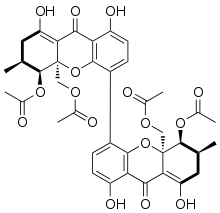
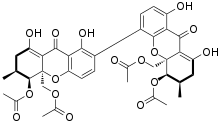

The phomoxanthones are a loosely defined class of natural products. The two founding members of this class are phomoxanthone A and phomoxanthone B. Other compounds were later also classified as phomoxanthones, although a unifying nomenclature has not yet been established. [1] The structure of all phomoxanthones is derived from a dimer of two covalently linked tetrahydroxanthones, and they differ mainly in the position of this link as well as in the acetylation status of their hydroxy groups. The phomoxanthones are structurally closely related to other tetrahydroxanthone dimers such as the secalonic acids and the eumitrins. While most phomoxanthones were discovered in fungi of the genus Phomopsis, most notably in the species Phomopsis longicolla, some have also been found in Penicillium sp. [2]
- Dicerandrol A [3] [2] [4]
- Dicerandrol B [3] [2] [4] [5]
- Dicerandrol C [3] [2] [4] [5]
- Penexanthone A [2] [4]
- Phomolactonexanthone A [4]
- Phomolactonexanthone B [4]
-
Phomoxanthone A
[1]
[6]
[5] and its derivatives:
- 1-acetylphomoxanthone A [7]
- 1,1′-diacetylphomoxanthone A [7]
- 1,1′,8-triacetylphomoxanthone A [7]
- 1,1′,8,8′-tetraacetylphomoxanthone A [7]
- 12-deacetylphomoxanthone A [5] [8] [7]
- 12,12′-dideacetylphomoxanthone A (deacetylphomoxanthone C) [4] [7]
- 12,12′,13,13′-tetradeacetylphomoxanthone A (deacetylphomoxanthone A) [1] [5] [7]
- Phomoxanthone B [1] and its derivatives:
- Masters, Kye-Simeon; Bräse, Stefan (2012). "Xanthones from Fungi, Lichens, and Bacteria: The Natural Products and Their Synthesis". Chemical Reviews. 112 (7): 3717–76. doi: 10.1021/cr100446h. PMID 22617028.
- Wezeman, Tim; Bräse, Stefan; Masters, Kye-Simeon (2015). "Xanthone dimers: A compound family which is both common and privileged". Natural Product Reports. 32 (1): 6–28. doi: 10.1039/C4NP00050A. PMID 25226564.
- ^ a b c d Isaka, M; Jaturapat, A; Rukseree, K; Danwisetkanjana, K; Tanticharoen, M; Thebtaranonth, Y (2001). "Phomoxanthones a and B, novel xanthone dimers from the endophytic fungus Phomopsis species". Journal of Natural Products. 64 (8): 1015–8. doi: 10.1021/np010006h. PMID 11520217.
- ^ a b c d e Cao, Shugeng; McMillin, Douglas W; Tamayo, Giselle; Delmore, Jake; Mitsiades, Constantine S; Clardy, Jon (2012). "Inhibition of Tumor Cells Interacting with Stromal Cells by Xanthones Isolated from a Costa Rican Penicillium sp". Journal of Natural Products. 75 (4): 793–7. doi: 10.1021/np2009863. PMC 3338863. PMID 22458669.
- ^ a b c Wagenaar, Melissa M; Clardy, Jon (2001). "Dicerandrols, New Antibiotic and Cytotoxic Dimers Produced by the Fungus Phomopsis longicolla Isolated from an Endangered Mint". Journal of Natural Products. 64 (8): 1006–9. doi: 10.1021/np010020u. PMID 11520215.
- ^ a b c d e f g h Ding, Bo; Yuan, Jie; Huang, Xishan; Wen, Weitao; Zhu, Xu; Liu, Yayue; Li, Hanxiang; Lu, Yongjun; He, Lei; Tan, Hongmei; She, Zhigang (2013). "New Dimeric Members of the Phomoxanthone Family: Phomolactonexanthones A, B and Deacetylphomoxanthone C Isolated from the Fungus Phomopsis sp". Marine Drugs. 11 (12): 4961–72. doi: 10.3390/md11124961. PMC 3877896. PMID 24335522.
- ^ a b c d e Rönsberg, David; Debbab, Abdessamad; Mándi, Attila; Vasylyeva, Vera; Böhler, Philip; Stork, Björn; Engelke, Laura; Hamacher, Alexandra; Sawadogo, Richard; Diederich, Marc; Wray, Victor; Lin, Wenhan; Kassack, Matthias U; Janiak, Christoph; Scheu, Stefanie; Wesselborg, Sebastian; Kurtán, Tibor; Aly, Amal H; Proksch, Peter (2013). "Pro-Apoptotic and Immunostimulatory Tetrahydroxanthone Dimers from the Endophytic Fungus Phomopsis longicolla". The Journal of Organic Chemistry. 78 (24): 12409–25. doi: 10.1021/jo402066b. PMID 24295452.
- ^ Elsässer, Brigitta; Krohn, Karsten; Flörke, Ulrich; Root, Natalia; Aust, Hans-Jürgen; Draeger, Siegfried; Schulz, Barbara; Antus, Sándor; Kurtán, Tibor (2005). "X-ray Structure Determination, Absolute Configuration and Biological Activity of Phomoxanthone A". European Journal of Organic Chemistry. 2005 (21): 4563. doi: 10.1002/ejoc.200500265.
- ^ a b c d e f g Frank, M; Niemann, H; Böhler, P; Stork, B; Wesselborg, S; Lin, W; Proksch, P (2015). "Phomoxanthone A--From Mangrove Forests to Anticancer Therapy". Current Medicinal Chemistry. 22 (30): 3523–32. doi: 10.2174/0929867322666150716115300. PMID 26179997.
- ^ Shiono, Y; Sasaki, T; Shibuya, F; Yasuda, Y; Koseki, T; Supratman, U (2013). "Isolation of a phomoxanthone a derivative, a new metabolite of tetrahydroxanthone, from a Phomopsis sp. Isolated from the mangrove, Rhizhopora mucronata". Natural Product Communications. 8 (12): 1735–7. doi: 10.1177/1934578X1300801220. PMID 24555286.
- ^ Choi, Jung Nam; Kim, Jiyoung; Ponnusamy, Kannan; Lim, Chaesung; Kim, Jeong Gu; Muthaiya, Maria John; Lee, Choong Hwan (2012). "Identification of a new phomoxanthone antibiotic from Phomopsis longicolla and its antimicrobial correlation with other metabolites during fermentation". The Journal of Antibiotics. 66 (4): 231–3. doi: 10.1038/ja.2012.105. PMID 23211934.
- ^ Rukachaisirikul, Vatcharin; Sommart, Ubonta; Phongpaichit, Souwalak; Sakayaroj, Jariya; Kirtikara, Kanyawim (2008). "Metabolites from the endophytic fungus Phomopsis sp. PSU-D15". Phytochemistry. 69 (3): 783–7. doi: 10.1016/j.phytochem.2007.09.006. PMID 17950385.