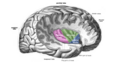
| Insular cortex | |
|---|---|
 Right insula, exposed by removing the
opercula | |
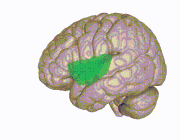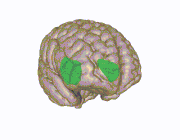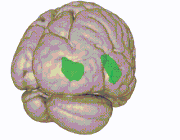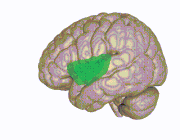 3D view of the insular cortex | |
| Details | |
| Part of | Cerebral cortex of brain |
| Artery | Middle cerebral |
| Identifiers | |
| Latin | cortex insularis |
| MeSH | D000087623 |
| NeuroNames | 111 |
| NeuroLex ID | birnlex_1117 |
| TA98 |
A14.1.09.149 A12.2.07.053 |
| TA2 | 5502 |
| FMA | 67329 |
| Anatomical terms of neuroanatomy | |
The insular cortex (also insula and insular lobe) is a portion of the cerebral cortex folded deep within the lateral sulcus (the fissure separating the temporal lobe from the parietal and frontal lobes) within each hemisphere of the mammalian brain.
The insulae are believed to be involved in consciousness and play a role in diverse functions usually linked to emotion or the regulation of the body's homeostasis. These functions include compassion, empathy, taste, perception, motor control, self-awareness, cognitive functioning, interpersonal relationships, and awareness of homeostatic emotions such as hunger, pain and fatigue. In relation to these, it is involved in psychopathology.
The insular cortex is divided into two parts: the anterior insula and the posterior insula in which more than a dozen field areas have been identified. The cortical area overlying the insula toward the lateral surface of the brain is the operculum (meaning lid). The opercula are formed from parts of the enclosing frontal, temporal, and parietal lobes.
Structure
Connections
The anterior part of the insula is subdivided by shallow sulci into three or four short gyri.
The anterior insula receives a direct projection from the basal part of the ventral medial nucleus of the thalamus and a particularly large input from the central nucleus of the amygdala. In addition, the anterior insula itself projects to the amygdala.
One study on rhesus monkeys revealed widespread reciprocal connections between the insular cortex and almost all subnuclei of the amygdaloid complex. The posterior insula projects predominantly to the dorsal aspect of the lateral and to the central amygdaloid nuclei. In contrast, the anterior insula projects to the anterior amygdaloid area as well as the medial, the cortical, the accessory basal magnocellular, the medial basal, and the lateral amygdaloid nuclei. [1]
The posterior part of the insula is formed by a long gyrus.
The posterior insula connects reciprocally with the secondary somatosensory cortex and receives input from spinothalamically activated ventral posterior inferior thalamic nuclei. It has also been shown that this region receives inputs from the ventromedial nucleus (posterior part) of the thalamus that are highly specialized to convey homeostatic information such as pain, temperature, itch, local oxygen status, and sensual touch. [2]
A human neuroimaging study using diffusion tensor imaging revealed that the anterior insula is interconnected to regions in the temporal and occipital lobe, opercular and orbitofrontal cortex, triangular and opercular parts of the inferior frontal gyrus. The same study revealed differences in the anatomical connection patterns between the left and right hemisphere. [3]
The 'circular sulcus of insula' (or sulcus of Reil [4]) is a semi-circular sulcus or fissure [4] that separates the insula from the neighboring gyri of the operculum [5] in the front, above, and behind. [4]
Cytoarchitecture
The insular cortex has regions of variable cell structure or cytoarchitecture, changing from granular in the posterior portion to agranular in the anterior portion. The insula also receives differential cortical and thalamic input along its length. The anterior insular cortex contains a population of spindle neurons (also called von Economo neurons), identified as characterising a distinctive subregion as the agranular frontal insula. [6]
Development
The insular cortex is considered a separate lobe of the telencephalon by some authorities. [7] Other sources see the insula as a part of the temporal lobe. [8] It is also sometimes grouped with limbic structures deep in the brain into a limbic lobe.[ citation needed] As a paralimbic cortex, the insular cortex is considered to be a relatively old structure.
Function
Multimodal sensory processing, sensory binding
Functional imaging studies show activation of the insula during audio-visual integration tasks. [9] [10]
Taste
The anterior insula is part of the primary gustatory cortex. [11] [12] Research in rhesus monkeys has also reported that apart from numerous taste-sensitive neurons, the insular cortex also responds to non-taste properties of oral stimuli related to the texture (viscosity, grittiness) or temperature of food. [13]
Speech
The sensory speech region, Wernicke’s area, and the motor speech region, Broca’s area, are interconnected by a large axonal fiber system known as the arcuate fasciculus which passes directly beneath the insular cortex. On account of this anatomical architecture, ischemic strokes in the insular region can disrupt the arcuate fasciculus. [14] Functional imaging studies on the cerebral correlates of language production also suggest that the anterior insula forms part of the brain network of speech motor control. [15] Moreover, electrical stimulation of the posterior insular can evoke speech disturbances such as speech arrest and reduced voice intensity. [16]
Lesion of the pre-central gyrus of the insula can also cause “pure speech apraxia” (i.e. the inability to speak with no apparent aphasic or orofacial motor impairments). [17] This demonstrates that the insular cortex forms part of a critical circuit for the coordination of complex articulatory movements prior to and during the execution of the motor speech plans. [17] Importantly, this specific cortical circuit is different from those that relate to the cognitive aspects of language production (e.g., Broca’s area on the inferior frontal gyrus). [17] Subvocal, or silent, speech has also been shown to activate right insular cortex, further supporting the theory that the motor control of speech proceeds from the insula. [18]
Interoceptive awareness
There is evidence that, in addition to its base functions, the insula may play a role in certain higher-level functions that operate only in humans and other great apes. The spindle neurons found at a higher density in the right frontal insular cortex are also found in the anterior cingulate cortex, which is another region that has reached a high level of specialization in great apes. It has been speculated that these neurons are involved in cognitive- emotional processes that are specific to primates including great apes, such as empathy and metacognitive emotional feelings. This is supported by functional imaging results showing that the structure and function of the right frontal insula is correlated with the ability to feel one's own heartbeat, or to empathize with the pain of others. It is thought that these functions are not distinct from the lower-level functions of the insula but rather arise as a consequence of the role of the insula in conveying homeostatic information to consciousness. [19] [20] The right anterior insula is engaged in interoceptive awareness of homeostatic emotions such as thirst, pain and fatigue, [21] and the ability to time one's own heartbeat. Moreover, greater right anterior insular gray matter volume correlates with increased accuracy in this subjective sense of the inner body, and with negative emotional experience. [22] It is also involved in the control of blood pressure, [23] in particular during and after exercise, [23] and its activity varies with the amount of effort a person believes he/she is exerting. [24] [25]
The insular cortex also is where the sensation of pain is judged as to its degree. [26] Lesion of the insula is associated with dramatic loss of pain perception and isolated insular infarction can lead to contralateral elimination of pinprick perception. [27] Further, the insula is where a person imagines pain when looking at images of painful events while thinking about their happening to one's own body. [28] Those with irritable bowel syndrome have abnormal processing of visceral pain in the insular cortex related to dysfunctional inhibition of pain within the brain. [29]
