Lawrence Que, Jr. | |
|---|---|
| Alma mater |
Ateneo de Manila University B.S. (1969) University of Minnesota Ph.D. (1973) |
| Known for | First isolation of a nonheme Fe(IV)=O complex |
| Awards | ACS Award in Inorganic Chemistry (2017) |
| Scientific career | |
| Fields | Bioinorganic Chemistry |
| Institutions |
University of Minnesota Cornell University |
| Thesis | Nuclear Magnetic Resonance (NMR) Studies of Stereochemically Nonrigid Complexes (1973) |
| Doctoral advisor | Louis H. Pignolet |
| Other academic advisors | Richard H. Holm, Eckhard Münck |
| Doctoral students | Sheila David |
| Website | Link |
Lawrence Que Jr. is a chemist who specializes in bioinorganic chemistry and is a Regents Professor at the University of Minnesota, Twin Cities. [1] He received the 2017 American Chemical Society (ACS) Award in Inorganic Chemistry for his contributions to the field., [2] and the 2008 ACS Alfred Bader Award in Bioinorganic Chemistry. [3]
Biography
Lawrence Que Jr. obtained his B.S. degree in chemistry from Ateneo de Manila University in Quezon City, Philippines in 1969, [4] [5] then received his Ph.D. degree in chemistry from the University of Minnesota in 1973 under the direction of Prof. Louis H. Pignolet. [5] [6] With Prof. Pignolet, Que studied stereochemical non-rigidity in coordination complexes with proton NMR spectroscopy. [7] [8] [9]
Que conducted postdoctoral studies with Prof. Richard H. Holm at the Massachusetts Institute of Technology from 1973 to 1974, where he studied iron-sulfur clusters in proteins [10] and the synthesis of model clusters. [11] [12] He then was a postdoctoral fellow with Prof. Eckard Münck at the Gray Freshwater Biological Institute of the University of Minnesota from 1975 to 1977. [5] [13] [14] With Prof. Münck, Que studied the mechanism of the protocatechuate 3,4-dioxygenase enzyme, using Mössbauer and EPR spectroscopies, as well as inhibition studies. [15] [16]
Que began his independent research career at Cornell University as an Assistant Professor of Chemistry in 1977. [4] He then moved back to the University of Minnesota in 1983 where he currently is a Regents Professor. [5] [14]
Que has published over 450 research manuscripts and 7 patents. He has presented almost 300 invited lectures and mentored almost 50 doctoral students. His inorganic chemistry research group at the University of Minnesota focuses on iron chemistry relevant to biocatalysis, in an attempt to better understand oxygen activation mechanisms of nonheme iron enzymes. His group is also working towards designing functional models for iron enzymes and capturing, observing, and categorizing highly active metal-based intermediates. He is also working on creating bio-inspired oxidation catalysts for green chemistry applications. For his contributions to the field of inorganic and bioinorganic chemistry, Que received the American Chemical Society's 2008 Alfred Bader Award in Bioinorganic Chemistry and 2017 Award in Inorganic Chemistry.
Research
Bioinspired catalysis
Que has been studying the behaviors of high-valent iron-oxo species in relation to their ability of hydroxylation. Previous high-valent iron-oxo species have been noted and studied by observing [(Por•)Fe(IV)=O]+ in heme systems. However, it is yet to be established that a high-valent state could be accessed without a nonheme ligand environment. Que and his group studied various nonheme iron based complexes and through elaborate mechanistic work proved that Fe(V)=O species can indeed exist without the supporting heme ligand.

Nonheme iron oxygenases

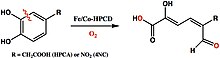
One of Que’s focuses is on the activation of dioxygen species in biological systems through non-heme iron active sites. More specifically, one of Que’s focuses is on homoprotocatechuate (HPCA) 2,3-dioxygenase, which mediates the electron transfer between catechol substrates and O2 to form a [M(II)(semiquinone)superoxo] intermediate. [17] Non-heme iron active sites, including those involving Mn(II) and Co(II), have proven to be equally potent as heme iron active sites with comparable or greater KMO2 and kcat. [18] By exploring nonheme iron oxygenases, Que hopes to optimize the first steps in the industrial production of methane gas, which would yield enormous energy savings for industry.
