This article is an
orphan, as no other articles
link to it. Please
introduce links to this page from
related articles; try the
Find link tool for suggestions. (June 2024) |
Glucose chain shortening and lengthening is the chemical processes for decreasing or increasing the carbon chain length of
glucose. Glucose can be shortened by oxidation and decarboxylation to generate
arabinose, a reaction known as the
Ruff degradation.
[1] To increase the glucose carbon chain, a series of chemical reactions can be used to add one more carbon at the
aldehyde end of glucose; this process is known as the
Kiliani–Fischer synthesis.
[2]

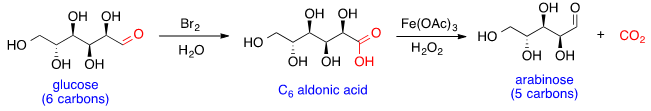
Glucose can react with
bromine in water to form the
aldonic acid, which can then undergo
oxidative decarboxylation with
hydrogen peroxide and
iron (III) acetate to form arabinose. This reaction can be conducted iteratively, shortening one carbon at a time to generate sugars with smaller chain lengths.
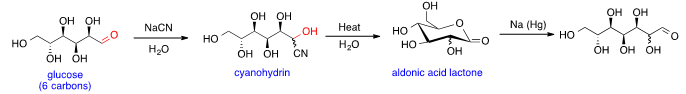
Glucose can be the substrate in Kiliani–Fischer synthesis that adds a carbon to the aldehyde group and forms sugars with one more carbon than the substrate. The first step is
nucleophilic addition of aqueous
cyanide to aldehyde group in glucose to generate the
cyanohydrin. Then cyanohydrin is hydrolyzed to form
aldonic acid lactone upon heating. Finally, the aldonic acid lactone is reduced with
sodium amalgam non-stereoselectively to produce
heptose; two isomers with both stereoisomers at C2 are generated.