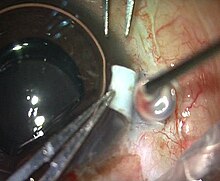
 Fibrin glue applied after drying the scleral bed in an
intraocular lens operation | |
| Combination of | |
|---|---|
| Fibrinogen | Glycoprotein |
| Thrombin | Coagulation factor |
| Clinical data | |
| Trade names | Artiss, Evicel, Tisseel, others |
| AHFS/ Drugs.com | Professional Drug Facts |
| License data | |
|
Pregnancy category | |
|
Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| DrugBank | |
| UNII | |
Fibrin glue (also called fibrin sealant) is a surgical formulation used to create a fibrin clot for hemostasis, cartilage repair surgeries or wound healing. It contains separately packaged human fibrinogen and human thrombin. [8] [9] [10] [11] [12] [13] [14] [15] [16] [17]
Medical uses
This glue is used as a supportive treatment in surgery (such as liver surgery) for the improvement of hemostasis where standard surgical techniques are insufficient or impractical. [18] [19]
It is also used for repairing dura mater tears and bronchial fistulas and for achieving hemostasis after spleen and liver trauma, [16] in "no sutures" corneal transplantation, pterygium excision with amniotic membrane or conjunctival autograft, and in eye trauma for corneal or conjunctival defects, [20] [21] [22] as well as for skin graft donor site wounds to reduce postoperative pain. [23]
It can also be used to treat pilonidal sinus disease but it is of unclear benefit as of 2017, due to insufficient research. [24]
Contraindications
The glue must not get into blood vessels, as this could lead to clotting in the form of thromboembolism or disseminated intravascular coagulation, or to anaphylaxis (a severe allergic reaction). [25]
Side effects
Possible adverse effects include bleeding disorder and allergic reactions such as flushing, stinging, generalised urticaria, angioedema, bronchospasm, and anaphylaxis. Other adverse effects in studies occurred in roughly equal proportions in treatment and placebo groups. [25]
Interactions
As fibrin glue contains proteins, it may be denatured by ethanol, iodine and heavy metals. These substances are frequently found in antiseptic solutions. [25]
Pharmacology
Mechanism of action
Thrombin is an enzyme that splits fibrinogen into fibrin monomers in 10 to 60 seconds, which aggregate to form a three-dimensional gel-like structure. Thrombin also activates factor XIII from the human body to factor XIIIa, which then cross-links the fibrin monomers to form a stable clot. Both these processes need calcium to work. As the wound heals, the clot is slowly degraded by the enzyme plasmin. [16] [25] [26]
Pharmacokinetics
In rabbit studies, only 1 to 2% of the applied thrombin dose reached the bloodstream. It reached highest blood plasma concentrations after 6 to 8 hours. [25]
Chemistry
Composition
Fibrin glue comes in two vials, respectively containing:
- fibrinogen: lyophilised pooled human concentrate
- thrombin: This used to be of bovine origin; modern formulations contain human thrombin. [11]
The two components are mixed immediately before application. [18] [27] The formulations also contain calcium salts. [25]
Formulations from different manufacturers may also contain aprotinin, fibronectin, plasminogen, and factor XIII. [28] [29]
Factors affecting structure
Factors that influence dimensional structure of fibrin gel giving rise to fine or coarse gel:
- Changing concentration of fibrinogen
- Changing concentration of thrombin – increased concentration increases ultimate tensile strength and Young modulus of gel
- Changing concentration of calcium
- pH
- Temperature
History
A formulation with human thrombin was approved for medical use in the United States in March 2003, and in the European Union in October 2008. [18] [27] [30]
References
- ^ a b "VeraSeal". Therapeutic Goods Administration (TGA). 12 November 2021. Retrieved 28 December 2021.
- ^ "Updates to the Prescribing Medicines in Pregnancy database". Therapeutic Goods Administration (TGA). 12 May 2022. Retrieved 13 May 2022.
- ^ "AusPAR: Human Fibrinogen / Human Thrombin". Therapeutic Goods Administration (TGA). 29 June 2022. Retrieved 17 July 2022.
- ^ "Health product highlights 2021: Annexes of products approved in 2021". Health Canada. 3 August 2022. Retrieved 25 March 2024.
- ^ "TachoSil sealant matrix - Summary of Product Characteristics (SmPC)". (emc). 31 January 2020. Archived from the original on 10 July 2020. Retrieved 10 July 2020.
- ^ "Tisseel Ready to use Solutions for Sealant - Summary of Product Characteristics (SmPC)". (emc). 30 December 2019. Retrieved 10 July 2020.
- ^ "Artiss Solutions for Sealant - Summary of Product Characteristics (SmPC)". (emc). 29 January 2019. Retrieved 10 July 2020.
- ^ Irwin RM, Bonassar LJ, Cohen I, Matuska AM, Commins J, Cole B, Fortier LA (8 November 2019). "The clot thickens: Autologous and allogeneic fibrin sealants are mechanically equivalent in an ex vivo model of cartilage repair". PLOS ONE. 14 (11): e0224756. Bibcode: 2019PLoSO..1424756I. doi: 10.1371/journal.pone.0224756. PMC 6839864. PMID 31703078.