High-valent iron-oxos

Que has worked on synthesizing oxoiron(IV) complexes. In 2003, Que reported the crystallographic and spectroscopic characterization of a low spin (S = 1) Fe(IV)=O. [19] The first crystallographic example of a high spin (S = 2) Fe(IV)=O was achieved in 2009, [20] and in 2011, Que proved that the ligand used in the previous study could also support a tricationic cyanoiron(IV) complex. [21]
References
- ^ "Current Regents Professors". University of Minnesota Awards. Retrieved 2018-02-10.
- ^ ACS Awards
- ^ ACS Awards
- ^ a b "Lawrence Que Jr., RSC Inorganic Mechanisms 2011 Awardee and Scientific Expert on Iron-containing Enzymes —". Asian Journal News. 2011-10-01. Retrieved 2021-05-12.
- ^ a b c d "Lawrence "Larry" Que Jr. | Department of Chemistry | College of Science and Engineering". cse.umn.edu. Retrieved 2021-05-12.
- ^ "Pignolet". www1.chem.umn.edu. Retrieved 2021-05-12.
- ^ Que, Lawrence; Pignolet, L. H. (1973). "Proton magnetic resonance study of the stereochemistry of four-coordinate nickel(II) complexes. Dihalobis(tertiary phosphine)nickel(II) complexes". Inorganic Chemistry. 12 (1): 156–163. doi: 10.1021/ic50119a036. ISSN 0020-1669.
- ^ Palazzotto, M. C.; Duffy, D. J.; Edgar, B. L.; Que, L.; Pignolet, L. H. (1973). "Dynamic stereochemistry of tris(chelate) complexes. I. Tris(dithiocarbamato) complexes of iron, cobalt, and rhodium". Journal of the American Chemical Society. 95 (14): 4537–4545. doi: 10.1021/ja00795a013. ISSN 0002-7863.
- ^ Que, L.; Pignolet, L. H. (1974-02-01). "Dynamic stereochemistry of tris-chelate complexes. II. Tris(dithiocarbamato) complexes of manganese (III), vanadium(III) chromium(III), gallium(III), and indium(III)". Inorganic Chemistry. 13 (2): 351–356. doi: 10.1021/ic50132a022. ISSN 0020-1669.
- ^ Que, L.; Holm, R. H.; Mortenson, L. E. (1975). "Extrusion of iron-sulfide (Fe2S2* and Fe4S4*) cores from the active sites of ferredoxin proteins". Journal of the American Chemical Society. 97 (2): 463–464. doi: 10.1021/ja00835a064. ISSN 0002-7863. PMID 1133364.
- ^ Que, L.; Bobrik, M. A.; Ibers, James A.; Holm, R. H. (1974). "Synthetic analogs of the active sites of iron-sulfur proteins. VII. Ligand substitution reactions of the tetranuclear clusters [Fe4S4(SR)2- and the structure of bis(tetramethylammonium) [tetra-.mu.-sulfide-tetrakis(benzenethiolato)tetrairon". Journal of the American Chemical Society. 96 (13): 4168–4178. doi: 10.1021/ja00820a018. ISSN 0002-7863. PMID 4854592.
- ^ Que, L.; Anglin, J. R.; Bobrik, M. A.; Davison, A.; Holm, R. H. (1974). "Synthetic analogs of the active sites of iron-sulfur proteins. IX. Formation and some electronic and reactivity properties of iron sulfide (Fe4S4) glycyl-L-cysteinylglycyl oligopeptide complexes obtained by ligand substitution reactions". Journal of the American Chemical Society. 96 (19): 6042–6048. doi: 10.1021/ja00826a014. ISSN 0002-7863. PMID 4416035.
