Diimines are organic compounds containing two imine (RCH=NR') groups. Common derivatives are 1,2- diketones and 1,3-diimines. These compounds are used as ligands and as precursors to heterocycles. Diimines are prepared by condensation reactions where a dialdehyde or diketone is treated with amine and water is eliminated. Similar methods are used to prepare Schiff bases and oximes.

The 1,2-diketimine ligands also called α-diimines and 1,4-diazabutadienes. They are derived from the condensation of 1,2-diketones and glyoxal with amines, often anilines. [1]
An example is glyoxal-bis(mesitylimine), a yellow solid that is synthesized by condensation of 2,4,6-trimethylaniline and glyoxal. [2] 2,2'-Bipyridine is a 1,2-diimine.
1,2-Diketimines are “ non-innocent ligands”, akin to the dithiolenes. [3]

For example, acetylacetone (2,4-pentanedione) and a primary alkyl- or arylamine will react, typically in acidified ethanol, to form a diketimine. 1,3-Diketimines are often referred to as H NacNac, a modification of the abbreviation Hacac for the conjugate acid of acetylacetone. These species form bidentate anionic ligands.
Substituted α-diimine ligands are useful in the preparation of post-metallocene catalysts for the polymerization and copolymerization of ethylene and alkenes. [6] [7]
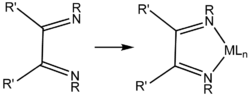
Diimines are precursors to NHC ligands by condensation with formaldehyde. [2]
- ^ Wang, F.; Chen, C. (2019). "A Continuing Legend: The Brookhart-Type α-Diimine Nickel and Palladium Catalysts". Polymer Chemistry. 10 (19): 2354–2369. doi: 10.1039/C9PY00226J.
- ^ a b Ison, Elon A.; Ison, Ana (2012). "Synthesis of Well-Defined Copper N-Heterocyclic Carbene Complexes and Their Use as Catalysts for a "Click Reaction": A Multistep Experiment That Emphasizes the Role of Catalysis in Green Chemistry". J. Chem. Educ. 89 (12): 1575–1577. Bibcode: 2012JChEd..89.1575I. doi: 10.1021/ed300243s.
- ^ Mashima, Kazushi (2020). "Redox-Active α-Diimine Complexes of Early Transition Metals: From Bonding to Catalysis". Bulletin of the Chemical Society of Japan. 93 (6): 799–820. doi: 10.1246/bcsj.20200056.
- ^ Haaf, Michael; Schmedake, Thomas A.; West, Robert (2000-10-01). "Stable Silylenes". Accounts of Chemical Research. 33 (10): 704–714. doi: 10.1021/ar950192g. ISSN 0001-4842. PMID 11041835.
- ^ Asay, Matthew; Jones, Cameron; Driess, Matthias (2011-02-09). "N-Heterocyclic Carbene Analogues with Low-Valent Group 13 and Group 14 Elements: Syntheses, Structures, and Reactivities of a New Generation of Multitalented Ligands†". Chemical Reviews. 111 (2): 354–396. doi: 10.1021/cr100216y. ISSN 0009-2665. PMID 21133370.
- ^ Ittel, S. D.; Johnson, L. K.; Brookhart, M. (2000). "Late-Metal Catalysts for Ethylene Homo- and Copolymerization". Chemical Reviews. 100 (4): 1169–1203. doi: 10.1021/cr9804644.
- ^ Guo, Lihua; Dai, Shengyu; Sui, Xuelin; Chen, Changle (2016). "Palladium and Nickel Catalyzed Chain Walking Olefin Polymerization and Copolymerization". ACS Catalysis. 6: 428–441. doi: 10.1021/acscatal.5b02426.
