
| |
| Names | |
|---|---|
|
Systematic IUPAC name
Dichloro[1,3-propanediylbis(diphenylphosphanuide-κP)]nickel | |
| Other names
1,3-bis(diphenylphosphino)propanenickel(II) chloride
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.132.628 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C27H26Cl2NiP2 | |
| Molar mass | 542.05 g·mol−1 |
| Appearance | Orange to red-orange powder |
| Melting point | 213 °C (415 °F; 486 K) |
| Insoluble | |
| Hazards | |
| GHS labelling: | |

 [1]
[1]
| |
| Danger [1] | |
| H315, H317, H319, H334, H335, H350 [1] | |
| P201, P261, P280, P305+P351+P338, P308+P313 [1] | |
| Safety data sheet (SDS) | External SDS |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Dichloro[1,3-bis(diphenylphosphino)propane]nickel a coordination complex with the formula NiCl2(dppp); where dppp is the diphosphine 1,3-bis(diphenylphosphino)propane. It is used as a catalyst in organic synthesis. The compound is a bright orange-red crystalline powder.
While the electronic and solid-state structure of the chloride congener is not known (due to low solubility in common analytical solvents), several studies have been carried out on the bromo and iodo derivatives. [2] The complexes display a temperature-dependent interconversion between square-planar and tetrahedral geometries (diamagnetic and paramagnetic) in polar organic solvents (Keq between 1-3.68, depending on the solvent and temperature). In contrast, dichloro(1,2-bis(diphenylphosphino)ethane)nickel adopts a static square-planar (diamagnetic) structure in solution.
NiCl2(dppp) is prepared by combining equal molar portions of nickel(II) chloride hexahydrate with 1,3-bis(diphenylphosphino)propane in 2-propanol. [3]
- Ni(H2O)6Cl2 + dppp → NiCl2(dppp) + 6 H2O
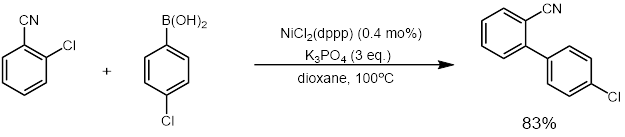
NiCl2(dppp) in an effective catalyst for coupling reactions such as the Kumada coupling [3] and Suzuki reactions (example below). [4] It also catalyzes other reactions that convert enol ethers, dithioacetals, and vinyl sulfides to olefins. [5] [6]
- ^ a b c d "1,3-Bis(diphenylphosphino)propane Nickel(II) Chloride". American Elements. Retrieved September 6, 2018.
- ^ Van Hecke, Gerald R.; Horrocks, Jr., William DeW. (1966). "Ditertiary Phosphine Complexes of Nickel. Spectral, Magnetic, and Proton Resonance Studies. A Planar-Tetrahedral Equilibrium". Inorganic Chemistry. 5 (11): 1968–1974. doi: 10.1021/ic50045a029.
- ^ a b Kumada, Makota; Tamao, Kohei; Sumitani, Koji (1978). "Phosphine-Nickel Complex Catalyzed Cross-Coupling of Grignard Reagents with Aryl and Alkenyl Halides: 1,2-Dibutylbenzene". Org. Synth. 58: 127. doi: 10.15227/orgsyn.058.0127.
- ^ Zhao, Yu-Long; Li, You; Li, Shui-Ming; Zhou, Yi-Guo; Sun, Feng-Yi; Gao, Lian-Xun; Han, Fu-She (1 June 2011). "A Highly Practical and Reliable Nickel Catalyst for Suzuki-Miyaura Coupling of Aryl Halides". Advanced Synthesis & Catalysis. 353 (9): 1543–1550. doi: 10.1002/adsc.201100101.
- ^ Tien-Yau Luh; Tien-Min Yuan. "Cross-Coupling Reactions". Encyclopedia of Reagents for Organic Synthesis doi: 10.1002/047084289X.rd100.pub2.
- ^ Ljungdahl, Thomas; Bennur, Timmanna; Dallas, Andrea; Emtenaes, Hans; Maartensson, Jerker (2008). "Two Competing Mechanisms for the Copper-Free Sonogashira Cross-Coupling Reaction". Organometallics. 27 (11): 2490–2498. doi: 10.1021/om800251s.