
The fluorenylmethoxycarbonyl protecting group (Fmoc) is a base-labile protecting group used in organic synthesis.
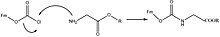
Fmoc carbamate is frequently used as a protecting group for amines, where the Fmoc group can be introduced by reacting the amine with fluorenylmethyloxycarbonyl chloride (Fmoc-Cl), e.g.: [1]
The other common method for introducing the Fmoc group is through 9-fluorenylmethylsuccinimidyl carbonate (Fmoc-OSu), which may itself be obtained by the reaction of Fmoc-Cl with the dicyclohexylammonium salt of N-hydroxysuccinimide. [2]
Reacting with 9-fluorenylmethyloxycarbonyl azide (itself made by reacting Fmoc-Cl with sodium azide) in sodium bicarbonate and aqueous dioxane is also a method to install Fmoc group. [3]
Because the fluorenyl group is highly fluorescent, certain UV-inactive compounds may be reacted to give the Fmoc derivatives, suitable for analysis by reversed phase HPLC. Analytical uses of Fmoc-Cl that do not use chromatography may be limited by the requirement that excess Fmoc-Cl be removed before an analysis of fluorescence.
The Fmoc group is rapidly removed by base. Piperidine is usually preferred for Fmoc group removal as it forms a stable adduct with the dibenzofulvene byproduct, preventing it from reacting with the substrate. [4] [5]
The use of Fmoc as a temporary protecting group for amine at the N-terminus in SPPS is very widespread for Fmoc/tBu approach, because its removal with piperidine solution does not disturb the acid-labile linker between the peptide and the resin. [6] A typical SPPS Fmoc deprotection is performed with a solution of 20% piperidine in N,N-dimethylformamide (DMF). [7]
- C13H9−CH2−OC(O)NHR + (CH2)5NH → (CH2)5NH+2 + [C13H8−CH2−OC(O)NHR]−
- [C13H8−CH2−OC(O)NHR]− → C13H8=CH2 + −OC(O)NHR
- −OC(O)NHR + (CH2)5NH+2 → HOC(O)NHR + (CH2)5NH
- HOC(O)NHR → CO2 + RNH2
- C13H8=CH2 + (CH2)5NH → C13H9−CH2N(CH2)5
Common deprotection cocktails for Fmoc during SPPS:
- 20% piperidine in DMF (Fmoc group has an approximate half life of 6 seconds in this solution) [7]
- 5% piperazine, 1% DBU and 1% formic acid in DMF. This method avoids the use of strictly controlled piperidine. [8] No side product was observed for a peptide with 9 residues synthesized with this method. [9]
- ^ Yamada, Kazuhiko; Hashizume, Daisuke; Shimizu, Tadashi; Ohki, Shinobu; Yokoyama, Shigeyuki (2008). "A solid-state 17O NMR, X-ray, and quantum chemical study of N-α-Fmoc-protected amino acids". Journal of Molecular Structure. 888 (1–3): 187–196. doi: 10.1016/j.molstruc.2007.11.059.
- ^ Paquet, A. (1982). "Introduction of 9-fluorenylmethyloxycarbonyl, trichloroethoxycarbonyl, and benzyloxycarbonyl amine protecting groups into O-unprotected hydroxyamino acids using succinimidyl carbonates". Canadian Journal of Chemistry. 60 (8): 976–980. doi: 10.1139/v82-146.
- ^ Carpino, Louis A.; Han, Grace Y. (1972). "9-Fluorenylmethoxycarbonyl amino-protecting group". The Journal of Organic Chemistry. 37 (22): 3404–3409. doi: 10.1021/jo00795a005.
- ^ Fields, Gregg B. (1995), Pennington, Michael W.; Dunn, Ben M. (eds.), "Methods for Removing the Finoc Group", Peptide Synthesis Protocols, Methods in Molecular Biology, vol. 35, Totowa, NJ: Humana Press, pp. 17–27, doi: 10.1385/0-89603-273-6:17, ISBN 978-1-59259-522-8, PMID 7894598, retrieved 2021-10-15
- ^ Wellings, Donald A.; Atherton, Eric (1997). "[4] Standard Fmoc protocols". Solid-Phase Peptide Synthesis. Methods in Enzymology. Vol. 289. pp. 44–67. doi: 10.1016/s0076-6879(97)89043-x. ISBN 9780121821906. PMID 9353717.
- ^ J. Jones, Amino Acid and Peptide Synthesis, 2nd edn., Oxford University Press, 2002
- ^ a b Wuts, P. G. M.; Greene, T.W. (2006). Greene's Protective Groups in Organic Synthesis. NY: J. Wiley. doi: 10.1002/0470053488. ISBN 9780470053485.
- ^ Ralhan, Krittika; KrishnaKumar, V. Guru; Gupta, Sharad (8 December 2015). "Piperazine and DBU: a safer alternative for rapid and efficient Fmoc deprotection in solid phase peptide synthesis". RSC Advances. 5 (126): 104417–104425. Bibcode: 2015RSCAd...5j4417R. doi: 10.1039/C5RA23441G. ISSN 2046-2069.
- ^ Lam, Pak-Lun; Wu, Yue; Wong, Ka-Leung (30 March 2022). "Incorporation of Fmoc-Dab(Mtt)-OH during solid-phase peptide synthesis: a word of caution". Organic & Biomolecular Chemistry. 20 (13): 2601–2604. doi: 10.1039/D2OB00070A. ISSN 1477-0539. PMID 35258068. S2CID 247175352.
-
 Media related to
Fmoc at Wikimedia Commons
Media related to
Fmoc at Wikimedia Commons
