Late-stage functionalization (LSF) is a desired, chemical or biochemical, chemoselective transformation on a complex molecule to provide at least one analog in sufficient quantity and purity for a given purpose without needing the addition of a functional group that exclusively serves to enable said transformation. [1]
Molecular complexity is an intrinsic property of each molecule and frequently determines the synthetic effort to make it. [2] [3] LSF can significantly diminish this synthetic effort, and thus enables access to molecules, which would otherwise not be available or too difficult to access. The requirements for LSF can be met by both C–H functionalization reactions and functional group manipulations. [1] LSF reactions are particularly relevant and often used in the fields of drug discovery and materials chemistry, [4] [5] [6] although no LSF has been implemented in a commercial process.

All LSF reactions are chemoselective but not every chemoselective reaction fulfills the requirements of the definition for LSF. [1] High chemoselectivity is required for a useful LSF with a predictable reaction outcome because complex molecules typically feature several distinct functional groups that need to be tolerated. In this sense, chemoselectivity is sometimes referred to as functional group tolerance. Furthermore, high chemoselectivity avoids often undesired over-functionalization of the valuable substrate, which is used as a limiting reagent in LSF reactions. [1]
Every C–H bond functionalization on a complex molecule classifies as LSF, except when a directing or activating group must be installed in a previous step of the synthesis to accomplish the transformation. For functional group manipulations, the distinction between LSF and functional-group-tolerant reactions is more subtle. For example, peptide bioconjugation reactions make use of the native functionality in amino acid side chains, and thus classify as LSF. In contrast, bioorthogonal 1,3-dipolar cycloadditions (see also copper-free click chemistry and Huisgen cycloaddition) generally require prior introduction of azide or cycloalkyne functionalities to biomolecules. Hence, such transformations do not classify as LSF despite their excellent functional group tolerance. [1] [7] [8]

Site-selectivity, also positional or regioselectivity, is generally desired but no requirement for LSF reactions because site-unselective LSF reactions can also be useful for special purposes. For example, site-unselective late-stage C–H functionalization reactions can provide quick access to several constitutional isomers of complex molecules relevant for biological testing in drug discovery. [1] [4] [5] [9] Site-selective reactions to access each possible constitutional isomer independently are scarce but highly desirable because cumbersome purification procedures are avoided, and other isomers are not produced as waste. Some LSF reactions provide one constitutional isomer in high selectivity based on innate substrate selectivity for a given reaction or based on catalyst control. The discovery of site-selective LSF reactions constitutes an important research objective in the field of synthetic methodology development. [1] [10] [11] [12]

- ^ a b c d e f g Börgel, Jonas; Ritter, Tobias (2020-08-06). "Late-Stage Functionalization". Chem. 6 (8): 1877–1887. doi: 10.1016/j.chempr.2020.07.007. ISSN 2451-9294.
- ^ von Korff, Modest; Sander, Thomas (2019-01-30). "Molecular Complexity Calculated by Fractal Dimension". Scientific Reports. 9 (1): 967. Bibcode: 2019NatSR...9..967V. doi: 10.1038/s41598-018-37253-8. ISSN 2045-2322. PMC 6353876. PMID 30700728.
- ^ Corey, E. J.; Wipke, W. Todd (1969-10-10). "Computer-Assisted Design of Complex Organic Syntheses". Science. 166 (3902): 178–192. Bibcode: 1969Sci...166..178C. doi: 10.1126/science.166.3902.178. ISSN 0036-8075. PMID 17731475.
- ^ a b Cernak, Tim; Dykstra, Kevin D.; Tyagarajan, Sriram; Vachal, Petr; Krska, Shane W. (2016-02-01). "The medicinal chemist's toolbox for late stage functionalization of drug-like molecules". Chemical Society Reviews. 45 (3): 546–576. doi: 10.1039/C5CS00628G. ISSN 1460-4744. PMID 26507237.
- ^ a b Moir, Michael; Danon, Jonathan J.; Reekie, Tristan A.; Kassiou, Michael (2019-11-02). "An overview of late-stage functionalization in today's drug discovery". Expert Opinion on Drug Discovery. 14 (11): 1137–1149. doi: 10.1080/17460441.2019.1653850. hdl: 1885/213256. ISSN 1746-0441. PMID 31411499. S2CID 199572352.
- ^ Wencel-Delord, Joanna; Glorius, Frank (2013-04-23). "C–H bond activation enables the rapid construction and late-stage diversification of functional molecules". Nature Chemistry. 5 (5): 369–375. Bibcode: 2013NatCh...5..369W. doi: 10.1038/nchem.1607. ISSN 1755-4349. PMID 23609086.
- ^ a b Lin, Shixian; Yang, Xiaoyu; Jia, Shang; Weeks, Amy M.; Hornsby, Michael; Lee, Peter S.; Nichiporuk, Rita V.; Iavarone, Anthony T.; Wells, James A.; Toste, F. Dean; Chang, Christopher J. (2017-02-10). "Redox-based reagents for chemoselective methionine bioconjugation". Science. 355 (6325): 597–602. Bibcode: 2017Sci...355..597L. doi: 10.1126/science.aal3316. ISSN 0036-8075. OSTI 1465412. PMC 5827972. PMID 28183972.
- ^ Jewett, John C.; Bertozzi, Carolyn R. (2010-03-22). "Cu-free click cycloaddition reactions in chemical biology". Chemical Society Reviews. 39 (4): 1272–1279. doi: 10.1039/B901970G. ISSN 1460-4744. PMC 2865253. PMID 20349533.
- ^ a b Nagib, David A.; MacMillan, David W. C. (2011-12-08). "Trifluoromethylation of arenes and heteroarenes by means of photoredox catalysis". Nature. 480 (7376): 224–228. Bibcode: 2011Natur.480..224N. doi: 10.1038/nature10647. ISSN 1476-4687. PMC 3310175. PMID 22158245.
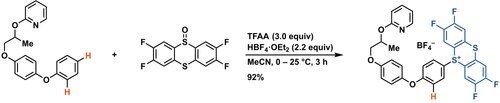
- ^ a b Berger, Florian; Plutschack, Matthew B.; Riegger, Julian; Yu, Wanwan; Speicher, Samira; Ho, Matthew; Frank, Nils; Ritter, Tobias (2019-03-13). "Site-selective and versatile aromatic C−H functionalization by thianthrenation". Nature. 567 (7747): 223–228. Bibcode: 2019Natur.567..223B. doi: 10.1038/s41586-019-0982-0. ISSN 1476-4687. PMID 30867606. S2CID 76660453.
- ^ a b Zhang, Kaidong; Shafer, Brian M.; Demars, Matthew D.; Stern, Harry A.; Fasan, Rudi (2012-11-14). "Controlled Oxidation of Remote sp3 C–H Bonds in Artemisinin via P450 Catalysts with Fine-Tuned Regio- and Stereoselectivity". Journal of the American Chemical Society. 134 (45): 18695–18704. doi: 10.1021/ja3073462. ISSN 0002-7863. PMC 3498520. PMID 23121379.
- ^ Lasso, Juan D.; Castillo-Pazos, Durbis J.; Li, Chao-Jun (2021-10-04). "Green chemistry meets medicinal chemistry: a perspective on modern metal-free late-stage functionalization reactions". Chemical Society Reviews. 50 (19): 10955–10982. doi: 10.1039/D1CS00380A. ISSN 1460-4744. PMID 34382989. S2CID 236988469.