (2008). "Reinvestigation of absolute stereostructure of (−)-rosiridol: Structures of monoterpene glycosides, rosiridin, rosiridosides A, B, and C, from Rhodiola sachalinesnsis". Chemical and Pharmaceutical Bulletin. 56 (5): 695–700. doi: 10.1248/cpb.56.695. PMID 18451561.
- ^ Panossian, Alexander; Wikman, Georg (2010). "Effects of adaptogens on the central nervous system and the molecular mechanisms associated with their stress-protective activity". Pharmaceuticals. 3 (1): 188–224. doi: 10.3390/ph3010188. PMC 3991026. PMID 27713248.
- ^ van Diermen, Daphne; Marston, Andrew; Bravo, Juan; Reist, Marianne; Carrupt, Pierre-Alain; Hostettmann, Kurt (2009-03-18). "Monoamine oxidase inhibition by Rhodiola rosea L. roots". Journal of Ethnopharmacology. 122 (2): 397–401. doi: 10.1016/j.jep.2009.01.007. ISSN 0378-8741.