Physiological studies in rhesus monkeys have shown that neurons in the insula respond to skin stimulation. [30] PET studies have also revealed that the human insula can also be activated by vibrational stimulation to the skin. [31]
Another perception of the right anterior insula is the degree of nonpainful warmth [32] or nonpainful coldness [33] of a skin sensation. Other internal sensations processed by the insula include stomach or abdominal distension. [34] [35] A full bladder also activates the insular cortex. [36]
One brain imaging study suggests that the unpleasantness of subjectively perceived dyspnea is processed in the right human anterior insula and amygdala. [37]
The cerebral cortex processing vestibular sensations extends into the insula, [38] with small lesions in the anterior insular cortex being able to cause loss of balance and vertigo. [39]
Other noninteroceptive perceptions include passive listening to music, [40] laughter and crying, [41] empathy and compassion, [42] and language. [43]
Motor control
In motor control, it contributes to hand-and-eye motor movement, [44] [45] swallowing, [46] gastric motility, [47] and speech articulation. [48] [49] It has been identified as a "central command” centre that ensures that heart rate and blood pressure increase at the onset of exercise. [50] Research upon conversation links it to the capacity for long and complex spoken sentences. [51] It is also involved in motor learning [52] and has been identified as playing a role in the motor recovery from stroke. [53]
Homeostasis
It plays a role in a variety of homeostatic functions related to basic survival needs, such as taste, visceral sensation, and autonomic control. The insula controls autonomic functions through the regulation of the sympathetic and parasympathetic systems. [54] [55] It has a role in regulating the immune system. [56] [57] [58]
Self
The insula has been identified as playing a role in the experience of bodily self-awareness, [59] [60] sense of agency, [61] and sense of body ownership. [62]
Social emotions
The anterior insula processes a person's sense of disgust both to smells [63] and to the sight of contamination and mutilation [64] — even when just imagining the experience. [65] This associates with a mirror neuron-like link between external and internal experiences.
In social experience, it is involved in the processing of norm violations, [66] emotional processing, [67] empathy, [68] and orgasms. [69]
The insula is active during social decision making. Tiziana Quarto et al. measured emotional intelligence (EI) (the ability to identify, regulate, and process emotions of themselves and of others) of sixty-three healthy subjects. Using fMRI EI was measured in correlation with left insular activity. The subjects were shown various pictures of facial expressions and tasked with deciding to approach or avoid the person in the picture. The results of the social decision task yielded that individuals with high EI scores had left insular activation when processing fearful faces. Individuals with low EI scores had left insular activation when processing angry faces. [70]
Emotions
The insular cortex, in particular its most anterior portion, is considered a limbic-related cortex. The insula has increasingly become the focus of attention for its role in body representation and subjective emotional experience. In particular, Antonio Damasio has proposed that this region plays a role in mapping visceral states that are associated with emotional experience, giving rise to conscious feelings. This is in essence a neurobiological formulation of the ideas of William James, who first proposed that subjective emotional experience (i.e., feelings) arise from our brain's interpretation of bodily states that are elicited by emotional events. This is an example of embodied cognition.
In terms of function, the insula is believed to process convergent information to produce an emotionally relevant context for sensory experience. To be specific, the anterior insula is related more to olfactory, gustatory, viscero-autonomic, and limbic function, whereas the posterior insula is related more to auditory-somesthetic-skeletomotor function. Functional imaging experiments have revealed that the insula has an important role in pain experience and the experience of a number of basic emotions, including anger, fear, disgust, happiness, and sadness. [71]
The anterior insular cortex (AIC) is believed to be responsible for emotional feelings, including maternal and romantic love, anger, fear, sadness, happiness, sexual arousal, disgust, aversion, unfairness, inequity, indignation, uncertainty, [72] disbelief, social exclusion, trust, empathy, sculptural beauty, a ‘state of union with God’, and hallucinogenic states. [73]
Functional imaging studies have also implicated the insula in conscious desires, such as food craving and drug craving. What is common to all of these emotional states is that they each change the body in some way and are associated with highly salient subjective qualities. The insula is well-situated for the integration of information relating to bodily states into higher-order cognitive and emotional processes. The insula receives information from "homeostatic afferent" sensory pathways via the thalamus and sends output to a number of other limbic-related structures, such as the amygdala, the ventral striatum, and the orbitofrontal cortex, as well as to motor cortices. [74]
A study using magnetic resonance imaging found that the right anterior insula is significantly thicker in people that meditate. [75] Other research into brain activity and meditation has shown an increase in grey matter in areas of the brain including the insular cortex. [76]
Another study using voxel-based morphometry and MRI on experienced Vipassana meditators was done to extend the findings of Lazar et al., which found increased grey matter concentrations in this and other areas of the brain in experienced meditators. [77]
The strongest evidence against a causative role for the insula cortex in emotion comes from Damasio et al. (2012) [78] which showed that a patient who suffered bilateral lesions of the insula cortex expressed the full complement of human emotions, and was fully capable of emotional learning.
Salience
Functional neuroimaging research suggests the insula is involved in two types of salience. Interoceptive information processing that links interoception with emotional salience to generate a subjective representation of the body. This involves, first, the anterior insular cortex with the pregenual anterior cingulate cortex ( Brodmann area 33) and the anterior and posterior mid-cingulate cortices, and, second, a general salience network concerned with environmental monitoring, response selection, and skeletomotor body orientation that involves all of the insular cortex and the mid-cingulate cortex. [79] A related idea is that the anterior insula, as part of the salience network, interacts with the mid-posterior insula to combine salient stimuli with autonomic information, leading to a high state of physiological awareness of salient stimuli. [80]
An alternative or perhaps complementary proposal is that the right anterior insular regulates the interaction between the salience of the selective attention created to achieve a task (the dorsal attention system) and the salience of arousal created to keep focused upon the relevant part of the environment (ventral attention system). [81] This regulation of salience might be particularly important during challenging tasks where attention might fatigue and so cause careless mistakes but if there is too much arousal it risks creating poor performance by turning into anxiety. [81]
Decision Making
Studies have shown that damage or dysfunction in the insular cortex can impair decision-making, emotional regulation, and social behavior. The insula is considered a key brain structure in the neural circuitry underlying complex decision-making processes. [82] It plays a significant role in integrating internal and external cues to facilitate adaptive choices.
