| |
| Names | |
|---|---|
|
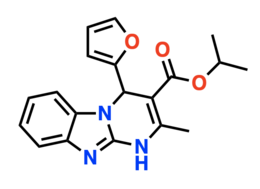
IUPAC names
(±)-Isopropyl 4-(Furan-2-yl)-2-methyl-1,4-dihydrobenzo-[4,5]imidazo[1,2-a]pyrimidine-3-carboxylate,
4-(2-Furanyl)-4,10-dihydro-2-methyl-pyrimido[1,2-a]benzimidazole-3-carboxylic acid 1-methylethyl ester
| |
| Other names
ISAM140
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C19H19N3O3 | |
| Molar mass | 337.37 |
| Appearance | White solid |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
ISAM-140 is a selective non- xanthinic adenosine A2B receptor atagonist. Discovered in 2016, [1] has a Ki of 3.49 nM in A2B receptor and >1000-fold selectivity with respect to the other three adenosine receptor subtypes. It has been shown to help immune system to attack cancer cells in in vitro assays, by rescuing T and NK cell proliferation, cytokine release and TIL infiltration. [2]
References
- ^ El Maatougui, Abdelaziz; Azuaje, Jhonny; González-Gómez, Manuel; Miguez, Gabriel; Crespo, Abel; Carbajales, Carlos; Escalante, Luz; García-Mera, Xerardo; Gutiérrez-de-Terán, Hugo; Sotelo, Eddy (2016-03-10). "Discovery of Potent and Highly Selective A 2B Adenosine Receptor Antagonist Chemotypes". Journal of Medicinal Chemistry. 59 (5): 1967–1983. doi: 10.1021/acs.jmedchem.5b01586. ISSN 0022-2623. PMID 26824742.
- ^ Tay, Apple Hui Min; Prieto-Díaz, Rubén; Neo, Shiyong; Tong, Le; Chen, Xinsong; Carannante, Valentina; Önfelt, Björn; Hartman, Johan; Haglund, Felix; Majellaro, Maria; Azuaje, Jhonny (2022-05-01). "A2B adenosine receptor antagonists rescue lymphocyte activity in adenosine-producing patient-derived cancer models". Journal for ImmunoTherapy of Cancer. 10 (5): e004592. doi: 10.1136/jitc-2022-004592. ISSN 2051-1426. PMC 9115112. PMID 35580926.