- ^ Jasniewski, Andrew J.; Que, Lawrence (2018-03-14). "Dioxygen Activation by Nonheme Diiron Enzymes: Diverse Dioxygen Adducts, High-Valent Intermediates, and Related Model Complexes". Chemical Reviews. 118 (5): 2554–2592. doi: 10.1021/acs.chemrev.7b00457. ISSN 0009-2665. PMC 5920527. PMID 29400961.
- ^ a b "Bailar Lecturer 2011-12 - Lawrence Que Jr. | Chemistry at Illinois". chemistry.illinois.edu. Retrieved 2021-05-12.
- ^ Que, L.; Lipscomb, J.D; Zimmermann, R.; Münck, E.; Ormejohnson, N.R; Orme-Johnson, W.H (1976-12-08). "Mössbauer and EPR spectroscopy on protocatechuate 3,4-dioxygenase from Pseudomonas aeruginosa". Biochimica et Biophysica Acta (BBA) - Enzymology. 452 (2): 320–334. doi: 10.1016/0005-2744(76)90182-0. ISSN 0005-2744. PMID 188463.
- ^ Que Jr, L.; Lipscomb, J. D.; Münck, E.; Wood, J. M. (1977-11-23). "Protocatechuate 3,4-dioxygenase: Inhibitor studies and mechanistic implications". Biochimica et Biophysica Acta (BBA) - Enzymology. 485 (1): 60–74. doi: 10.1016/0005-2744(77)90193-0. ISSN 0005-2744. PMID 199266.
- ^ a b c Fielding, Andrew J.; Kovaleva, Elena G.; Farquhar, Erik R.; Lipscomb, John D.; Que, Lawrence (February 2011). "A Hyperactive Cobalt-Substituted Extradiol-Cleaving Catechol Dioxygenase". Journal of Biological Inorganic Chemistry. 16 (2): 341–355. doi: 10.1007/s00775-010-0732-0. ISSN 0949-8257. PMC 3192431. PMID 21153851.
- ^ a b c Fielding, Andrew J.; Lipscomb, John D.; Que, Lawrence (2012-01-18). "Characterization of an O2 Adduct of an Active Cobalt-Substituted Extradiol-Cleaving Catechol Dioxygenase". Journal of the American Chemical Society. 134 (2): 796–799. doi: 10.1021/ja2095365. ISSN 0002-7863. PMC 3262093. PMID 22175783.
- ^ a b Rohde, Jan-Uwe; In, Jun-Hee; Lim, Mi Hee; Brennessel, William W.; Bukowski, Michael R.; Stubna, Audria; Münck, Eckard; Nam, Wonwoo; Que, Lawrence (2003-02-14). "Crystallographic and Spectroscopic Characterization of a Nonheme Fe(IV)=O Complex". Science. 299 (5609) (published 14 Feb 2003): 1037–1039. doi: 10.1126/science.299.5609.1037. ISSN 0036-8075. PMID 12586936.
- ^ England, J.; Martinho, M.; Farquhar, E. R.; Frisch, J. R.; Bominaar, E. L.; Münck, E.; Que, L. Jr. (2009). "A Synthetic High-Spin Oxoiron(IV) Complex: Generation, Spectroscopic Characterization, and Reactivity". Angew. Chem. Int. Ed. 48 (20): 3622–3626. doi: 10.1002/anie.200900863. PMC 2719303. PMID 19373820.
- ^ England, J.; Farquhar, E. R.; Guo, Y.; Cranswick, M. A.; Ray, K.; Münck, E.; Que, L. Jr. (2011). "Characterization of a Tricationic Trigonal Bipyramidal Iron(IV) Cyanide Complex, with a Very High Reduction Potential, and Its Iron(II) and Iron(III) Congeners". Inorg. Chem. 50 (7): 2885–2896. doi: 10.1021/ic102094d. PMC 3065519. PMID 21381646.
External links
- Lawrence Que Jr. publications indexed by Google Scholar