Auditory perception
Recent research indicates that the insular cortex is involved in auditory perception. Responses to sound stimuli were obtained using intracranial EEG recordings acquired from patients with epilepsy. The posterior part of the insula showed auditory responses that resemble those observed in Heschl’s gyrus, whereas the anterior part responded to the emotional contents of the auditory stimuli. [83] Clinical data additionally shows that bilateral damage to the insula after ischemic injury or trauma can lead to auditory agnosia. [84] Functional magnetic resonance studies have also demonstrated that the insular cortex participates in many key auditory processes such as tuning into novel auditory stimuli and allocating auditory attention. [85]
Direct recordings from the posterior part of the insula showed responses to unexpected sounds within regular auditory streams, a process known as auditory deviance detection. Researchers observed a mismatch negativity (MMN) potential, a well known event related potential, as well as the high frequency activity signals originating from local neurons. [86]
Simple auditory illusions and hallucinations were elicited by electrical functional mapping. [87] [83]
Clinical significance
Progressive expressive aphasia
Progressive expressive aphasia is the deterioration of normal language function that causes individuals to lose the ability to communicate fluently while still being able to comprehend single words and intact other non-linguistic cognition. It is found in a variety of degenerative neurological conditions including Pick's disease, motor neuron disease, corticobasal degeneration, frontotemporal dementia, and Alzheimer's disease. It is associated with hypometabolism [88] and atrophy of the left anterior insular cortex. [89]
Addiction
A number of functional brain imaging studies have shown that the insular cortex is activated when drug users are exposed to environmental cues that trigger cravings. This has been shown for a variety of drugs, including cocaine, alcohol, opiates, and nicotine. Despite these findings, the insula has been ignored within the drug addiction literature, perhaps because it is not known to be a direct target of the mesocortical dopamine system, which is central to current dopamine reward theories of addiction. Research published in 2007 [90] has shown that cigarette smokers suffering damage to the insular cortex, from a stroke for instance, have their addiction to cigarettes practically eliminated. These individuals were found to be up to 136 times more likely to undergo a disruption of smoking addiction than smokers with damage in other areas. Disruption of addiction was evidenced by self-reported behavior changes such as quitting smoking less than one day after the brain injury, quitting smoking with great ease, not smoking again after quitting, and having no urge to resume smoking since quitting. The study was conducted on average eight years after the strokes, which opens up the possibility that recall bias could have affected the results. [91] More recent prospective studies, which overcome this limitation, have corroborated these findings [92] [93] This suggests a significant role for the insular cortex in the neurological mechanisms underlying addiction to nicotine and other drugs, and would make this area of the brain a possible target for novel anti-addiction medication. In addition, this finding suggests that functions mediated by the insula, especially conscious feelings, may be particularly important for maintaining drug addiction, although this view is not represented in any modern research or reviews of the subject. [94]
A recent study in rats by Contreras et al. [95] corroborates these findings by showing that reversible inactivation of the insula disrupts amphetamine conditioned place preference, an animal model of cue-induced drug craving. In this study, insula inactivation also disrupted "malaise" responses to lithium chloride injection, suggesting that the representation of negative interoceptive states by the insula plays a role in addiction. However, in this same study, the conditioned place preference took place immediately after the injection of amphetamine, suggesting that it is the immediate, pleasurable interoceptive effects of amphetamine administration, rather than the delayed, aversive effects of amphetamine withdrawal that are represented within the insula.
A model proposed by Naqvi et al. (see above) is that the insula stores a representation of the pleasurable interoceptive effects of drug use (e.g., the airway sensory effects of nicotine, the cardiovascular effects of amphetamine), and that this representation is activated by exposure to cues that have previously been associated with drug use. A number of functional imaging studies have shown the insula to be activated during the administration of addictive psychoactive drugs. Several functional imaging studies have also shown that the insula is activated when drug users are exposed to drug cues, and that this activity is correlated with subjective urges. In the cue-exposure studies, insula activity is elicited when there is no actual change in the level of drug in the body. Therefore, rather than merely representing the interoceptive effects of drug use as it occurs, the insula may play a role in memory for the pleasurable interoceptive effects of past drug use, anticipation of these effects in the future, or both. Such a representation may give rise to conscious urges that feel as if they arise from within the body. This may make addicts feel as if their bodies need to use a drug, and may result in persons with lesions in the insula reporting that their bodies have forgotten the urge to use, according to this study.
Subjective certainty in ecstatic seizures
A common quality in mystical experiences is a strong feeling of certainty which cannot be expressed in words. Fabienne Picard proposes a neurological explanation for this subjective certainty, based on clinical research of epilepsy. [96] [97] According to Picard, this feeling of certainty may be caused by a dysfunction of the anterior insula, a part of the brain which is involved in interoception, self-reflection, and in avoiding uncertainty about the internal representations of the world by "anticipation of resolution of uncertainty or risk". This avoidance of uncertainty functions through the comparison between predicted states and actual states, that is, "signaling that we do not understand, i.e., that there is ambiguity." [98] Picard notes that "the concept of insight is very close to that of certainty," and refers to Archimedes' "Eureka!" [99] [100] Picard hypothesizes that during ecstatic seizures the comparison between predicted states and actual states no longer functions, and that mismatches between predicted state and actual state are no longer processed, blocking " negative emotions and negative arousal arising from predictive uncertainty," which will be experienced as emotional confidence. [101] Picard concludes that "[t]his could lead to a spiritual interpretation in some individuals." [101]
Other clinical conditions
The insular cortex has been suggested to have a role in anxiety disorders, [102] emotion dysregulation, [103] and anorexia nervosa. [104]
History
The insula was first described by Johann Christian Reil while describing cranial and spinal nerves and plexuses. [105] Henry Gray in Gray's Anatomy is responsible for it being known as the Island of Reil. [105] John Allman and colleagues showed that anterior insular cortex contains spindle neurons.
Additional images
-
Location and structure of the insular cortex
-
Coronal section of brain immediately in front of pons (Insula labeled at upper right)
-
Horizontal section of left cerebral hemisphere
-
Insular cortex highlighted in green on coronal T1 MRI images
-
Insular cortex highlighted in green on sagittal T1 MRI images
-
Insular cortex highlighted in green on transversal T1 MRI images
See also
References
- ^ MUFSON, E; MESULAM, M; PANDYA, D (1 July 1981). "Insular interconnections with the amygdala in the rhesus monkey". Neuroscience. 6 (7): 1231–1248. doi: 10.1016/0306-4522(81)90184-6. PMID 6167896. S2CID 46366616.
