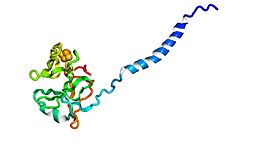
 Rieske protein from
cytochrome b6f complex. (
PDB:
1vf5) | |||||||||
| Identifiers | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Symbol | Rieske | ||||||||
| Pfam | PF00355 | ||||||||
| InterPro | IPR005806 | ||||||||
| PROSITE | PDOC00177 | ||||||||
| SCOP2 | 1rie / SCOPe / SUPFAM | ||||||||
| TCDB | 3.E.2 | ||||||||
| OPM superfamily | 92 | ||||||||
| OPM protein | 1q90 | ||||||||
| CDD | cd03467 | ||||||||
| |||||||||
| Cytochrome B6-F complex Fe-S subunit, alpha helical transmembrane domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
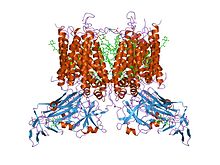 crystal structure of cytochrome b6f complex from m.laminosus | |||||||||
| Identifiers | |||||||||
| Symbol | CytB6-F_Fe-S | ||||||||
| Pfam | PF08802 | ||||||||
| InterPro | IPR014909 | ||||||||
| |||||||||
Rieske proteins are
iron–sulfur protein (ISP) components of
cytochrome bc1 complexes and
cytochrome b6f complexes and are responsible for electron transfer in some biological systems. John S. Rieske and co-workers first discovered the protein and in 1964 isolated an acetylated form of the bovine
mitochondrial protein.
[1] In 1979, Trumpower's team isolated the "oxidation factor" from bovine
mitochondria and showed it was a reconstitutively-active form of the Rieske iron-sulfur protein
[2]
It is a unique [2Fe-2S] cluster in that one of the two Fe atoms is coordinated by two
histidine residues rather than two
cysteine residues. They have since been found in plants, animals, and
bacteria with widely ranging electron
reduction potentials from -150 to +400 mV.
[3]
Biological function
Ubiquinol-cytochrome-c reductase (also known as bc1 complex or complex III) is an enzyme complex of bacterial and mitochondrial oxidative phosphorylation systems. It catalyses the oxidation-reduction reaction of the mobile components ubiquinol and cytochrome c, contributing to an electrochemical potential difference across the mitochondrial inner or bacterial membrane, which is linked to ATP synthesis. [4] [5]
The complex consists of three subunits in most bacteria, and nine in mitochondria: both bacterial and mitochondrial complexes contain cytochrome b and cytochrome c1 subunits, and an iron–sulfur 'Rieske' subunit, which contains a high potential 2Fe-2S cluster. [6] The mitochondrial form also includes six other subunits that do not possess redox centres. Plastoquinone- plastocyanin reductase (b6f complex), present in cyanobacteria and the chloroplasts of plants, catalyses the oxidoreduction of plastoquinol and cytochrome f. This complex, which is functionally similar to ubiquinol-cytochrome c reductase, comprises cytochrome b6, cytochrome f and Rieske subunits. [7]
The Rieske subunit acts by binding either a ubiquinol or plastoquinol anion, transferring an electron to the 2Fe-2S cluster, then releasing the electron to the cytochrome c or cytochrome f heme iron. [4] [7] The reduction of the Rieske center increases the affinity of the subunit by several orders of magnitude, stabilizing the semiquinone radical at the Q(P) site. [8] The Rieske domain has a [2Fe-2S] center. Two conserved cysteines coordinate one Fe ion while the other Fe ion is coordinated by two conserved histidines. The 2Fe-2S cluster is bound in the highly conserved C-terminal region of the Rieske subunit.
Rieske protein family
The homologues of the Rieske proteins include ISP components of cytochrome b6f complex, aromatic-ring-hydroxylating dioxygenases (phthalate dioxygenase, benzene, naphthalene and toluene 1,2-dioxygenases) and arsenite oxidase ( EC 1.20.98.1). Comparison of amino acid sequences has revealed the following consensus sequence:
- Cys-Xaa-His-(Xaa)15–17-Cys-Xaa-Xaa-His

3D structure
The crystal structures of a number of Rieske proteins are known. The overall fold, comprising two subdomains, is dominated by antiparallel β-structure and contains variable numbers of α-helices. The smaller "cluster-binding" subdomains in mitochondrial and chloroplast proteins are virtually identical, whereas the large subdomains are substantially different in spite of a common folding topology. The [Fe2S2] cluster-binding subdomains have the topology of an incomplete antiparallel β-barrel. One iron atom of the Rieske [Fe2S2] cluster in the domain is coordinated by two cysteine residues and the other is coordinated by two histidine residues through the Nδ atoms. The ligands coordinating the cluster originate from two loops; each loop contributes one Cys and one His.
Subfamilies
- Rieske iron–sulfur protein, C-terminal InterPro: IPR005805
- Arsenite oxidase, small subunit InterPro: IPR014067
Human proteins containing this domain
References
- ^ Rieske JS, Maclennan DH, Coleman R (1964). "Isolation and properties of an iron-protein from the (reduced coenzyme Q)-cytochrome C reductase complex of the respiratory chain". Biochem. Biophys. Res. Commun. 15 (4): 338–344. doi: 10.1016/0006-291X(64)90171-8.
- ^ Trumpower BL, Edwards CA (1979). "Purification of a reconstitutively active iron-sulfur protein (oxidation factor) from succinate:cytochrome c reductase complex of bovine heart mitochondria". J Biol Chem. 254 (17): 8697–706. doi: 10.1016/S0021-9258(19)86947-8. PMID 224062.