- ^ Shah NV, Meislin R (December 2013). "Current state and use of biological adhesives in orthopedic surgery". Orthopedics. 36 (12): 945–956. doi: 10.3928/01477447-20131120-09. PMID 24579215.
- ^ Smith BL, Matuska AM, Greenwood VL, Gilat R, Wijdicks CA, Cole BJ (15 April 2022). "Autologous Fibrin Sealants Have Comparable Graft Fixation to an Allogeneic Sealant in a Biomechanical Cadaveric Model of Chondral Defect Repair". Arthroscopy, Sports Medicine, and Rehabilitation. 4 (3): e1075–e1082. doi: 10.1016/j.asmr.2022.03.003. ISSN 2666-061X. PMC 9210474. PMID 35747626. S2CID 248208731.
- ^ a b Atrah HI (April 1994). "Fibrin glue". BMJ. 308 (6934): 933–934. doi: 10.1136/bmj.308.6934.933. PMC 2539755. PMID 8173397.
- ^ Thompson DF, Letassy NA, Thompson GD (December 1988). "Fibrin glue: a review of its preparation, efficacy, and adverse effects as a topical hemostat". Drug Intelligence & Clinical Pharmacy. 22 (12): 946–952. doi: 10.1177/106002808802201203. PMID 2468466. S2CID 37017252.
- ^ "Fibrin Glue for Anal Fistula - Digestive Disorders / Gastroenterology". MedHelp.
- ^ Shinohara K, Kobayashi E, Yoshida T, Toyama N, Kiyozaki H, Fujimura A, Miyata M (1998). "Effect of fibrin glue on small and large bowel anastomoses in the rat". European Surgical Research. 30 (1): 8–12. doi: 10.1159/000008552. PMID 9493689. S2CID 46792418.
- ^ Spotnitz WD, Mintz PD, Avery N, Bithell TC, Kaul S, Nolan SP (August 1987). "Fibrin glue from stored human plasma. An inexpensive and efficient method for local blood bank preparation". The American Surgeon. 53 (8): 460–462. PMID 2440358.
- ^ a b c Saxena S, Jain P, Shukla J (2003). "Preparation of two component Fibrin Glue and its clinical evaluation in skin grafts and flaps". Indian J Plast Surg. 36 (1): 14–17. doi: 10.1055/s-0043-1778572.
- ^ Mücke T, Wolff KD (2009). "Performing microvascular anastomosis with fibrin glue--faster, easier, and more reliable?". Microsurgery. 29 (1): 80–81. doi: 10.1002/micr.20556. PMID 18946885. S2CID 31854904.
- ^
a
b
c
"Evicel EPAR". European Medicines Agency. 17 September 2018. Retrieved 12 June 2020.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ "Evicel Fibrin Sealant (Human)". U.S. Food and Drug Administration (FDA). 22 July 2017.
- ^ Narendran N, Mohamed S, Shah S (July 2007). "No sutures corneal grafting--a novel use of overlay sutures and fibrin glue in Deep Anterior Lamellar Keratoplasty". Contact Lens & Anterior Eye. 30 (3): 207–209. doi: 10.1016/j.clae.2007.02.007. PMID 17379570.
- ^ Dal Pizzol MM, Roggia MF, Kwitko S, Marinho DR, Rymer S (2009). "[Use of fibrin glue in ocular surgery]" [Use of fibrin glue in ocular surgery]. Arquivos Brasileiros de Oftalmologia (in Portuguese). 72 (3): 308–312. doi: 10.1590/s0004-27492009000300006. hdl: 10183/37501. PMID 19668958.
- ^ "Fibrin Sealant Fibrin Gluing Haemostasis autologous". vivostat.com.
- ^ Sinha S, Schreiner AJ, Biernaskie J, Nickerson D, Gabriel VA (November 2017). "Treating pain on skin graft donor sites: Review and clinical recommendations". The Journal of Trauma and Acute Care Surgery. 83 (5): 954–964. doi: 10.1097/TA.0000000000001615. PMID 28598907. S2CID 44520644.
- ^ Lund J, Tou S, Doleman B, Williams JP (January 2017). "Fibrin glue for pilonidal sinus disease". The Cochrane Database of Systematic Reviews. 1 (1): CD011923. doi: 10.1002/14651858.CD011923.pub2. PMC 6464784. PMID 28085995.
- ^ a b c d e f "Evicel: EPAR – Product Information" (PDF). European Medicines Agency. 12 June 2020.
- ^ Spotnitz WD (April 2010). "Fibrin sealant: past, present, and future: a brief review". World Journal of Surgery. 34 (4): 632–634. doi: 10.1007/s00268-009-0252-7. PMID 19820991. S2CID 20760524.
- ^ a b "Evicel Fibrin Sealant (Human)- fibrinogen human and thrombin human kit". DailyMed. 17 December 2019. Retrieved 12 June 2020.
- ^ "Tisseel". Swedish official drug catalog.
- ^ KEGG drug: Factor XIII with fibrinogen. Accessed 9 July 2020.
- ^ "Evicel". U.S. Food and Drug Administration (FDA). 5 June 2017. Archived from the original on 22 July 2017. Retrieved 12 June 2020.