- ^ Craig AD, Chen K, Bandy D, Reiman EM (2000). "Thermosensory activation of insular cortex". Nat. Neurosci. 3 (2): 184–90. doi: 10.1038/72131. PMID 10649575. S2CID 7077496.
- ^ JAKAB, A; MOLNAR, P; BOGNER, P; BERES, M; BERENYI, E (1 Oct 2011). "Connectivity-based parcellation reveals interhemispheric differences in the insula". Brain Topography. 25 (3): 264–271. doi: 10.1007/s10548-011-0205-y. PMID 22002490. S2CID 12293575.
- ^ a b c Johannes Sobotta. "Sobotta's Atlas and Text-book of human anatomy 1909". p. 145. Retrieved November 10, 2013.
- ^ "Definition: 'Circular Sulcus Of Insula'". MediLexicon. Archived from the original on 2013-06-04. Retrieved 2012-03-30.
- ^ Bauernfeind A; et al. (April 2013). "A volumetric comparison of the insular cortex and its subregions in primates". Human Evolution. 64 (4): 263–279. doi: 10.1016/j.jhevol.2012.12.003. PMC 3756831. PMID 23466178.
- ^ Brain, MSN Encarta. Archived 2009-10-31.
- ^ Kolb, Bryan; Whishaw, Ian Q. (2003). Fundamentals of human neuropsychology (5th ed.). [New York]: Worth. ISBN 978-0-7167-5300-1.
- ^ Bushara, KO; Grafman, J; Hallett, M (1 January 2001). "Neural correlates of auditory-visual stimulus onset asynchrony detection". The Journal of Neuroscience. 21 (1): 300–4. doi: 10.1523/JNEUROSCI.21-01-00300.2001. PMC 6762435. PMID 11150347.
- ^ Bushara, KO; Hanakawa, T; Immisch, I; Toma, K; Kansaku, K; Hallett, M (February 2003). "Neural correlates of cross-modal binding". Nature Neuroscience. 6 (2): 190–5. doi: 10.1038/nn993. PMID 12496761. S2CID 1098979.
- ^ Marieb, Elaine N.; Hoehn, Katja (2008). Anatomy & Physiology, Third Edition. Boston: Benjamin Cummings/Pearson. pp. 391–395. ISBN 978-0-8053-0094-9.
- ^ Pritchard, TC; Macaluso, DA; Eslinger, PJ (August 1999). "Taste perception in patients with insular cortex lesions". Behavioral Neuroscience. 113 (4): 663–71. doi: 10.1037/0735-7044.113.4.663. PMID 10495075.
- ^ Verhagen, Justus V.; Kadohisa, Mikiko; Rolls, Edmund T. (September 2004). "Primate Insular/Opercular Taste Cortex: Neuronal Representations of the Viscosity, Fat Texture, Grittiness, Temperature, and Taste of Foods". Journal of Neurophysiology. 92 (3): 1685–1699. doi: 10.1152/jn.00321.2004. ISSN 0022-3077. PMID 15331650.
-
^
https://academic.oup.com/brain/article-abstract/103/2/337/378338. Retrieved 2023-12-11.
{{ cite web}}: Missing or empty|title=( help) - ^ Bohland, Jason W.; Guenther, Frank H. (2006-08-15). "An fMRI investigation of syllable sequence production". NeuroImage. 32 (2): 821–841. doi: 10.1016/j.neuroimage.2006.04.173. ISSN 1053-8119. PMID 16730195. S2CID 9909543.
- ^ Afif, Afif; Minotti, Lorella; Kahane, Philippe; Hoffmann, Dominique (November 2010). "Anatomofunctional organization of the insular cortex: A study using intracerebral electrical stimulation in epileptic patients". Epilepsia. 51 (11): 2305–2315. doi: 10.1111/j.1528-1167.2010.02755.x. ISSN 0013-9580. PMID 20946128.
- ^
a
b
c
https://academic.oup.com/cercor/article/31/8/3723/6213947. Retrieved 2023-12-11.
{{ cite web}}: Missing or empty|title=( help) - ^ Kato, Yutaka; Muramatsu, Taro; Kato, Motoichiro; Shintani, Masuro; Kashima, Haruo (2007-03-26). "Activation of right insular cortex during imaginary speech articulation". NeuroReport. 18 (5): 505–509. doi: 10.1097/WNR.0b013e3280586862. ISSN 0959-4965. PMID 17496812. S2CID 2040545.
- ^ Benedetto De Martino; Dharshan Kumaran; Ben Seymour; Raymond J. Dolan (August 2006). "Frames, Biases, and Rational Decision-Making in the Human Brain". Science. 313 (6): 684–687. Bibcode: 2006Sci...313..684D. doi: 10.1126/science.1128356. PMC 2631940. PMID 16888142.
- ^ Gui Xue; Zhonglin Lu; Irwin P. Levin d; Antoine Bechara (2010). "The impact of prior risk experiences on subsequent risky decision-making: The role of the insula". NeuroImage. 50 (2): 709–716. doi: 10.1016/j.neuroimage.2009.12.097. PMC 2828040. PMID 20045470.
- ^ Emeran A. Mayer (August 2011). "Gut feelings: the emerging biology of gut–brain communication". Nature Reviews Neuroscience. 12 (8): 453–466. doi: 10.1038/nrn3071. PMC 3845678. PMID 21750565.
- ^ Critchley HD, Wiens S, Rotshtein P, Ohman A, Dolan RJ (February 2004). "Neural systems supporting interoceptive awareness". Nat. Neurosci. 7 (2): 189–95. doi: 10.1038/nn1176. hdl: 21.11116/0000-0001-A2FB-D. PMID 14730305. S2CID 13344271.
- ^ a b Lamb K, Gallagher K, McColl R, Mathews D, Querry R, Williamson JW (April 2007). "Exercise-induced decrease in insular cortex rCBF during postexercise hypotension". Med Sci Sports Exerc. 39 (4): 672–9. doi: 10.1249/mss.0b013e31802f04e0. PMID 17414805.
- ^ Williamson JW, McColl R, Mathews D, Mitchell JH, Raven PB, Morgan WP (April 2001). "Hypnotic manipulation of effort sense during dynamic exercise: cardiovascular responses and brain activation". J. Appl. Physiol. 90 (4): 1392–9. doi: 10.1152/jappl.2001.90.4.1392. PMID 11247939. S2CID 8653997.
