| |
| Names | |
|---|---|
|
IUPAC name
1,3,4,6,7,9,9b-heptaazaphenalene-2,5,8-tricarbodiimide
| |
| Other names | |
| Identifiers | |
3D model (
JSmol)
|
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C9H3N13 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
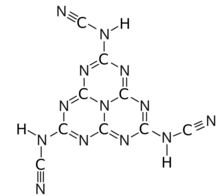
Hydromelonic acid,
[1] is an elusive
chemical compound with formula C
9H
3N
13 or (HNCN)
3(C
6N
7), whose molecule would consist of a
heptazine H3(C
6N
7) molecule, with three
cyanamido groups H–N=C=N– or N≡C–NH– substituted for the
hydrogen atoms.
The compound had not been properly isolated as of 2010, due to its tendency to
polymerize.
[3]
[4] However, removal of three protons yields the melonate
[5] (formerly hydromelonate
[1]) anion (NCN)
3(C
6N
7)3−
, whose salts are stable and have been known since the 19th century. Removal of only two protons yields the divalent hydrogenmelonate anion H(NCN)
3(C
6N
7)2−
.
[3]
[5]
History
In 1834
Justus von Liebig described the compounds that he named
melamine,
melam, and
melon.
[6] In 1835
Leopold Gmelin prepared
a potassium salt (later identified as K
3[(NCN)
3(C
6N
7)]) by heating
potassium ferrocyanide with
sulfur); recognizing their connection to the compounds described by Liebig, he named the salt "hydromelonate" and the corresponding acid "hydromelonic".
[1] In the following years Liebig prepared the same salt by other methods, such as by fusing
potassium thiocyanate with
antimony trichloride,
[7] and eventually determined the formula C
9N
13H
3 for the acid.
[8]
[9]
The correct structure for the cation was published in 1937 by Linus Pauling and J. H. Sturdivant. [9]
Starting in the 1970s, melonates have attracted new interest, motivated by research in
cubic
carbon nitride c-C
3N
4.
[3] Salts of many cations have been synthesized and studied,
[2]
[10]
[4]
[11] including mixed-cation salts
[5] and
calcium hydrogenmelonate Ca[HC
9N
13]·7H
2O.
[3] The structure of potassium melonate pentahydrate K
3C
9N
13·5H
2O was elucidated only in 2005.
[5]
Preparation
Sodium and potassium melonates are still routinely prepared by variations of one of Liebig's methods, [7] namely by reacting melon with molten potassium thiocyanate, potassium cyanate, or sodium thiocyanate. [2] Other salts can be prepared by double displacement reactions. [3]
Melonates

3(C
6N
7)3−
.
Alkali melonates are soluble in water without decomposition. [10] Treatment of melonates with alkali gives salts of cyameluric acid. [12]
Potassium melonate pentahydrate has planar sheets of melonate anions with potassium cations between the layers. The same generals structure is seen in the anhydrous salt (except that the melonate ions are asymmetric due to an NHN– group being turned by 180°) and in the salts Rb
3C
9N
13·3H
2O and Cs
3C
9N
13·3H
2O.
[3]
[5]
Melonate salts decompose when melted with cyanates to form tricyanomelaminates, the first selective decomposition reaction leading from heptazines to triazines. [2]
Pyrolysis of
iron(III) melonate Fe[C
9N
13 at very high temperature yields
carbon in the form of
nanotubes.
[4]
See also
-
Melem, (H
2N)
3(C
6N
7) -
Cyameluric acid, (HO)
3(C
6N
7)
References
- ^ a b c d L. Gmelin, Ann. Pharmacie, 15, 252 (1835).
- ^
a
b
c
d
e Andreas Sattler and Wolfgang Schnick (2009): "On the Formation and Decomposition of the Melonate Ion in Cyanate and Thiocyanate Melts and the Crystal Structure of Potassium Melonate,K
3[C
6N
7(NCN)
3". European Journal of Inorganic Chemistry, volume 2009, issue 33, pages 4972-4981. doi: 10.1002/ejic.200900585 - ^ a b c d e f Sophia J. Makowski, Daniel Gunzelmann, Jürgen Senker, Wolfgang Schnick (2009): "Protonated Melonate Ca[HC6N7(NCN)3]·7H2O – Synthesis, Crystal Structure, and Thermal Properties". Zeitschrift für anorganische und allgemeine Chemie (ZAAC), volume 635, issue 15, pages 2434-2439. doi: 10.1002/zaac.200900231
- ^
a
b
c Corinna Clauss, Marcus Schwarz, and Edwin Kroke (2010): "Microwave-induced decomposition of nitrogen-rich iron salts and CNT formation from iron(III)–melonate Fe[C
9N
13". Carbon, volume 48, issue 4,pages 1137-1145. doi: 10.1016/j.carbon.2009.11.036 - ^ a b c d e Fabian Karl Keßler (2019), Structure and Reactivity of s-Triazine-Based Compounds in C/N/H Chemistry. Doctoral thesis, Fakultät für Chemie und Pharmazie, Ludwig-Maximilians-Universität München
- ^ J. Liebig, Ibid., 10, 1 (1834).
- ^ a b J. Liebig, Ann. Chem. Pharm., 50, 337 (1844).
- ^ J. Liebig, Ibid., 95, 257 (1855).
- ^ a b Linus Pauling and J. H. Sturdivant (1937): "The Structure of Cyameluric Acid, Hydromelonic Acid and Related Substances". Proceedings of the National Academy of Sciences, volume 23, issue 12, page 615–620. doi: 10.1073/pnas.23.12.615
- ^
a
b Corinna Clauss, Jörg Wagler, Marcus Schwarz, Anke Schwarzer, and Edwin Kroke (2010): "Lithium Melonate, Li
3[C
6N
7(NCN)
3]·6H
2O – Synthesis, Crystal Structure and Thermal Properties of a Novel Precursor for Graphitic Carbon Nitrides". 'Zeitschrift für anorganische und allgemeine Chemie (ZAAC), vlume 636, issue 1, pages 196-200. doi: 10.1002/zaac.200900326 -
^ Wolfgang Schnick and Sophia Makowski (2010): "
Rb
3[C
6N
7(NCN)
3]•3H
2O and Cs
3[C
6N
7(NCN)
3]•3H
2O – Synthesis, Crystal Structure and Thermal Behavior of two Novel Alkali Melonates". CSSD HAL, article hal-00511998 version 1 doi: 10.1002/zaac.200900232 - ^ W. Henneberg, Ibid., 73, 228 (1850).