Size of this PNG preview of this SVG file:
690 × 600 pixels. Other resolutions:
276 × 240 pixels |
552 × 480 pixels |
884 × 768 pixels |
1,179 × 1,024 pixels |
2,357 × 2,048 pixels |
1,152 × 1,001 pixels.
Original file (SVG file, nominally 1,152 × 1,001 pixels, file size: 1.28 MB)
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 18:21, 23 September 2021 |
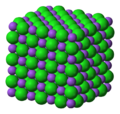
 | 1,152 × 1,001 (1.28 MB) | Goran tek-en | {{Information |description ={{en|1=The crystal structure of sodium chloride, NaCl, a typical ionic compound, emphasising the octahedral environment of one sodium cation, Na<sup>+</sup>, illustrated as a <span style="color:#8F58C0; background-color:#8F58C0;">__</span> purple sphere and one chloride anion, Cl<sup>−</sup>, illustrated as a <span style="color:#28D028;background-color:#28D028;">__</span> green sphere. The yellow stipples represent the [[:en:electro... |
File usage
The following pages on the English Wikipedia use this file (pages on other projects are not listed):
Global file usage
The following other wikis use this file:
- Usage on pt.wikipedia.org
- Usage on tr.wikipedia.org