- ^ Williamson JW, McColl R, Mathews D, Ginsburg M, Mitchell JH (September 1999). "Activation of the insular cortex is affected by the intensity of exercise". J. Appl. Physiol. 87 (3): 1213–9. CiteSeerX 10.1.1.492.2730. doi: 10.1152/jappl.1999.87.3.1213. PMID 10484598. S2CID 1078691.
- ^ Baliki MN, Geha PY, Apkarian AV (February 2009). "Parsing pain perception between nociceptive representation and magnitude estimation". J. Neurophysiol. 101 (2): 875–87. doi: 10.1152/jn.91100.2008. PMC 3815214. PMID 19073802.
- ^ Birklein, Frank; Rolke, Roman; Müller-Forell, Wibke (2005-11-08). "Isolated insular infarction eliminates contralateral cold, cold pain, and pinprick perception". Neurology. 65 (9): 1381. doi: 10.1212/01.wnl.0000181351.82772.b3. ISSN 0028-3878. PMID 16275823.
- ^ Ogino Y, Nemoto H, Inui K, Saito S, Kakigi R, Goto F (May 2007). "Inner experience of pain: imagination of pain while viewing images showing painful events forms subjective pain representation in human brain". Cereb. Cortex. 17 (5): 1139–46. doi: 10.1093/cercor/bhl023. PMID 16855007.
- ^ Song GH, Venkatraman V, Ho KY, Chee MW, Yeoh KG, Wilder-Smith CH (December 2006). "Cortical effects of anticipation and endogenous modulation of visceral pain assessed by functional brain MRI in irritable bowel syndrome patients and healthy controls". Pain. 126 (1–3): 79–90. doi: 10.1016/j.pain.2006.06.017. PMID 16846694. S2CID 21437784.
- ^ Schneider, Richard J.; Friedman, David P.; Mishkin, Mortimer (1993-09-03). "A modality-specific somatosensory area within the insula of the rhesus monkey". Brain Research. 621 (1): 116–120. doi: 10.1016/0006-8993(93)90305-7. ISSN 0006-8993. PMID 8221062. S2CID 20207990.
- ^ Burton, H.; Videen, T. O.; Raichle, M. E. (January 1993). "Tactile-Vibration-Activated Foci in Insular and Parietal-Opercular Cortex Studied with Positron Emission Tomography: Mapping the Second Somatosensory Area in Humans". Somatosensory & Motor Research. 10 (3): 297–308. doi: 10.3109/08990229309028839. ISSN 0899-0220. PMID 8237217.
- ^ Olausson H, Charron J, Marchand S, Villemure C, Strigo IA, Bushnell MC (November 2005). "Feelings of warmth correlate with neural activity in right anterior insular cortex". Neurosci. Lett. 389 (1): 1–5. doi: 10.1016/j.neulet.2005.06.065. PMID 16051437. S2CID 20068852.
- ^ Craig AD, Chen K, Bandy D, Reiman EM (February 2000). "Thermosensory activation of insular cortex". Nat. Neurosci. 3 (2): 184–90. doi: 10.1038/72131. PMID 10649575. S2CID 7077496.
- ^ Ladabaum U, Minoshima S, Hasler WL, Cross D, Chey WD, Owyang C (February 2001). "Gastric distention correlates with activation of multiple cortical and subcortical regions". Gastroenterology. 120 (2): 369–76. doi: 10.1053/gast.2001.21201. PMID 11159877.
- ^ Hamaguchi T, Kano M, Rikimaru H, et al. (June 2004). "Brain activity during distention of the descending colon in humans". Neurogastroenterol. Motil. 16 (3): 299–309. doi: 10.1111/j.1365-2982.2004.00498.x. PMID 15198652. S2CID 20437580.[ dead link]
- ^ Matsuura S, Kakizaki H, Mitsui T, Shiga T, Tamaki N, Koyanagi T (November 2002). "Human brain region response to distention or cold stimulation of the bladder: a positron emission tomography study". J. Urol. 168 (5): 2035–9. doi: 10.1016/s0022-5347(05)64290-5. PMID 12394703.
- ^ von Leupoldt, A.; Sommer, T.; Kegat, S.; Baumann, H. J.; Klose, H.; Dahme, B.; Buchel, C. (24 January 2008). "The Unpleasantness of Perceived Dyspnea Is Processed in the Anterior Insula and Amygdala". American Journal of Respiratory and Critical Care Medicine. 177 (9): 1026–1032. doi: 10.1164/rccm.200712-1821OC. PMID 18263796.[ permanent dead link]
- ^ Kikuchi M, Naito Y, Senda M, et al. (April 2009). "Cortical activation during optokinetic stimulation — an fMRI study". Acta Otolaryngol. 129 (4): 440–3. doi: 10.1080/00016480802610226. PMID 19116795. S2CID 42990194.
- ^ Papathanasiou ES, Papacostas SS, Charalambous M, Eracleous E, Thodi C, Pantzaris M (2006). "Vertigo and imbalance caused by a small lesion in the anterior insula". Electromyogr Clin Neurophysiol. 46 (3): 185–92. PMID 16918202.
- ^ Brown S, Martinez MJ, Parsons LM (September 2004). "Passive music listening spontaneously engages limbic and paralimbic systems". NeuroReport. 15 (13): 2033–7. doi: 10.1097/00001756-200409150-00008. PMID 15486477. S2CID 12308683.
- ^ Sander K, Scheich H (October 2005). "Left auditory cortex and amygdala, but right insula dominance for human laughing and crying". J Cogn Neurosci. 17 (10): 1519–31. doi: 10.1162/089892905774597227. PMID 16269094. S2CID 9509954.
- ^ "Interview with Tania Singer | the Center for Compassion and Altruism Research and Education". Archived from the original on 2010-07-14. Retrieved 2010-07-04.
- ^ Bamiou DE, Musiek FE, Luxon LM (May 2003). "The insula (Island of Reil) and its role in auditory processing. Literature review". Brain Res. Brain Res. Rev. 42 (2): 143–54. doi: 10.1016/S0165-0173(03)00172-3. PMID 12738055. S2CID 22339177.
- ^ Anderson TJ, Jenkins IH, Brooks DJ, Hawken MB, Frackowiak RS, Kennard C (October 1994). "Cortical control of saccades and fixation in man. A PET study". Brain. 117 (Pt 5): 1073–84. doi: 10.1093/brain/117.5.1073. PMID 7953589.
- ^ Fink GR, Frackowiak RS, Pietrzyk U, Passingham RE (April 1997). "Multiple nonprimary motor areas in the human cortex". J. Neurophysiol. 77 (4): 2164–74. doi: 10.1152/jn.1997.77.4.2164. PMID 9114263. S2CID 15881491.
