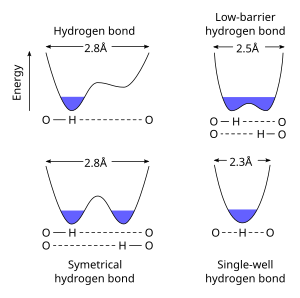
A Low-barrier hydrogen bond (LBHB) is a special type of hydrogen bond. LBHBs can occur when the pKa of the two heteroatoms are closely matched, which allows the hydrogen to be more equally shared between them. This hydrogen-sharing causes the formation of especially short, strong hydrogen bonds. [1]
Description
Standard hydrogen bonds are longer (e.g. 2.8 Å for an O···O h-bond), and the hydrogen ion clearly belongs to one of the heteroatoms. When pKa of the heteroatoms is closely matched, a LBHB becomes possible at a shorter distance (~2.55 Å). When the distance further decreases (< 2.29 Å) the bond is characterized as a single-well or short-strong hydrogen bond. [3]
Proteins
Low barrier hydrogen bonds occur in the water-excluding environments of proteins. [4] Multiple residues act together in a charge-relay system to control the pKa values of the residues involved. LBHBs also occur on the surfaces of proteins, but are unstable due to their proximity to bulk water, and the conflicting requirements of strong salt-bridges in protein-protein interfaces. [4]
Enzyme catalysis
Low-barrier hydrogen bonds have been proposed to be relevant to enzyme catalysis in two types of circumstance. [5] Firstly, a low-barrier hydrogen bond in a charge relay network within an active site could activate a catalytic residue (e.g. between acid and base within a catalytic triad). Secondly, an LBHB could form during catalysis to stabilise a transition state (e.g. with substrate transition state in an oxyanion hole). Both of these mechanisms are contentious, with theoretical and experimental evidence split on whether they occur. [6] [7] Since the 2000s, the general consensus has been that LBHBs are not used by enzymes to aid catalysis. [7] [8] However, in 2012, a low-barrier hydrogen bond has been proposed to be involved in phosphate-arsenate discrimination for a phosphate transport protein. [9] This finding might indicate the possibility of low-barrier hydrogen bonds playing a catalytic role in ion size selection for some very rare cases.
References
- ^ Gilli, G.; Gilli, P. (2000-09-26). "Towards an unified hydrogen-bond theory". Journal of Molecular Structure. 552 (1–3): 1–15. Bibcode: 2000JMoSt.552....1G. doi: 10.1016/S0022-2860(00)00454-3.
- ^ Day, Victor W.; Hossain, Md. Alamgir; Kang, Sung Ok; Powell, Douglas; Lushington, Gerald; Bowman-James, Kristin (2007). "Encircled Proton". J. Am. Chem. Soc. 129 (28): 8692–3. doi: 10.1021/ja0724745. PMID 17585768.
- ^ Schiøtt B, Iversen BB, Madsen GK, Larsen FK, Bruice TC (October 1998). "On the electronic nature of low-barrier hydrogen bonds in enzymatic reactions". Proc. Natl. Acad. Sci. U.S.A. 95 (22): 12799–802. Bibcode: 1998PNAS...9512799S. doi: 10.1073/pnas.95.22.12799. PMC 23598. PMID 9788994.
- ^ a b Ishikita, Hiroshi; Saito, Keisuke (2014-02-06). "Proton transfer reactions and hydrogen-bond networks in protein environments". Journal of the Royal Society Interface. 11 (91): 20130518. doi: 10.1098/rsif.2013.0518. ISSN 1742-5689. PMC 3869154. PMID 24284891.
- ^ Cleland, W. W.; Frey, P. A.; Gerlt, J. A. (2 October 1998). "The Low Barrier Hydrogen Bond in Enzymatic Catalysis". Journal of Biological Chemistry. 273 (40): 25529–25532. doi: 10.1074/jbc.273.40.25529. PMID 9748211.
- ^ Ash, E. L. (7 November 1997). "A Low-Barrier Hydrogen Bond in the Catalytic Triad of Serine Proteases? Theory Versus Experiment". Science. 278 (5340): 1128–1132. Bibcode: 1997Sci...278.1128A. doi: 10.1126/science.278.5340.1128. PMID 9353195.
- ^ a b Schutz, Claudia N.; Warshel, Arieh (1 April 2004). "The low barrier hydrogen bond (LBHB) proposal revisited: The case of the Asp ··· His pair in serine proteases". Proteins: Structure, Function, and Bioinformatics. 55 (3): 711–723. doi: 10.1002/prot.20096. PMID 15103633. S2CID 34229297.
- ^ Warshel, Arieh; Sharma, Pankaz K.; Kato, Mitsunori; Xiang, Yun; Liu, Hanbin; Olsson, Mats H. M. (August 2006). "Electrostatic Basis for Enzyme Catalysis". Chemical Reviews. 106 (8): 3210–3235. doi: 10.1021/cr0503106. PMID 16895325.
- ^ Elias, Mikael; Wellner, Alon; Goldin-Azulay, Korina; Chabriere, Eric; Vorholt, Julia A.; Erb, Tobias J.; Tawfik, Dan S. (2012-11-01). "The molecular basis of phosphate discrimination in arsenate-rich environments". Nature. 491 (7422): 134–137. Bibcode: 2012Natur.491..134E. doi: 10.1038/nature11517. ISSN 0028-0836. PMID 23034649. S2CID 205230854.