-
^ Brown, E.N. and Friemann, R. and Karlsson, A. and Parales, J.V. and Couture, M.M. and Eltis, L.D. and Ramaswamy, S. (2008). "Determining Rieske cluster reduction potentials". J. Biol. Inorg. Chem. 13 (8): 1301–1313.
doi:
10.1007/s00775-008-0413-4.
PMID
18719951.
S2CID
3303144.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ a b Harnisch U, Weiss H, Sebald W (May 1985). "The primary structure of the iron-sulfur subunit of ubiquinol-cytochrome c reductase from Neurospora, determined by cDNA and gene sequencing". Eur. J. Biochem. 149 (1): 95–9. doi: 10.1111/j.1432-1033.1985.tb08898.x. PMID 2986972.
- ^ Gabellini N, Sebald W (February 1986). "Nucleotide sequence and transcription of the fbc operon from Rhodopseudomonas sphaeroides. Evaluation of the deduced amino acid sequences of the FeS protein, cytochrome b and cytochrome c1". Eur. J. Biochem. 154 (3): 569–79. doi: 10.1111/j.1432-1033.1986.tb09437.x. PMID 3004982.
- ^ Kurowski B, Ludwig B (October 1987). "The genes of the Paracoccus denitrificans bc1 complex. Nucleotide sequence and homologies between bacterial and mitochondrial subunits". J. Biol. Chem. 262 (28): 13805–11. doi: 10.1016/S0021-9258(19)76497-7. PMID 2820981.
- ^ a b Madueño F, Napier JA, Cejudo FJ, Gray JC (October 1992). "Import and processing of the precursor of the Rieske FeS protein of tobacco chloroplasts". Plant Mol. Biol. 20 (2): 289–99. doi: 10.1007/BF00014496. PMID 1391772. S2CID 2306978.
- ^ Link TA (July 1997). "The role of the 'Rieske' iron sulfur protein in the hydroquinone oxidation (Q(P)) site of the cytochrome bc1 complex. The 'proton-gated affinity change' mechanism". FEBS Lett. 412 (2): 257–64. doi: 10.1016/S0014-5793(97)00772-2. PMID 9256231. S2CID 35375512.
Further reading
- Iwata S, Saynovits M, Link TA, Michel H (May 1996). "Structure of a water soluble fragment of the 'Rieske' iron-sulfur protein of the bovine heart mitochondrial cytochrome bc1 complex determined by MAD phasing at 1.5 A resolution". Structure. 4 (5): 567–79. doi: 10.1016/S0969-2126(96)00062-7. PMID 8736555.
- Huang JT, Struck F, Matzinger DF, Levings CS (December 1991). "Functional analysis in yeast of cDNA coding for the mitochondrial Rieske iron-sulfur protein of higher plants". Proc. Natl. Acad. Sci. U.S.A. 88 (23): 10716–20. Bibcode: 1991PNAS...8810716H. doi: 10.1073/pnas.88.23.10716. PMC 53001. PMID 1961737.
- Brandt U, Yu L, Yu CA, Trumpower BL (April 1993). "The mitochondrial targeting presequence of the Rieske iron-sulfur protein is processed in a single step after insertion into the cytochrome bc1 complex in mammals and retained as a subunit in the complex". J. Biol. Chem. 268 (12): 8387–90. doi: 10.1016/S0021-9258(18)52883-0. PMID 8386158.
- Ferraro, D.J., Gakhar, L. and Ramaswamy, S. (2005). "Rieske business: structure-function of Rieske non-heme oxygenases". Biochem. Biophys. Res. Commun. 338 (1): 175–190.
doi:
10.1016/j.bbrc.2005.08.222.
PMID
16168954.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - Mason, J.R. & Cammack, R. (1992). "The electron-transport proteins of hydroxylating bacterial dioxygenases". Annu. Rev. Microbiol. 46: 277–305. doi: 10.1146/annurev.mi.46.100192.001425. PMID 1444257.
- Schmidt, C.L. (2004). "Rieske iron-sulfur proteins from extremophilic organisms". J. Bioenerg. Biomembr. 36 (1): 107–113. doi: 10.1023/B:JOBB.0000019602.96578.78. PMID 15168614. S2CID 23790442.
- Schneider, D. & Schmidt, C.L. (2005). "Multiple Rieske proteins in prokaryotes: where and why?". Biochim. Biophys. Acta. 1710 (1): 1–12. doi: 10.1016/j.bbabio.2005.09.003. PMID 16271700.
- Brown, E.N. and Friemann, R. and Karlsson, A. and Parales, J.V. and Couture, M.M. and Eltis, L.D. and Ramaswamy, S. (2008). "Determining Rieske cluster reduction potentials". J. Biol. Inorg. Chem. 13 (8): 1301–1313.
doi:
10.1007/s00775-008-0413-4.
PMID
18719951.
S2CID
3303144.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)
External links
- PDB: 1RIE - X-ray structure of Rieske protein (water-soluble fragment) of the bovine mitochondrial cytochrome bc1 complex
- PDB: 1RFS - X-ray structure of Rieske protein (water-soluble fragment) of the spinach chloroplast cytochrome b6 fcomplex
- PDB: 1FQT - X-ray structure of Rieske-type ferredoxin associated with biphenyl dioxygenase from Burkholderia cepacia
- PDB: 1G8J - X-ray structure of Rieske subunit of arsenite oxidase from Alcaligenes faecalis
- PDB: 2I7F - X-ray structure of the Sphingomonas yanoikuyae B1 Rieske ferredoxin
- PDB: 2QPZ - X-ray structure of the Pseudomonas Naphthalene 1,2-dioxygenase Rieske ferredoxin
- InterPro: IPR005806 - InterPro entry for Rieske [2Fe-2S] region