- ^ Sörös P, Inamoto Y, Martin RE (August 2009). "Functional brain imaging of swallowing: an activation likelihood estimation meta-analysis". Hum Brain Mapp. 30 (8): 2426–39. doi: 10.1002/hbm.20680. PMC 6871071. PMID 19107749. S2CID 15438676.
- ^ Penfield W, Faulk ME (1955). "The insula; further observations on its function". Brain. 78 (4): 445–70. doi: 10.1093/brain/78.4.445. PMID 13293263.
- ^ Dronkers NF (November 1996). "A new brain region for coordinating speech articulation". Nature. 384 (6605): 159–61. Bibcode: 1996Natur.384..159D. doi: 10.1038/384159a0. PMID 8906789. S2CID 4305696.
- ^ Ackermann H, Riecker A (May 2004). "The contribution of the insula to motor aspects of speech production: a review and a hypothesis". Brain Lang. 89 (2): 320–8. doi: 10.1016/S0093-934X(03)00347-X. PMID 15068914. S2CID 36867434.
- ^ Nowak M, Holm S, Biering-Sørensen F, Secher NH, Friberg L (June 2005). ""Central command" and insular activation during attempted foot lifting in paraplegic humans". Hum Brain Mapp. 25 (2): 259–65. doi: 10.1002/hbm.20097. PMC 6871668. PMID 15849712.
- ^ Borovsky A, Saygin AP, Bates E, Dronkers N (June 2007). "Lesion correlates of conversational speech production deficits". Neuropsychologia. 45 (11): 2525–33. doi: 10.1016/j.neuropsychologia.2007.03.023. PMC 5610916. PMID 17499317.
- ^ Mutschler I, Schulze-Bonhage A, Glauche V, Demandt E, Speck O, Ball T (2007). Fitch T (ed.). "A rapid sound-action association effect in human insular cortex". PLOS ONE. 2 (2): e259. Bibcode: 2007PLoSO...2..259M. doi: 10.1371/journal.pone.0000259. PMC 1800344. PMID 17327919.
- ^ Weiller C, Ramsay SC, Wise RJ, Friston KJ, Frackowiak RS (February 1993). "Individual patterns of functional reorganization in the human cerebral cortex after capsular infarction". Annals of Neurology. 33 (2): 181–9. doi: 10.1002/ana.410330208. PMID 8434880. S2CID 25131597.
- ^ Oppenheimer SM, Gelb A, Girvin JP, Hachinski VC (September 1992). "Cardiovascular effects of human insular cortex stimulation". Neurology. 42 (9): 1727–32. doi: 10.1212/wnl.42.9.1727. PMID 1513461. S2CID 32371468.
- ^ Critchley HD (December 2005). "Neural mechanisms of autonomic, affective, and cognitive integration". J. Comp. Neurol. 493 (1): 154–66. doi: 10.1002/cne.20749. PMID 16254997. S2CID 32616395.
- ^ Pacheco-López G, Niemi MB, Kou W, Härting M, Fandrey J, Schedlowski M (March 2005). "Neural substrates for behaviorally conditioned immunosuppression in the rat". J. Neurosci. 25 (9): 2330–7. doi: 10.1523/JNEUROSCI.4230-04.2005. PMC 6726099. PMID 15745959.
- ^ Ramírez-Amaya V, Alvarez-Borda B, Ormsby CE, Martínez RD, Pérez-Montfort R, Bermúdez-Rattoni F (June 1996). "Insular cortex lesions impair the acquisition of conditioned immunosuppression". Brain Behav. Immun. 10 (2): 103–14. doi: 10.1006/brbi.1996.0011. PMID 8811934. S2CID 24813018.
- ^ Ramírez-Amaya V, Bermúdez-Rattoni F (March 1999). "Conditioned enhancement of antibody production is disrupted by insular cortex and amygdala but not hippocampal lesions". Brain Behav. Immun. 13 (1): 46–60. doi: 10.1006/brbi.1998.0547. PMID 10371677. S2CID 20527835.
- ^ Karnath HO, Baier B, Nägele T (August 2005). "Awareness of the functioning of one's own limbs mediated by the insular cortex?". J. Neurosci. 25 (31): 7134–8. doi: 10.1523/JNEUROSCI.1590-05.2005. PMC 6725240. PMID 16079395.
- ^ Craig AD (January 2009). "How do you feel—now? The anterior insula and human awareness". Nature Reviews Neuroscience. 10 (1): 59–70. doi: 10.1038/nrn2555. PMID 19096369. S2CID 2340032.
- ^ Farrer C, Frith CD (March 2002). "Experiencing oneself vs another person as being the cause of an action: the neural correlates of the experience of agency". NeuroImage. 15 (3): 596–603. doi: 10.1006/nimg.2001.1009. PMID 11848702. S2CID 768408.
- ^ Tsakiris M, Hesse MD, Boy C, Haggard P, Fink GR (October 2007). "Neural signatures of body ownership: a sensory network for bodily self-consciousness". Cereb. Cortex. 17 (10): 2235–44. doi: 10.1093/cercor/bhl131. PMID 17138596.
- ^ Wicker B, Keysers C, Plailly J, Royet JP, Gallese V, Rizzolatti G (October 2003). "Both of us disgusted in My insula: the common neural basis of seeing and feeling disgust". Neuron. 40 (3): 655–64. doi: 10.1016/S0896-6273(03)00679-2. PMID 14642287. S2CID 766157.
- ^ Wright P, He G, Shapira NA, Goodman WK, Liu Y (October 2004). "Disgust and the insula: fMRI responses to pictures of mutilation and contamination". NeuroReport. 15 (15): 2347–51. doi: 10.1097/00001756-200410250-00009. PMID 15640753. S2CID 6864309.
- ^ Jabbi M, Bastiaansen J, Keysers C (2008). Lauwereyns J (ed.). "A common anterior insula representation of disgust observation, experience and imagination shows divergent functional connectivity pathways". PLOS ONE. 3 (8): e2939. Bibcode: 2008PLoSO...3.2939J. doi: 10.1371/journal.pone.0002939. PMC 2491556. PMID 18698355.
- ^ Sanfey AG, Rilling JK, Aronson JA, Nystrom LE, Cohen JD (June 2003). "The neural basis of economic decision-making in the Ultimatum Game". Science. 300 (5626): 1755–8. Bibcode: 2003Sci...300.1755S. doi: 10.1126/science.1082976. PMID 12805551. S2CID 7111382.
- ^ Phan KL, Wager T, Taylor SF, Liberzon I (June 2002). "Functional neuroanatomy of emotion: a meta-analysis of emotion activation studies in PET and fMRI". NeuroImage. 16 (2): 331–48. doi: 10.1006/nimg.2002.1087. PMID 12030820. S2CID 7150871.
