| |
| |
| Names | |
|---|---|
|
IUPAC name
iron(III) acetate
| |
| Other names
basic iron(III) acetate , iron(III) oxyacetate, iron(III) Acetate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
| |
| |
| Properties | |
| C14H27Fe3O18 | |
| Molar mass | 650.9 g/mol |
| Appearance | brownish-red powder |
| Insoluble | |
| Solubility | soluble in ethanol [2] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
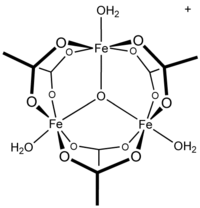
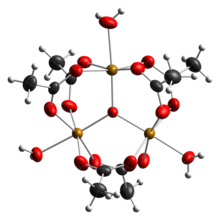
Ferric acetate is the acetate salt of the coordination complex [Fe3O(OAc)6(H2O)3+ (OAc− is CH3CO2−). Commonly the salt is known as "basic iron acetate". [3] The formation of the red-brown complex was once used as a test for ferric ions. [4]
Structure and synthesis
Basic iron acetate forms on treating aqueous solutions of iron(III) sources with acetate salts. [5] A typical precursor is freshly precipitated iron oxide/hydroxide, which is halide-free. [6]
Early work showed that the cation is trinuclear. [7] The Fe centres are equivalent, each being octahedral, being bound to six oxygen ligands, including a triply bridging oxide at the center of the equilateral triangle. [8] The compound was an early example of a molecular complex of iron that features an oxide ligand. The cation has idealized D3h symmetry.
Reactions
The terminal aqua ligands on the trimetallic framework can be substituted with other ligands, such as pyridine and dimethylformamide. Many different salts are known by exchanging the anion, e.g. [Fe3(μ3-O)(OAc)6(H2O)3]Cl. Reduction of the cation affords the neutral mixed-valence derivative that contains one ferrous and two ferric centers. [3] Mixed metal species are known such as [Fe2CoO(OAc)6(H2O)3]. [9]
Related compounds
Chromium(III), ruthenium(III), vanadium(III), manganese(III) and rhodium(III) form analogous compounds. [10] Iron(III) acetate (lacking the oxo ligand) has been claimed as a red coloured compound from the reaction of silver acetate and iron(III) chloride. [11]
Uses
Materials prepared by heating iron, acetic acid, and air, loosely described as basic iron acetates, are used as dyes and mordants. [3]
Iron acetate is often brushed upon untreated wood to give it an aged appearance.
See also
- Iron(II) acetate
- Cobalt(II) acetate
- Rhodium(II) acetate
- Manganese(III) acetate
- Chromium(III) acetate
References
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. pp. 4–63. ISBN 0-8493-0487-3.
- ^ "Iron(III) Acetate". EndMemo. Retrieved 18 April 2015.
- ^ a b c J., Burgess; M. V., Twigg (2005). R. Bruce, King; J., Wiley (eds.). Encyclopedia of inorganic chemistry (2nd ed.). New York: Wiley. ISBN 978-0-470-86078-6.
- ^ H., Brearley; F., Ibbotson (1902). The Analysis of Steel-Works Materials. London ; New York: Longmans, Green. Archived from the original on 18 April 2015.
- ^ W., Simon (March 2007). Manual of Chemistry. p. 474. ISBN 978-1406733358.
- ^ H. Lux (1963). "Basic Iron(III) Acetate". In Georg Brauer (ed.). Handbook of Preparative Inorganic Chemistry. p. 1508.
- ^ Weinland, R.; Dinkelacker, P. (July 1909). "Über Salze einer Hexaacetato(formiato)-trichrombase. II". Berichte der Deutschen Chemischen Gesellschaft. 42 (3): 2997–3018. doi: 10.1002/cber.19090420318.
- ^ Figgis, B. N.; Robertson, G. B. (13 February 1965). "Crystal-Molecular Structure and Magnetic Properties of Cr3(CH3.COO)6OCl.5H2O". Nature. 205 (4972): 694–695. Bibcode: 1965Natur.205..694F. doi: 10.1038/205694a0. S2CID 4283321. This paper describes the isostructure chromium and iron compounds.
- ^ Blake, Antony B.; Yavari, Ahmad; Hatfield, William E.; Sethulekshmi, C. N. (1985). "Magnetic and spectroscopic properties of some heterotrinuclear basic acetates of chromium(III), iron(III), and divalent metal ions". Journal of the Chemical Society, Dalton Transactions (12): 2509. doi: 10.1039/DT9850002509.
- ^ Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, ISBN 0-12-352651-5
- ^ Paul, Ram C.; Narula, Ramesh C.; Vasisht, Sham K. (December 1978). "Iron(III) acetates". Transition Metal Chemistry. 3 (1): 35–38. doi: 10.1007/BF01393501. S2CID 94447648.