- ^ Singer T (2006). "The neuronal basis and ontogeny of empathy and mind reading: review of literature and implications for future research". Neurosci Biobehav Rev. 30 (6): 855–63. doi: 10.1016/j.neubiorev.2006.06.011. PMID 16904182. S2CID 15411628.
- ^ Ortigue S, Grafton ST, Bianchi-Demicheli F (August 2007). "Correlation between insula activation and self-reported quality of orgasm in women". NeuroImage. 37 (2): 551–60. doi: 10.1016/j.neuroimage.2007.05.026. PMID 17601749. S2CID 3377994.
- ^ Quarto, Tiziana; Blasi, Giuseppe; Maddalena, Chiara; Viscanti, Giovanna; Lanciano, Tiziana; Soleti, Emanuela; Mangiulli, Ivan; Taurisano, Paolo; Fazio, Leonardo (2016-02-09). "Association between Ability Emotional Intelligence and Left Insula during Social Judgment of Facial Emotions". PLOS ONE. 11 (2): e0148621. Bibcode: 2016PLoSO..1148621Q. doi: 10.1371/journal.pone.0148621. ISSN 1932-6203. PMC 4747486. PMID 26859495.
- ^ Wager, Tor (June 2002). "Functional Neuroanatomy of Emotion: A Meta-Analysis of Emotion Activation Studies in PET and fMRI". NeuroImage. 16 (2): 331–48. doi: 10.1006/nimg.2002.1087. PMID 12030820. S2CID 7150871.
- ^ Vilares I, Howard JD, Fernandes HL, Gottfried JA, Kording KP (2012). "Differential Representations of Prior and Likelihood Uncertainty in the Human Brain". Current Biology. 22 (18): 1641–1648. doi: 10.1016/j.cub.2012.07.010. PMC 3461114. PMID 22840519.
- ^ Craig, A. D. (Bud) (2009). "How do you feel — now? The anterior insula and human awareness" (PDF). Nature Reviews Neuroscience. 10 (1): 59–70. doi: 10.1038/nrn2555. PMID 19096369. S2CID 2340032. Archived from the original (PDF) on 2013-01-07.
- ^ Craig, A. D. (Bud) (2002). "A new view of pain as a homeostatic emotion" (PDF). Trends in Neurosciences. 26 (6): 303–307. doi: 10.1016/s0166-2236(03)00123-1. PMID 12798599. S2CID 19794544. Archived from the original (PDF) on 2010-06-22. Retrieved 2009-09-03.
- ^ Lazar SW, Kerr CE, Wasserman RH, Gray JR, Greve DN, Treadway MT, McGarvey M, Quinn BT, Dusek JA, Benson H, Rauch SL, Moore CI, Fischl B (2005). "Meditation experience is associated with increased cortical thickness". NeuroReport. 16 (17): 1893–7. doi: 10.1097/01.wnr.0000186598.66243.19. PMC 1361002. PMID 16272874.
- ^ Fox, Kieran C.R.; Nijeboer, Savannah; Dixon, Matthew L.; Floman, James L.; Ellamil, Melissa; Rumak, Samuel P.; Sedlmeier, Peter; Christoff, Kalina (June 2014). "Is meditation associated with altered brain structure? A systematic review and meta-analysis of morphometric neuroimaging in meditation practitioners". Neuroscience & Biobehavioral Reviews. 43: 48–73. doi: 10.1016/j.neubiorev.2014.03.016. PMID 24705269. S2CID 207090878.
- ^ Hölzel, Britta K.; Ott, Ulrich; Gard, Tim; Hempel, Hannes; Weygandt, Martin; Morgen, Katrin; Vaitl, Dieter (2008). "Investigation of mindfulness meditation practitioners with voxel-based morphometry". Social Cognitive and Affective Neuroscience. 3 (1): 55–61. doi: 10.1093/scan/nsm038. PMC 2569815. PMID 19015095.
- ^ Damasio, A.; Damasio, H.; Tranel, D. (2013). "Persistence of Feelings and Sentience after Bilateral Damage of the Insula". Cerebral Cortex. 23 (4): 833–846. doi: 10.1093/cercor/bhs077. PMC 3657385. PMID 22473895.
- ^ Taylor KS, Seminowicz DA, Davis KD (September 2009). "Two systems of resting state connectivity between the insula and cingulate cortex". Hum Brain Mapp. 30 (9): 2731–45. doi: 10.1002/hbm.20705. PMC 6871122. PMID 19072897. S2CID 12917288.
- ^ Menon, Vinod; Uddin, Lucina Q. (2010-05-29). "Saliency, switching, attention and control: a network model of insula function". Brain Structure and Function. 214 (5–6): 655–667. doi: 10.1007/s00429-010-0262-0. ISSN 1863-2653. PMC 2899886. PMID 20512370.
- ^ a b Eckert MA, Menon V, Walczak A, Ahlstrom J, Denslow S, Horwitz A, Dubno JR (2009). "At the heart of the ventral attention system: the right anterior insula". Hum. Brain Mapp. 30 (8): 2530–41. doi: 10.1002/hbm.20688. PMC 2712290. PMID 19072895.
- ^ Billeke, Pablo; Ossandon, Tomas; Perrone-Bertolotti, Marcela; Kahane, Philippe; Bastin, Julien; Jerbi, Karim; Lachaux, Jean-Philippe; Fuentealba, Pablo (1 June 2020). "Human Anterior Insula Encodes Performance Feedback and Relays Prediction Error to the Medial Prefrontal Cortex". Cerebral Cortex. 30 (7): 4011–4025. doi: 10.1093/cercor/bhaa017. PMID 32108230.
- ^ a b Zhang, Yang; Zhou, Wenjing; Wang, Siyu; Zhou, Qin; Wang, Haixiang; Zhang, Bingqing; Huang, Juan; Hong, Bo; Wang, Xiaoqin (2019-02-01). "The Roles of Subdivisions of Human Insula in Emotion Perception and Auditory Processing". Cerebral Cortex. 29 (2): 517–528. doi: 10.1093/cercor/bhx334. ISSN 1047-3211. PMID 29342237. S2CID 36927038.
- ^ Nieuwenhuys, Rudolf (2012-01-01), Hofman, Michel A.; Falk, Dean (eds.), "Chapter 7 - The insular cortex: A review", Progress in Brain Research, Evolution of the Primate Brain, 195, Elsevier: 123–163, doi: 10.1016/B978-0-444-53860-4.00007-6, ISBN 978-0-444-53860-4, PMID 22230626, retrieved 2023-12-11
- ^ Bamiou, Doris-Eva; Musiek, Frank E; Luxon, Linda M (2003-05-01). "The insula (Island of Reil) and its role in auditory processing: Literature review". Brain Research Reviews. 42 (2): 143–154. doi: 10.1016/S0165-0173(03)00172-3. ISSN 0165-0173. PMID 12738055. S2CID 22339177.
- ^ Blenkmann, Alejandro O.; Collavini, Santiago; Lubell, James; Llorens, Anaïs; Funderud, Ingrid; Ivanovic, Jugoslav; Larsson, Pål G.; Meling, Torstein R.; Bekinschtein, Tristan; Kochen, Silvia; Endestad, Tor (December 2019). "Auditory deviance detection in the human insula: An intracranial EEG study". Cortex. 121: 189–200. doi: 10.1016/j.cortex.2019.09.002. hdl: 10852/75077. PMID 31629197. S2CID 202749677.
- ^ Afif, Afif; Minotti, Lorella; Kahane, Philippe; Hoffmann, Dominique (November 2010). "Anatomofunctional organization of the insular cortex: A study using intracerebral electrical stimulation in epileptic patients: Functional Organization of the Insula". Epilepsia. 51 (11): 2305–2315. doi: 10.1111/j.1528-1167.2010.02755.x. PMID 20946128. S2CID 2506125.
- ^ Nestor PJ, Graham NL, Fryer TD, Williams GB, Patterson K, Hodges JR (November 2003). "Progressive non-fluent aphasia is associated with hypometabolism centred on the left anterior insula". Brain. 126 (Pt 11): 2406–18. doi: 10.1093/brain/awg240. PMID 12902311.
- ^ Gorno-Tempini ML, Dronkers NF, Rankin KP, et al. (March 2004). "Cognition and anatomy in three variants of primary progressive aphasia". Annals of Neurology. 55 (3): 335–46. doi: 10.1002/ana.10825. PMC 2362399. PMID 14991811.
- ^ Nasir H. Naqvi; David Rudrauf; Hanna Damasio; Antoine Bechara. (January 2007). "Damage to the Insula Disrupts Addiction to Cigarette Smoking". Science. 315 (5811): 531–4. Bibcode: 2007Sci...315..531N. doi: 10.1126/science.1135926. PMC 3698854. PMID 17255515.
- ^ Vorel SR, Bisaga A, McKhann G, Kleber HD (July 2007). "Insula damage and quitting smoking". Science. 317 (5836): 318–9, author reply 318–9. doi: 10.1126/science.317.5836.318c. PMID 17641181. S2CID 8917168.
- ^ Suner-Soler, R. (2011). "Smoking Cessation 1 Year Poststroke and Damage to the Insular Cortex". Stroke. 43 (1): 131–136. doi: 10.1161/STROKEAHA.111.630004. PMID 22052507.
- ^ Gaznick, N. (2013). "Basal Ganglia Plus Insula Damage Yields Stronger Disruption of Smoking Addiction Than Basal Ganglia Damage Alone". Nicotine. 16 (4): 445–453. doi: 10.1093/ntr/ntt172. PMC 3954424. PMID 24169814.
- ^ Hyman, Steven E. (2005-08-01). "Addiction: A Disease of Learning and Memory". Am J Psychiatry. 162 (8): 1414–22. doi: 10.1176/appi.ajp.162.8.1414. PMID 16055762.
- ^ Marco Contreras; Francisco Ceric; Fernando Torrealba (January 2007). "Inactivation of the Interoceptive Insula Disrupts Drug Craving and Malaise Induced by Lithium". Science. 318 (5850): 655–8. Bibcode: 2007Sci...318..655C. doi: 10.1126/science.1145590. hdl: 10533/135157. PMID 17962567. S2CID 23499558.
- ^ Picard, Fabienne (2013), "State of belief, subjective certainty and bliss as a product of cortical dysfuntion", Cortex, 49 (9): 2494–2500, doi: 10.1016/j.cortex.2013.01.006, PMID 23415878, S2CID 206984751
- ^ Gschwind, Markus; Picard, Fabienne (2016), "Ecstatic Epileptic Seizures: a glimpse into the multiple roles of the insula", Frontiers in Behavioral Neuroscience, 10: 21, doi: 10.3389/fnbeh.2016.00021, PMC 4756129, PMID 26924970
- ^ Picard 2013, p.2496-2498
- ^ Picard 2013, p.2497-2498
- ^ See also satori in Japanese Zen
- ^ a b Picard 2013, p.2498
- ^ Paulus MP, Stein MB (August 2006). "An insular view of anxiety". Biol. Psychiatry. 60 (4): 383–7. doi: 10.1016/j.biopsych.2006.03.042. PMID 16780813. S2CID 17889111.
- ^ Thayer JF, Lane RD (December 2000). "A model of neurovisceral integration in emotion regulation and dysregulation". J Affect Disord. 61 (3): 201–16. doi: 10.1016/S0165-0327(00)00338-4. PMID 11163422.
- ^ Gaudio S, Wiemerslage L, Brooks SJ, Schiöth HB (2016). "A systematic review of resting-state functional-MRI studies in anorexia nervosa: Evidence for functional connectivity impairment in cognitive control and visuospatial and body-signal integration" (PDF). Neurosci Biobehav Rev. 71: 578–589. doi: 10.1016/j.neubiorev.2016.09.032. PMID 27725172. S2CID 16526824.
- ^ a b Binder DK, Schaller K, Clusmann H (November 2007). "The seminal contributions of Johann-Christian Reil to anatomy, physiology, and psychiatry". Neurosurgery. 61 (5): 1091–6, discussion 1096. doi: 10.1227/01.neu.0000303205.15489.23. PMID 18091285. S2CID 8152708.
External links
- Insular cortex in the Brede Database at the Technical University of Denmark. Location and literature citations for the insula
- synd/1212 at Who Named It?
- "Anatomy diagram: 13048.000-1". Roche Lexicon - illustrated navigator. Elsevier. Archived from the original on 2014-11-07.
- Stained brain slice images which include the "insular cortex" at the BrainMaps project
- Thomas P. Naidicha; et al. (1 February 2004). "The Insula: Anatomic Study and MR Imaging Display at 1.5 T". American Journal of Neuroradiology. 25 (2): 222–32. PMC 7974606. PMID 14970021.
- Kakigia R, Nakataa H, Inuia K, Hiroea N, et al. (October 2005). "Intracerebral pain processing in a Yoga Master who claims not to feel pain during meditation". Eur J Pain. 9 (5): 581–9.
doi:
10.1016/j.ejpain.2004.12.006.
PMID
16139187.
S2CID
17598717.
As for fMRI recording, there were remarkable changes in levels of activity in the...SII-insula (mainly the insula)